Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
What are the uses of methylene chloride? |
|
Answer» SOLUTION :Methylene chloride is USED as: (i) Aerosol spray propellant. (II) Solvent in paint REMOVER. (iii) Process solvent in the manufacture of drugs. (iv) A metal cleaning agent. |
|
| 2. |
Which of the following is least likely to behave as Lewis base ? |
|
Answer» `H_2O` |
|
| 3. |
What could be the harmful effects of improper management of industrial and domestic solid waste in a city ? |
|
Answer» Solution :All the solid wastes either domestic or INDUSTRIAL are of two types : (i) Biodegradable (ii) Non-biodegradable If domestic waste in a city is not PROPERLY managed, it MAY find its way into sewers or may be eaten up by the cattle. The polythene bags, if swallowed by the cattle, can result into their death. SIMILARLY, if industrial waste is not properly managed, it will cause POLLUTION of the air, soil and water. |
|
| 4. |
Who had given explanation of chemical bonding with respect to electron ? |
| Answer» SOLUTION :KOSSEL and LEWIS | |
| 5. |
What are X and Y in the reaction? 3B_(2)H_(6) + 6X to 3[BH_(2) (X)_(2)]^(+) [BH_(4)]^(-) overset("heat")(to) Y + 12 H_(2) |
|
Answer» `X = NH_(3), Y = B_(3)N_(3)H_(6)` 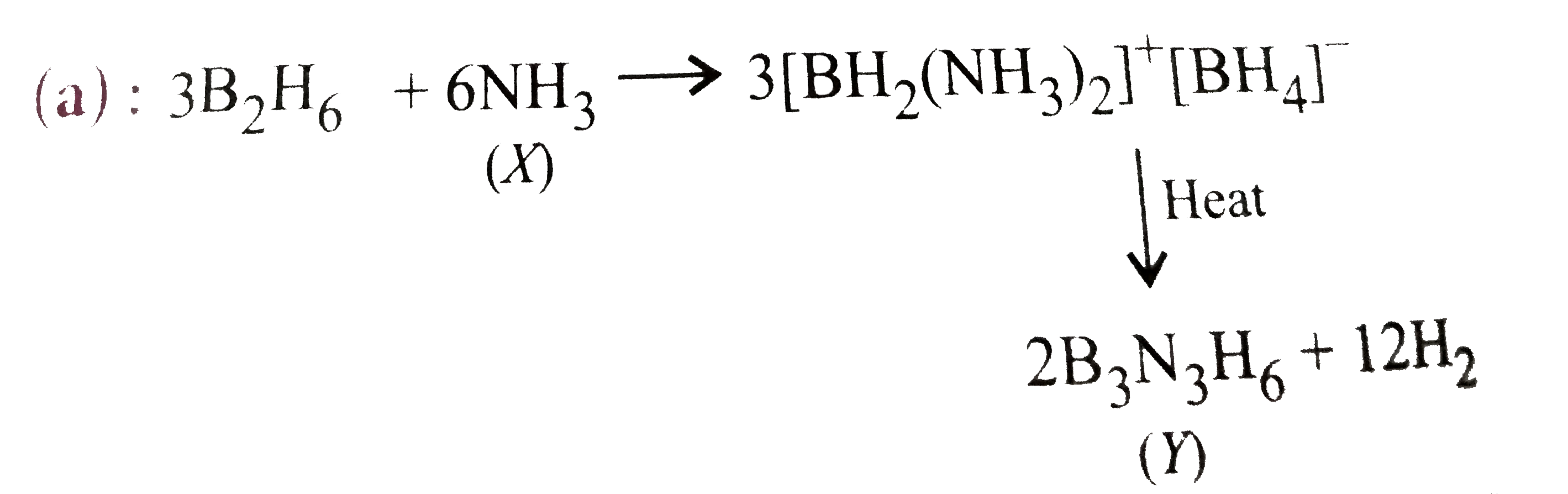
|
|
| 6. |
The substance used as a smoke screen in warfare is |
|
Answer» `SiCl_4` |
|
| 7. |
Which statement is wrong for NO ? |
|
Answer» It is anhydride of nitrous acid |
|
| 8. |
Which of the following reagent on reaction with conc. NaOh followed by H^(oplus)gives following easter ? |
|
Answer» |
|
| 9. |
When KMnO_(4) acts as an oxidising agent and ultimately forms MnO_(4)^(-2),MnO_(2),Mn_(2)O_(3) and Mn^(+2) then the number of electrons transferred in each case respectively is |
|
Answer» 4,3,1,5 |
|
| 11. |
which of the following stateement is coorect ? |
|
Answer» Lanthanum is the firstelement of Lanthanide |
|
| 12. |
Which Q_(sp) will be high from 0.0001 M BaCrO_4 and 0.0001 M Ag_2CrO_4 ? |
|
Answer» SOLUTION :`Q_(sp) (BaCrO_4)=(0.0001)^2=1xx10^(-8)` `Q_(sp)(Ag_2CrO_4)=4S^3=4(0.0001)^3=4xx10^(-12)` THEREFORE `Q_(sp)` of `BaCrO_4` is less. |
|
| 13. |
What is the relationship between K_(w)andpK_(w). |
| Answer» SOLUTION :`pK_(W)=-log_(10)K_(w)" at "298K,pK_(w)` of water is 14. | |
| 15. |
Which are Homogenous and Heterogenous equilibrium ? (a)H_(2(g)) + I_(2(g)) hArr 2HI_((g)) (b)Ca(OH)_(2(s)) hArr Ca_((aq))^(2+) + 20H_((aq))^(-) (c ) CaCO_(3(s)) hArr CaO_((s))+CO_(2(aq)) (d)Ag_2O_((cs))+2H_((aq))^(+) hArr H_2O_((l)) + 2Ag_((aq))^(+) (e) NH_(3(g))+H_2O_((aq)) hArr NH_(4(aq))^(+) + OH_((aq))^(-) (f)CH_3COOC_2H_(5(l)) + H_2O_((l)) hArr CH_2COOH_((l)) +C_2H_5OH_((l)) |
|
Answer» Solution :Homogenous : (a),(E) ,(F) Heterogenous : (B), (C) , (d) |
|
| 16. |
Which of the following sets of quantum numbers is correct for an electron is 4f - orbital ? |
|
Answer» N = 4, l = 3, m = 4, s = +1/2 |
|
| 17. |
Write the atomic number of the element, present in the third period and seventeenth group of the periodic table. |
|
Answer» |
|
