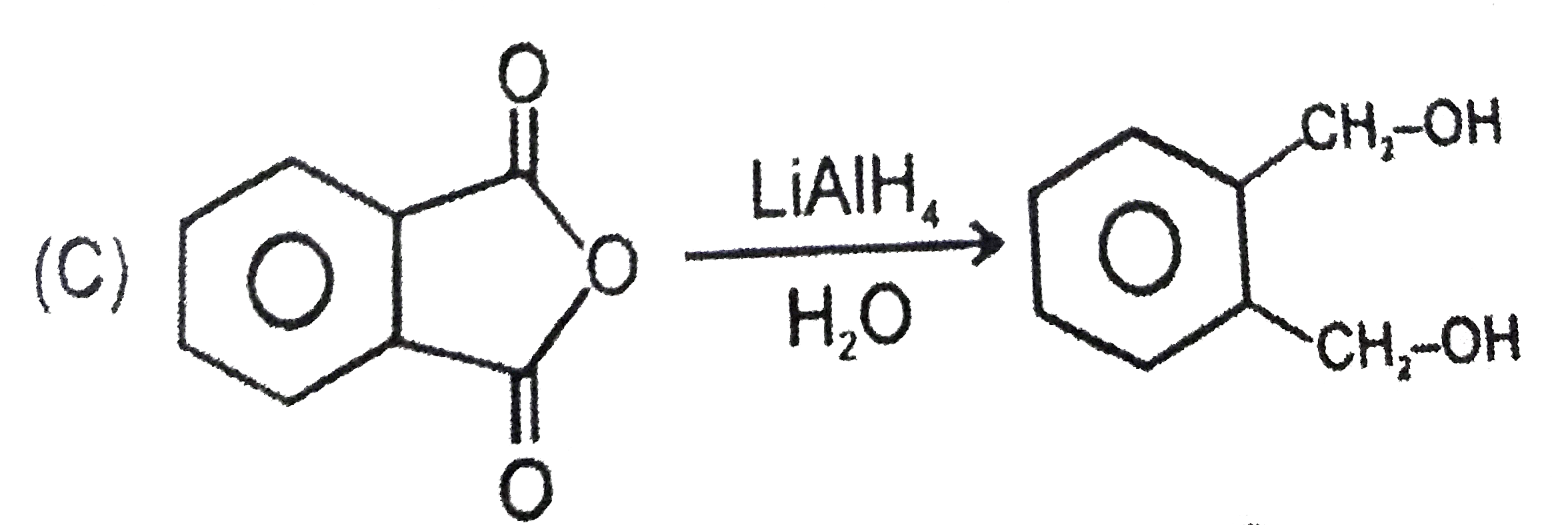
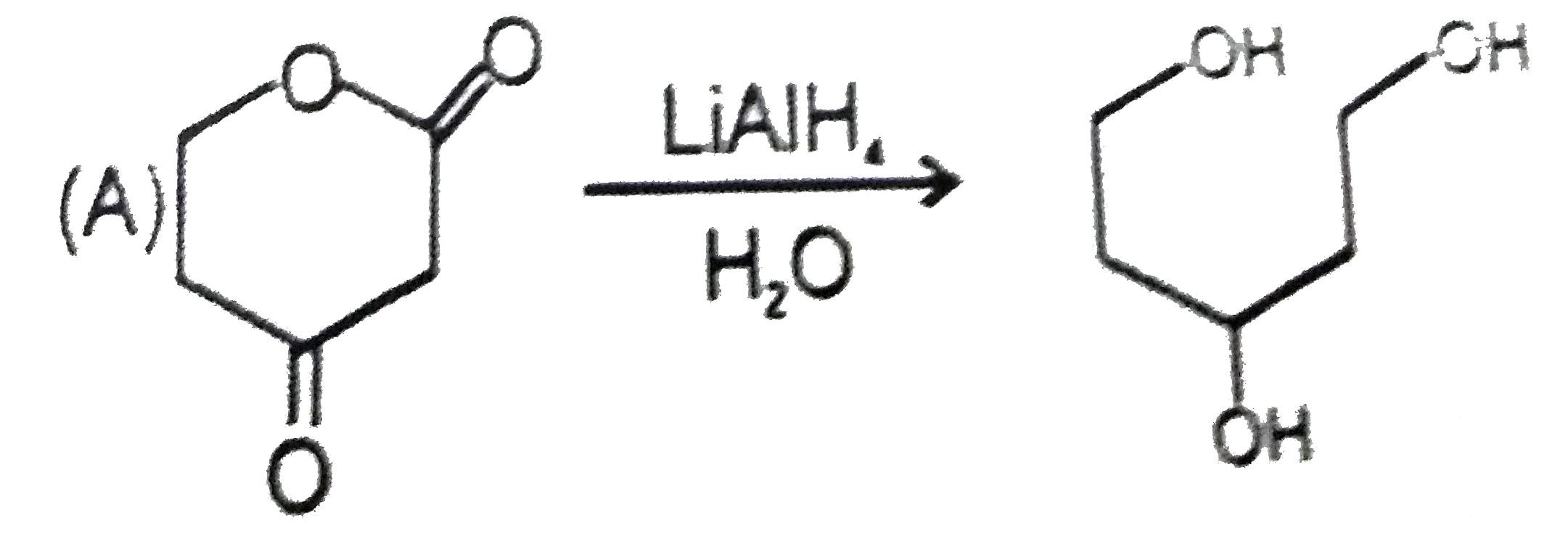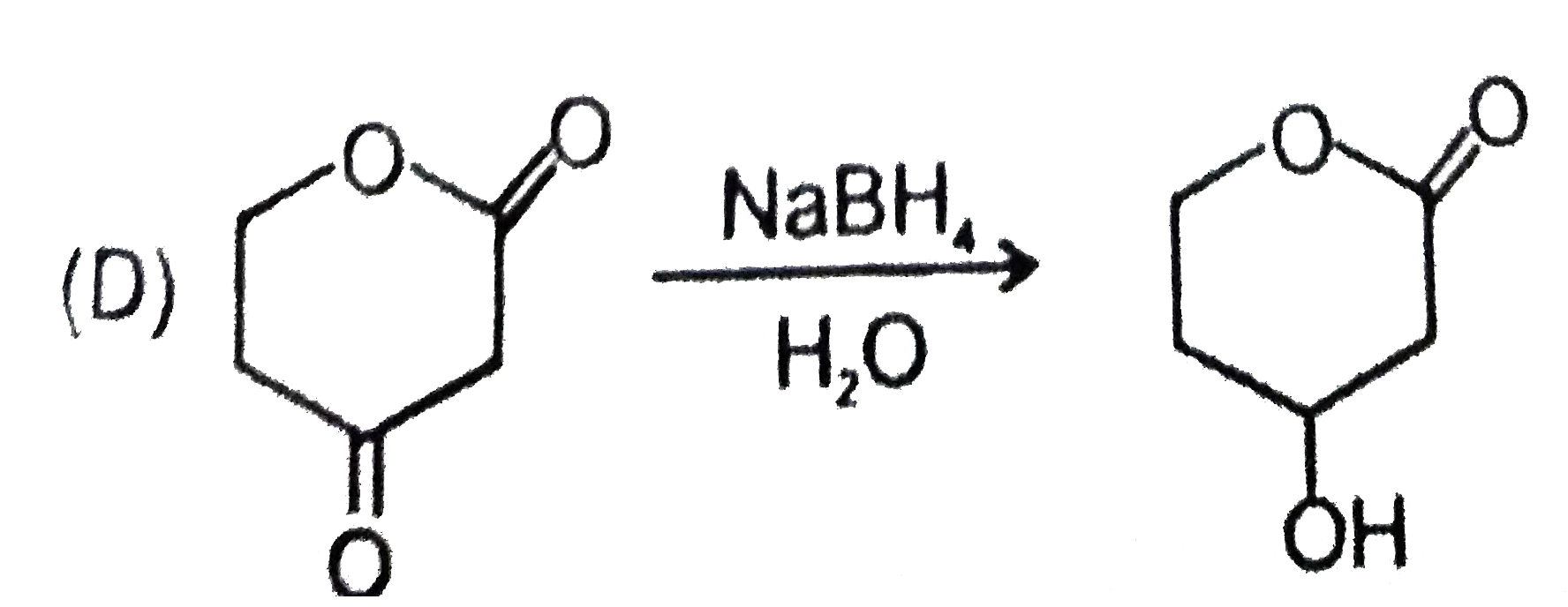Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
What do you mean by ozone hole? What are its consequences? |
|
Answer» SOLUTION :Depletion of ozone LAYER creates some sort of holes in the blanket of ozone which SURROUNDS us in the atmosphere and this is known as ozone hole. (i) With the depletion of the ozone layer, UV radiations filters into the troposphere which leads to ageing of SKIN, cataract, sunburn, skin cancer etc. (ii) By killing many of the phytoplanktons, it can damage the fish productivity. (III) Evaporation rate increases through the surface and stomata of leaves which can decrease the moisture content of the soil. |
|
| 2. |
Whatis not same in ._(17)^(35) CI and ._(17)^(37) CI ? |
|
Answer» ELECTRON |
|
| 3. |
Which of the following is//are matched properly ? |
|
Answer» `H_(2)O_(2)+2OH^(-)to2H_(2)O+O_(2)+2e^(-)` …….. REDUCING property in alkaline medium |
|
| 5. |
Write the K_p and K_c for the following reactions. (i) 2SO_(2)(g)+ O_(2)(g) hArr 2 SO_(3)(g) (ii) 2CO(g) hArr CO_(2) + C(s) |
|
Answer» Solution :`K_(c) = ([SO_3]^2)/([SO_2]^2[O_2]), K_p = (P_(SO_3)^(2))/(P_(SO_2)^2 . PO_(2))` (ii) C(s) has CONTANT concentration `K_(c)= ([CO_2])/([CO]^2) , K_p = (P_(CO_2))/(P_(CO)^(2))` |
|
| 6. |
Which one of the wrong statement |
|
Answer» The uncertainty principle is `DeltaE xx Deltat ge h//4pi` |
|
| 7. |
When 0.1 mole of BaCl_(2) is treated with 0.5 mole of Na_(3)PO_(4), the maximum number of moles of Barium phosphate formed is |
|
Answer» 0.25 |
|
| 8. |
Which of the following statements is wrong ? |
|
Answer» Ozone is not responsible for greenhouse effect. |
|
| 9. |
Write the expression of K_(p) for the reaction. N_(2)O(g)hArr2NO_(2)(g) |
|
Answer» <P> Solution :`K_(p)=((P_(NO_(2))))/((P_(N_(2)O_(4))))` |
|
| 10. |
Which is more acidic, acetylene or benzene? |
|
