Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
When dissolved in dilute sulphuric acid, 0.275 g of a metal evolved 119.7 mL of hydrogen at 20°C and 763 mm pressure. What is the equivalent mass of the metal ? |
|
Answer» <P> Solution :In the PRESENT case,`P_1 = 763 mm, V_(1) = 119.7 ML, T_2 = 20°C = 293` K, MASS of metal = 0.275 g If the volume of hydrogen at N.T.P is `V_2`, we have from gas equation `(P_(1)V_(1))/T_(1) = (P_(2)V_(2))/T_(2)` or `(763 xx 119.7)/293 = (760 xx V_(2))/273` `therefore V_(2)=(793 xx 119.7 xx 273)/(760 xx 293) = 111.97` mL Mass of 111.97 mL of `H_2` at S.T.P. `=2/(22400) xx 111.97 = 0.011` g Hence, Equivalent weight of the metal `=("Mass of the metal")/("Mass of "H_(2) "evolved at S.T.P") = 0.275/0.011 = 25` |
|
| 2. |
Write the structure of pyrophosphoric aicd. |
|
Answer» <P> Solution :`HO-underset(|)overset(||)P-O-underset(|)overset(||)P-OH` Pyrophosphoric `(H_(4)P_(2)O_(7))` ACID |
|
| 3. |
W/V percentage of 1 M H_2O_2solution is |
|
Answer» 3.03 |
|
| 5. |
The total number of reagents(s) which give white precipitate with solution of sodium oxalates. (MnO_(2)+H_(2)SO_(4)),CaCI_(2),AgNO_(3),BaCI_(2),KMnO_(4),(K_(2)Cr_(2)O_(7)+H_(2)SO_(4)) |
|
Answer» |
|
| 6. |
What do you mean by dihedral angle or torsional angel ? |
| Answer» SOLUTION :Magnitude of torsional strain depends upon the angle of rotation about C-C BOND. This angle is also CALLED DIHEDRAL angle or torsional angle. | |
| 7. |
Three reactions involving H_(2)PO_(4)^(-) are given below : (i) H_(3)PO_(4) + H_(2)O rarrH+_(3)O^(+) + H_(2)PO_(4)^(-) (ii) H_(2)PO_(4)^(-) + H_(2)O rarr HPO_(4)^(2-) + H_(3)O^(+) (iii) H_(2)PO_(4)^(-) + OH^( -) rarr H_(3)PO_(4) + O^(2-) In which of theabove does H_(2)PO_(4)^(-) act as an acid ? |
|
Answer» (iii)only (ii), `H_(2)PO_(4)^(-)` is donating a proton which is ACCEPTED by `H_(2)O`. Hence it is an acid. In (iii) , `H_(2)PO_(4)^(-)` is accepting a proton. Hence it is a base Thus, only in (ii), `H_(2)PO_(4)^(-)` acts an acid. |
|
| 8. |
The solution of salt of (HCl + NH_4OH) is ____ |
|
Answer» Acidic |
|
| 10. |
Why can dilute solutions of hydrogen peroxide not be concentrated by heating ? How can a concentrated solution of hydrogen peroxide be obtained ? |
|
Answer» Solution :`H_2O_2` cannot be CONCENTRATED by heating because it decomposes much below its boiling point to GIVE `H_2O` and `O_2`. `2H_2O_2 to 2H_2O+ O_2` Therefore, concentrated of `H_2O_2` is carried out in a number of STAGE (i) The `H_2O_2` formed (about 1%) is EXTRACTED with water and the aqucous solution is concentrated by distillation under REDUCED pressure. to give 30% `H_2O_2` solution. |
|
| 11. |
What are the products in the following sequence of reaction Poly (F) is: |
|
Answer» Nylon-6,6 |
|
| 12. |
What is electronic configuration ? |
| Answer» Solution :Distribution of ELECTRONS in an ATOMS is known as ELECTRONIC configuration. | |
| 13. |