| 18. |
The relative reactivity of 1^(@), 2^(@), 3^(@), hydrogen's towards chlorination is 1:3.8:5. Calculate the percentage of all mono chlorinated products obtained from 2-methylbutane. |
|
Answer» Solution :In 2-methyl butane, there are 9 primary hdyrogen, 2 SECONDARY and 1 tertiaryhydrogen ATOMS. The realtivere activity of `1^(@), 2^(@)` and `3^(@)` hydrogen atoms towards chlorination is 1:3.8:5. RELATIVE amount of product after chlorination = number of hydrogen atom C relative REACTIVITY. `{:("Relative",1^(@) "halide",2^(@) "halide",3^(@) "halide",),("Amount",9 xx 1 = 9,2 xx 3.8 = 7.6,1 xx 5 = 5,):}` Total amount of monochloro product = 9+7.6 +5 = 21.6 Percentage of `1^(@)` monochloro product `= 9 xx 100/21.6 = 41.7%` Percentage of `2^(@)` monochloro product `= 7.6 xx 100//21.6 = 35.2%` Percentage of `3^(@)` monochloro product `= 5 xx 100//21.6 = 23.1%` |
|
| 19. |
What is diagonal relationship due to ? |
| Answer» Solution :Elements having similar size of ATOMS or ions , similar electronegativity or similar polarising power show diagonal RELATIONSHIP . For example , Li and Mg , and Be and AL have MANY similar properties . | |
| 20. |
What is vapour pressure ? "In open vessel rate of vaporization is constant still equilibrium is not establish" - Explain. |
|
Answer» Solution :At definite temperature in CLOSE vessel, equilibrium is established between liquid and its vapour. At this TIME the pressure of vapour is called .vapour pressure of that liquid.. Equilibrium is possible in close vessel only and at equilibrium rate of vaporisation and condensation become same. At equilibrium rate of forward and reverse REACTION is also same. In open vessel - It is not possible to reach equilibrium in an open SYSTEM. In watch glass if water is PLACED in open atmosphere than the molecules of liquid vapour disperse in room. So rate of liquid `to`gas is more than rate of gas `to`liquid. So, equilibrium is not established. In this way in open vessel , `"liquid"_((l)) ne "liquid"_((g))`...(i) In open vessel also at constant temperature rate of vaporisation remain constant but equilibrium state is not attain. Condensation rate `lt` vaporisation, both two remain constant. |
|
| 21. |
Which of the following are antioxidants ? |
|
Answer» BHT |
|
| 22. |
Which is wrong according to Kinetic theory? |
|
Answer» The average K.E. of the MOLECULES is DIRECTLY PROPORTIONAL to the absolute temperature. |
|
| 24. |
Which methods are used for removal or reduction of soluble salts and turbidity of water? |
|
Answer» REVERSE osmosis |
|
| 25. |
What is sabatier sendersens reaction |
| Answer» Solution :The processof ADDITIONOF `H_(2)` tounstauratedcompoundin KNOWNAS HYDROGENATION . Theaboveprocesscan becatalysed by nickedat 298 kthisreactionisknownas Sabatier -Sendersens reactionforexample | |
| 26. |