Answer» SOLUTION :Acetylene is more ACIDIC than BENZENE as `CH -= C^(Theta)` is more stable than 
|
|
| 11. |
Which oxide of nitrogen is not common pollutant ? |
|
Answer» `NO_(2)` |
|
| 12. |
The reddish brown coloured gas formed when nitric oxide is oxidised by air is |
| Answer» Solution :`2NO+O_(2)to2NO_(2)` | |
| 13. |
Write two uses of PCBs. |
| Answer» SOLUTION :(i) As a FERTILIZER (II) As a DETERGENT in CLEANING | |
| 14. |
Write self ionization reaction and ionic product of water. |
|
Answer» Solution :The self-ionization REACTION of water at `25^@C` is as FOLLOWING. `H_2O_((l)) + H_2O_((l)) HARR H_3O_((AQ))^(+) + OH_((aq))^(-)` `K_w=[H_3O^+][OH^-]=1.0xx10^(-14)` |
|
| 15. |
Which of the following reactions result in the formation of conjugated diene? |
|
Answer» TREATING vinyl BROMIDE with `(CH_2=CH)_2` CuLi |
|
| 16. |
Which of the following molecule(s) does not have existence? |
|
Answer» `Ibr_(7)` |
|
| 17. |
Two substances when separated on the basis of partition coefficient between two liquid phases, the technique is known as |
|
Answer» Column CHROMATOGRAPHY |
|
| 18. |
Which one of the following statement is is incorrect |
|
Answer» `H_(2)` REACTS with `Cl_(2)` to form HCl, an electron pair SHARED between H and Cl |
|
| 19. |
What atoms are indicated by the following eletronic configurations ? (i) 1s^(2) 2s^(2) 2p^(1)(ii) [Ar] 4s^(2) 3d^(1) |
|
Answer» Solution :(i) Total number of electrons in `1s^(2) 2s^(2) 2p^(1) =2+2+1=5 :.` Atomic number of the element `=5` Hence, the element is Boron (B) (II) Total number of electrons in `[AR] 4s^(2) 3D^(1)=18+2+1=21 :.` Atomic number of the element `=21` Hence, the element is Scandium (SC) |
|
| 20. |
What are fumes? Give one example. |
| Answer» Solution :Fumes are one of the non-viable particulate pollutants air. They are obtained by condensation of VAPOURS released during SUBLIMATION, DISTILLATION, boiling and calcination. For example, ORGANIC solvents, metals and metallic oxides form fume particles. | |