The varous types of hydrides and examples of each type are given below : {:(,"Hydride type",,"Compound"),((A),"Electron deficient",(i),LiH),((B),"Saline",(ii),CH_(4)),((C ),"Electron-precise",(iii),NH_(3)),((D),"Interstitial",(iv),B_(2)H_(6)),((E ),"Electron rich",(v),CrH):} Choose the correct matching from the codes given below : |
|
Answer» (A) - (ii), (B) - (iv), (C )-(v), (D)-(iii), (E )-(i) Saline - LiH Electron precise `- CH_(4)` Interstitial - CRH Electron rich `- NH_(3)` |
|
| 14. |
The type of hybrid orbitals employed in the formation of SF_(6) molecules is |
|
Answer» `SP^(3)d` |
|
| 15. |
The solubility of H_2O_2 is 48 gm in 500 ml calculate volume strength and %w/v. |
|
Answer» 34.62,19.2 `=48/(34xx0.5)=2.82 M H_2O_2` %w/v = `(Mxx34)/10 = (2.82xx34)/10`=9.6 V=M x 11.2 = 2.82 x 11.2 = 31.58 |
|
| 16. |
Which of the following statement (s) is/are true about the following eliminations ? |
|
Answer» HOFFMANN PRODUCT is MAJOR product in I |
|
| 17. |
Which of the following properties of lithium cannot be attributed to its anomalous properties |
|
Answer» Lithium chloride crystallizes as hydrated lithium chloride . |
|
| 18. |
Which is the most reactive alkane towards bromination in presence of sunlight ? |
|
Answer» `CH_3CH_3` |
|
| 19. |
Which of the following molecules represents the order of hybridization sp^(2), sp^(2), sp, sp from left to right atoms ? |
|
Answer» `HC -= C-C-=CH` |
|
| 20. |
What iselectromagneticradiation ? |
|
Answer» SOLUTION :JamesMaxwellwas the FIRST to GIVE acomprehensiveexplanationabout theinteractionbetweenthe chargebodiesand thebehavior of ELECTRICAL andmagneticfields. Whenelectrically chargedparticlemovesunderaccelerationaltermatingelectricalandmagneticfieldareproduced andtransmitted . thesefieldare transmittedin the formsofwaves calledelectromagneticwavesofelectromagneticradiation. |
|
| 21. |
Which one of the following is an incorrect statement for physiosorption ? |
|
Answer» It is reversible process |
|
| 22. |
which salt can be produced by the reaction of carbon monoxide and caustic soda (NaOH)? |
|
Answer» `CH_(3)COOH` |
|
| 23. |
What are Cabanions ? |
|
Answer» Solution :Let us consider the heterolytic fission of the bond C-X present in a ORGANIC molecules . If the carbon atom FORMER takes away the bonding electron PAIR and ACQUIRES a negative charge . The resulting carbon anion is known as Carbanion . ` (##SUR_CHE_XI_V02_C12_E02_064_S01.png" width="80%"> |
|
| 24. |
Which one of the metal is used to convert para hydrogen into ortho hydrogen? |
|
Answer» COPPER |
|
| 25. |
Which of the following gases is not a greenhouse gas ? |
|
Answer» CO |
|
| 26. |
The redispersal of a freshly precipitated substance into a sol by the addition of an electrolyte in common is known as |
|
Answer» Aggregation |
|
| 27. |
Whichone (atom//ion )in the followingpairshas highestelectrongainenthalpy ? (i)O^(-)S(ii) O, S^(-)(iii) O^(-), S^(-)(iv)N^(-)P |
|