When a salt reacts with water to form acidic (or ) basic solution the process is called salt hydrolysis. The P^(H)of salt solution can be calculated using the following relation P^(H) =(1)/(2)[P^(k_w) +P^(Ka) + log C ] for salt of weak acid and strong base. P^(H)=(1)/(2)[P^(K_w) -P^(K_a) -log C ] for salt of weakbase and strong acid , P^(H) =(1)/(2)[P^(K_w) +P^(K_a) - P^(K_b) ] For a salt of weakacid and weakbase where .c. represents the concentration of salt . When a weak acid (or)a weak base is not completlyneatralised by strong base (or ) strong acid respectively, then formation of buffer takes places. The P^(H)of buffer solution can be calculated using the following relation P^(H)=P^(Ka) +log "" ((["salt")])/(["Acid "]) , P^(OH) =P^(Kb) +log ""(["salt"])/(["base"])Answer the following questions using the following data pK_a (CH_3COOH) =4.7447 , pK_b (NH_4OH) =4.7447 , P^(K_W)=14. One mole CH_3 COOHand one moleCH_3 COONa are dissolved in water one litre aqueous solution The P^(H)of the resulting solution will be |
|
Answer» ` 9.2553` |
|
| 27. |
When a salt reacts with water to form acidic (or ) basic solution the process is called salt hydrolysis. The P^(H)of salt solution can be calculated using the following relation P^(H) =(1)/(2)[P^(k_w) +P^(Ka) + log C ] for salt of weak acid and strong base. P^(H)=(1)/(2)[P^(K_w) -P^(K_a) -log C ] for salt of weakbase and strong acid , P^(H) =(1)/(2)[P^(K_w) +P^(K_a) - P^(K_b) ] For a salt of weakacid and weakbase where .c. represents the concentration of salt . When a weak acid (or)a weak base is not completlyneatralised by strong base (or ) strong acid respectively, then formation of buffer takes places. The P^(H)of buffer solution can be calculated using the following relation P^(H)=P^(Ka) +log "" ((["salt")])/(["Acid "]) , P^(OH) =P^(Kb) +log ""(["salt"])/(["base"])Answer the following questions using the following data pK_a (CH_3COOH) =4.7447 , pK_b (NH_4OH) =4.7447 , P^(K_W)=14. 0.001 M NH_4 Cl aqueous solution has P^(H) |
|
Answer» `6.127` ` = 6.127` |
|
| 28. |
What is the correct reading of a device with precision 1/100 of a unit ? (i) 3.600 g (ii) 3.60 g (iii) 3.6 g |
|
Answer» |
|
| 29. |
Write suitable chemical equations to show the nature of Calcium oxide and carbon dioxide |
Answer» Solution :(i) REACTIONS of acidic OXIDES with alkalis :  (ii) Reactions of AMPHOTERIC oxides with acids and bases :  (iii) Reactions of BASIC oxides with acids : `Tl_2 O_3+ underset("dil.")(3H_2 SO_4) + 4H_2 O to underset("Thallium sulphate heptahydrate")(Tl_2 (SO_4)_3 . 7H_2 O)` |
|
| 30. |
Write the structural formula of: (a) o-ethylanisole (b) p-nitroaniline (c ) 2, 3-dibromo-1-phenylpentane (d) 4-ethyl-1-fluoro-2-nitrobenzene |
Answer» Solution :(a) o-Ethylanisole: `C_(6)H_(5)OCH_(3)` its common name is anisole. So  Note: Generally the substitute ortho positions written in left side. (b) p-nitroaniline: Aniline MEANS `C_(6)H_(5)NH_(2) and -NO_(2)` is at pera position so, `NO_(2) - C_(6)H_(4)- NH_(2)`  (c ) 2, 3-dibromo-1-phenylpentane : In this five carbon containing parent chain, the PHENYL, bromo and bromo substitutes are present at 1, 2 and 3 position respectively and written as under `overset(5)C - overset(4)(C )-underset(underset(Br)(|))overset(3)(C )- underset(underset(Br)(|))overset(2)(C )- underset(underset(C_(6)H_(5))(|))overset(1)(C )`  (d) 4-ethyl-1-fluoro-2-nitrobenzene: 