| 21. |
Which would you expect to have a higher melting point, magnesium oxide or magnesium fluoride? Explain your reasoning. |
|
Answer» Solution :(i) Magnesium OXIDE has very strong ionic bonds as compared to magnesium fluoride. (ii) `Mg^(2+) and O^(2-)` have charges of +2 and -2, respectively. (iii) Oxygen ion is smaller than fluoride ion. (iv) The smaller the ionic radii, the smalller the bonds LENGTH in MGO and the bond is STRONGER than `MgF_(2)`. (V) Due to more strong bond nature in MgO, it has high melting point than `MgF_(2)` |
|
| 22. |
What is the ratio of the Rydberg constant for helium to Hydrogen atom? |
|
Answer» `1//2` `(R_(He))/(R_(H))=(2xx2^2)/(1xx1^(2))=8` `therefore(R_(H))/(R_(He))=(1)/(8)`. |
|
| 23. |
Which elements are consider as alkaline earth metals? Give brief about them. |
|
Answer» Solution :The group-2 ELEMENTS comprise beryllium, magnesium, calcium, STRONTIUM, barium and radium. They follow alkali METALS in the PERIODIC table. These (except beryllium) are known as alkaline earth metals. The first element beryllium differs from the rest of the members and shows diagonal relationship to ALUMINIUM.  `*ppm (part per million), ** Percentage by weight |
|
| 24. |
When trans-2-butane reacts with Br_2//C Cl_4, X number of products are formed. Whereas when trans-2-butene reacts with HBr Y number of products are formed . Report your answer as Y X . |
|
Answer» |
|
| 25. |
Write two uses of green chemistry in routine life. |
|
Answer» Solution :(1) For drycleaning of cloths. (2) For the bleaching of PAPER. (3) In the synthesis of chemical. |
|
| 26. |
When bauxite powder is mixed with coke and reacted with nitrogen at 2075 K, carbon monoxide and X are formed. What is the gas formed when X is reacted with water ? |
|
Answer» `NH_(3)` `Al_(2)O_(3) + 3C + N_(2) rarr 2AIN + 3CO uarr` `AIN + 3H_(2)O rarr Al(OH)_(3)+NH_(3)` |
|
| 27. |
What sorts of informations can you draw from the following reaction ? (CN)_(2(g))+2OH_((g))^(-)toCN_((aq))^(-)+CNO_((aq))^(-)+H_(2)O_((l)) |
Answer» Solution :Oxidation number of C in `(CN)_(2),CN^(-)andCNO^(-)` is given below :  In a given reaction oxidation number of C is DIFFERENT. `overset(+3)((CN))_(2(g))+2OH_((g))^(-)tooverset(+2)(CN_((AQ))^(-))+overset(+4)(CNO_((aq))^(-))+H_(2)O_((l))` In a disproportionation reaction an element in one oxidation state is simultaneously oxidised and reduced. So, we can say that dissolution of cyanide in basic medium is DISPROPORTIONATE reaction. |
|
| 28. |
What are free radical initiators? |
| Answer» Solution :The types of reagents that PROMOTE homolytic cleavage in substrate are CALLED as free radical initiators. They are short LIVED and are HIGHLY REACTIVE. | |
| 29. |
Which is a bigger ion among Na^(+), F^(-), O^(2-) and Mg^(2+)? Why? |
|
Answer» SOLUTION :`O^(2-)` is BIGGER ion AMONG the given four. Among isoelectronic IONS, the more the negative CHARGE on the ion, the more its size. The order is `O^(2-) gt F^(-) gt Na^(+) gt Mg^(2+)` |
|
| 30. |
Write Lewis dot symbols for atoms of the following elements: Mg, Na, B, O, N, Br. |
|
Answer» Solution :`""_(12)MG=2,8,2" "therefore` Lewis symbol = Mg. `""_(11) N_(a) = 2,8,1" "therefore` Lewis symbol = Na `""_(5)B = 2,3" "therefore` Lewis symbol `= *overset(*)(B*)` `""_(8)O = 2,6""therefore`Lewis symbol `= : overset(* *)B:` `""_(7)N = 2,5" "therefore`Lewis symbol `= : UNDERSET(*)overset(*)N*` `""_(35)Br = 2,8, 18, 7" "therefore` Lewis symbol `= :underset(* *)overset(* *)Br` |
|
| 31. |
Write the conjugate acids for the following Bronsted bases : NH_2^(-) , NH_3 and HCOO^-. |
|
Answer» Solution :Conjugate acid =BASE + `H^+` The conjugate acid should have ONE extra PROTON and +1 CHARGE them base. Conjugate acid of`NH_2^(-) =(NH_2^(-) + H^+) = NH_3` Conjugate acid of `NH_3=(NH_3+H^(+))=NH_4^(+)` Conjugate acid of `HCOO^(-) = (HCOO^(-) + H^+)` =HCOOH |