|
|
| 31. |
What do you expect the nature of Hydrides is, if formed by elements of atomic numbers 15, 19, 23 and 44 with dihydrogen ? Compare their behaviour towards water. |
|
Answer» Solution :(i) The element (Z = 15) is a p-block element so it form covalent HYDRIDE eg. `PH_3` (ii) The element (Z = 19) is a s-block element so, it form ionic hydride e.g. KH (iii) The element (Z = 23) is d-block element. So it form nonstoichiometric hydride. e.g. `VH_(1.6)` (iv) The element (Z = 44) is from d-block and 8 GROUP. So it does not form hydride. Above four hydride, only `H_2` is obtained in reac tion of KH with `H_2O`. `KH + H_2O to KOH + H_2` |
|
| 32. |
The value of K_p for the reaction, CO_(2(g)) + C_((s)) hArr 2CO_((g))is 3.0 at 1000 K. If initially p_(CO_2)= 0.48 bar and p_(CO) = 0 bar and pure graphite is present, calculate the equilibrium partial pressures of CO and CO_2. |
|
Answer» SOLUTION :Suppose, the DECREASE in concentration (pressure) of `CO_2` in reaction = x bar. So, according to stoichiometry of reaction, the increase in pressure of `CO_2` is x bar. `{:("Reaction equilibrium:",CO_(2(g))+ , C_((s)) hArr , 2CO_((g))),("INITIAL pressure :","0.48 bar",-,0),("Change of pressure :","-x bar",-,+ 2x "bar"),("At equilibrium :", (0.48-x)"bar",,2x "bar"):}` For this chemical equilibrium , equilibrium constant `K_p`, `therefore K_p=(P^2CO)/(PCO^2)` where , `K_p`=3.0 `p_(CO)`=2x bar `p_(CO_2)`=(0.48-x) bar `therefore 3.0=(2x)^2/((0.48-x))` `therefore 4x^2=3(0.48-x)=1.44-3x` `therefore 4x^2+3x-1.44=0` `x=(-b pm sqrt(b^2-4ac))/(2a)` So, a=4 , b=3 , C=-1.44 `therefore x=(-3pmsqrt((3)^2-4(4)(-1.44)))/(2(4))` `=(-3pmsqrt(9+23.04))/8=(-3pmsqrt(32.04))/8` `=(-3pm5.660)/8` `=2.66/8` OR `(-8.66)/8` =0.3325 OR -1.0825 The value of x cannot be negative . `therefore` x=0.3325 bar So, `p_(CO)`=2x=2(0.3325)=0.6650 barand `p_(CO_2)` =(0.48-x)=(0.48-0.3325)=0.1475 bar =0.15 bar |
|
| 33. |
Which hydrated elements of group 13 are found as polymer in nature ? |
| Answer» Solution :HYDRATES of Al, Ga and In are FOUND as POLYMER in NATURE. Hydrates of Boron does not found as polymer | |
| 35. |
Which of the following statements is/are correct |
|
Answer» The IUPAC name of amyl alcohol is pentanol. |
|
| 36. |
Which of the following statements (s) is (are) correct ? |
|
Answer» All REACTION are OXIDATION and reduction reactions |
|
| 37. |
What are liquid solutions ? Explain with example. |
Answer» SOLUTION :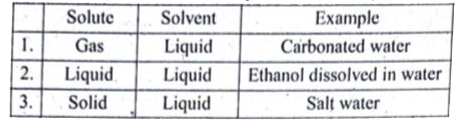
|
|
| 38. |
The wavelength range of the visible spectrum extends from violet (400 nm) to red (750 nm). Express these wavelengths in frequencies(Hz), (1nm=10^(-9)m). |
| Answer» Solution :Frequency of violet light `V=c/lambda=(3.00xx10^8ms^(-1))/(400xx10^(-9))=7.50xx10^14Hz` Frequency of RED light `lambda=c/lambda=(3.00xx10^8ms^(-1))/(750xx10^(-9))=4.0xx10^14Hz` The range of VISIBLE SPECTRUM is from `4.0xx10^14` to `7.5xx10^14` Hz.in terms of frequency UNITS. | |