|
| 32. |
When the resonance structure represented ? |
|
Answer» SOLUTION :(i) If MOLECULE has alternate MULTIPLE BOND (ii) The atom which has double bond possess lone PAIR |
|
| 33. |
What is the principle of distillation? |
| Answer» Solution :EVERY pure LIQUID has a definite BOILING POINT | |
| 34. |
Which of the following statements is correct regarding ease of dehydration ? |
|
Answer» Primary `GT` Secondary |
|
| 35. |
What product(s) wouldyou expect from the following solvolysis ? |
Answer» SOLUTION :We OBSERVE that this cyclohexyl bromide is tertiary, and therefore in methanol it should lose a bromide ion to FORM a tertiary carbocation. Because the carbocation is trigonal planar at the positive CARBON, it can react with a solvent molecule (methanol) to form two PRODUCTS. 
|
|
| 36. |
Which of the following reaction is/are correct ? |
|
Answer»
|
|
| 37. |
The volutme of oxygen liberated from 15 ml of 20 volume is |
|
Answer» Solution :1 ml ----- 20 ml 15 ml ----------? VOLUME `=20 times 15=300ml` |
|
| 38. |
Which is the pH of 10^(-8) M NaOH from 8.0, 6.0 and 7.05 ? Why ? |
| Answer» SOLUTION :NaOH is BASE. So the pH of BASIC solution is more than 7.0. So it is 7.05 and POH = 6.95. | |
| 40. |
Which of the following molecules have zero dipole moment ? cis-1,2-dichloroethene, trans-1,2-dichloroethene, 1,1-dichloroethene, trans-2-pentene, cis-2-pentene, 1-butyne, 2-butyne , trans-2-butene, cis-2-butene. |
|
Answer» |
|
| 41. |
What are oxidation and reduction organic reactions? Give an example. |
|
Answer» SOLUTION :Oxidation reaction : Most of the oxidation reaction of organic compounds involves GAIN of oxygen or loss of hydrogen. E.g. : `underset("(acetaldehyde)")(CH_3CHO) underset(H^+ |K_2Cr_2O_7)OVERSET((O))to underset("(Acetic ACID)")(CH_3COOH)` REDUCTION reactions : Most of the reduction of organic compounds involves gain of hydrogen or loss of oxygen. 
|
|
| 42. |
Which of the following represents aromatic alcohol? |
|
Answer» Phenol 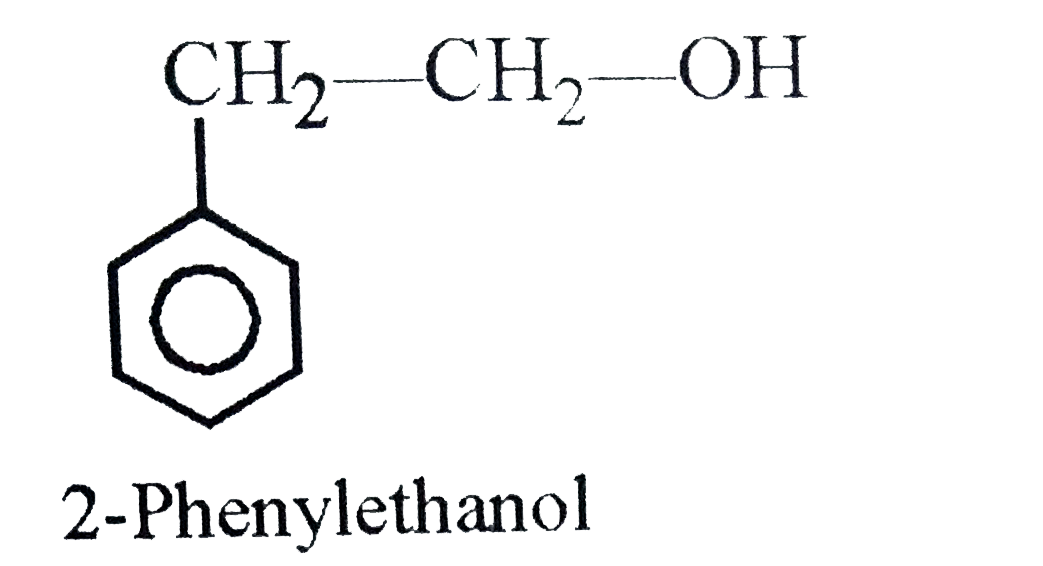
|
|
| 43. |
What happens when sodium carbonate undergoes hydrolysis? |
|
Answer» Solution :Sodium carbonate is a salt of a strong base like NaOH and WEAK acid like `H_(2) CO_(3), Na_(2) CO_(3)` undergoes hydrolysis in water to form an alkaline solution. `Na_(2) CO_(3) + H_(2) O to H_(2) CO_(3) + NaOH` |
|
| 44. |
What is the maximum number of orbitals that can be identified with the following quantum number n = 3, I = 1, m = 0. |
|
Answer» 1 So it should be `3P_(Z)` |
|
| 45. |
Write the structure of cis and trans isomer of But-2-ene. |
Answer» SOLUTION : 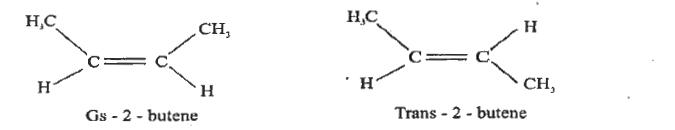
|
|
| 46. |
What happens when HCOOH is heated at 373K with conc. H_2SO_4 ? |
| Answer» | |
| 47. |
What volume of 0.2 M Ba( MnO_(4))_2solution is required for complete oxidation of 25 gm of 89.6% pure Fe Co, in acidic medium according to the reaction MnO_(4)^(-) +FeCr_(2)O_(4) rarrFe^(+3) +Cr_(2)O_(7)^(2-)+Mn^(+2) |
|
Answer» 700 ml n-factor of `Ba(MnO_(4))_(2) = 10` n-factor of `FeCr_(2)O_(4)= 7 ` `:. V xx0.2xx10=(25xx89.6xx10^(3))/(100xx((224)/7))` `:. V=350 ml` |
|
| 48. |
Which has more surface tension from the following ? (i) H_(2)O (ii) Soap |
| Answer» SOLUTION :Surface tension of SOAP solution is less than `H_(2)O` because solubility INCREASES, surface tension increases. | |
| 49. |
Ultraviolet radiation is absorbed by |
|
Answer» exosphere |
|
| 50. |
Which of the following reacts with nitrogen when heated in air Anomalous propaerrties |
|
Answer» Al |
|