| 39. |
Which of the following species will have the largest and the smallest size ? Mg, Mg^(2+) , Al , Al^(3+) |
|
Answer» Solution :RADIUS of Mg is maximum and `Al^(3+)` is minium. SIZE of Mg is more and Al is less , because Mg and Al are in same period. In same period we go to left to right atomic radius decreases. So, `Al LT Mg`. Volume of position ions are less than PARENTAL atoms. So , `Mg^(2+) lt Mg " and " Al^(3+) lt Al`. In isoelectroic species, POSITIVE charge increases radius an volume decreases. So, `Al^(+3) lt Mg^(+2)`. `Mg^(+2) ` having Al more nuclear effective charge so, `Mg^(2+) lt Al`. |
|
| 40. |
The type of bonds present in ammonium chloride are |
|
Answer» Only IONIC and dative |
|
| 41. |
Which of the following binds with haemoglobin and reduce the oxygen carrying capacity of blood? |
| Answer» Answer :D | |
| 42. |
Why are potassium and caesium , rather than lithium used in photoelectric cells ? |
| Answer» Solution :Potassium and CAESIUM have much lower IONIZATION enthalpy than that of lithium . As a RESULT , these metals on exposure to light , easily emit electrons but lithium does not . THEREFORE , K and Cs rather than Li are USED in photoelectric cells. | |
| 43. |
Write the functional group of the following compounds (i) Aldehyde (ii) Ester (iii) Ether (iv) alcohol |
|
Answer» SOLUTION :ALDEHYDE `""RCHO""-UNDERSET(O)underset(||)C-H` (ii) ESTER `"" RCOOR^1"" -underset(O)underset(||)OR^I` (iii) Ether `""R-O-R "" -O-` (iv) alcohol `""R-OH ""-OH` |
|
| 44. |
White of an egg is partly coagulated by heating which can be again obtained back by some pepsin and little HCl. This process is called |
|
Answer» Peptization |
|
| 45. |
The total charge (coulombs) required for complete electrolysis is |
|
Answer» 24125 Moles of `Na^+` discharged at cathode =2 `therefore` The no. of ELECTRONS required for this purpuse = 2 moles `therefore` Total CHARGE required = 2 faradays `=2xx96500` =193000 coulombs. |
|
| 46. |
Whichseriesof linesobtainedin ultravioletregionin hydrogenspectra? |
| Answer» ANSWER :C | |
| 47. |
What are the main postulates of Valence Shell Electron Pair Repulsion (VSEPR) theory? |
|
Answer» Solution :(i) The shape of a molecule depends UPON the no. of ELECTRON pairs AROUND the centra ATOM. (ii) There is a repulsive force between the electron pairs, which tend to repel one another. (iii) The electron pairs in space tend to occupy such positions that they are at maximum distance, so that the repulsive force will be minimum. (iv) A multiple bond is treated as IFIT 1s a single bond and the remaining electron pairs which onstitute the bond may be regarded as single super pair. |
|
| 48. |
Which of the following factors is of no significance for roasting sulphide ores to oxides and not subjecting the sulphide ores to carbon reduction directly ? |
|
Answer» `CO_2` is more VOLATILE than `CS_2` |
|
| 49. |
When will the total heat absorbed by system during the process is completely used up in work? |
| Answer» SOLUTION :When `Deltau=0, q=-w`. | |