Answer» <P> (ii) Onsimilargroundsthe electron gainenthalpyof `S^(-)` is+ vewhilethat OFO is - ve (iii) dueto smallsizerepulsionsbetween `O^(-)` and theincomingelectron is muchmore thanin `S^(-)` .thereforeelectron gainenthalpyof `O^(-)` is more+ vethanthatof `S^(-)` (iv) DUE torepulsion between`N^(-)` and theincomingelectron gainenthalpyof `N^(-)` positivewhilethat P is -ve |
|
| 28. |
What is difference between barometer and nanometer ? |
|
Answer» SOLUTION :Barometer is USED for measurement of pressure of atmosphere and nanometer is used for measurement of pressure of gases. Example : Pressure of barometer is 740 mm of Hg and difference between two end of nanometer is 2.1 CM. Then calculate pressure of Gas ? Pressure of Gas = Pressure of barometer - Difference between two end `= 740-(2.1xx10) [because 2.1 cm = 2.1xx10 mm]` = 719 mm Hg. |
|
| 29. |
Which of the following is less than zero during adsorption? |
| Answer» Answer :D | |
| 30. |
Which of the following cations is not hydrolyzed in aqueous solution ? (i) Ba^(2+),(ii) Ca^(2+) (iii) Na^(+),(iv) K^(+) |
|
Answer» (i), (ii) |
|
| 31. |
What is [H^(+)] in mol/L of a solution that is 0.20 M in CH_(3)CO ON aand 0.10M in CH_(3)CO OH ? K_(a) for CH_(3)CO OH = 1.8 xx 10^(-5). |
|
Answer» `9.0xx10^(-6)` `pH = pK_(a) +log. (["SALT"])/(["ACID"])` `=-log.(1.8xx10^(-5))+log.(0.20)/(0.10)` or `-log(h^(+)]=log.(2)/(1.8xx10^(-5))` or `log.(1)/([H^(+)])=log.(2)/(1.8xx10^(-5))` or `[H^(+)]=(1.8xx10^(-5))/(2) = 0.9xx10^(-5)M` `=9xx10^(-6)M` |
|
| 32. |
Which set of quantum numberis notpossible |
|
Answer» |
|
| 33. |
Which is the correct Lewis structure of N_(2) ? |
|
Answer»
 In `N_(2)` TOTAL valence ELECTRON = 5 + 5 = 10 Only OPTION (A) is correct for no. of electrons. 
|
|
| 34. |
Which of the following is a stronger base? Give reason to justify your answer. {:(CH_(2)=CH-NJ_(2)""CH_(2)=N-CH_(3)),(""I""II):} |
|
Answer» |
|
| 35. |
The tendency of BF_(3),BCl_(3) and BBr_(3) to have as Lewisacid decreases in the sequence |
|
Answer» `BF_(3) lt BCl_(3) gt BBr_(3)` |
|
| 36. |
which of the following is not matched correctly with its use? |
|
Answer» PIEZOELECTRIC MATERIAL - Quartz |
|
| 37. |
What would be the pH of a solution obtained by mixing 10 g of acetic acid and 15 g of sodium acetate and making the volume equal to 1L. Dissociation constant of acetic acid at 25^(@)C is1.75xx10^(-5). |
|
Answer» Solution :`pH=pK_(a) + log. (["Salt"])/(["Acid"])=pK_(a) + log. ([CH_(3)CO ON a])/([CH_(3)CO OH])` `[CH_(3)CO OH]=(10)/(60) "mol L"^(-1)` (molar MASS of `CH_(3)CO OH = 60 g "mol"^(-1)`) `[CH_(3)CO Ona]=(15)/(82) "mol" L^(-1)` (molar mass of `CH_(3)CO Ona = 82 g "mol" ^(-1)`) `pK_(a)=-log K_(a)=-log(1.75xx10^(-5))=5-0.2430 = 4.757` `:. pH = 4.757 + log(15//82)/(10//60) = 4.757+ log. ((15)/(82)xx(60)/(10))` `=4.757 + log 1.098 = 4.757 + 0.0406 = 4.80` |
|
| 38. |
The reagent(s) used for softening the temporary hardnes of water is(are): |
|
Answer» `Ca_(3)(PO_(4))_(2)` |
|
| 39. |
Which of the following contain same number of carbon atoms as in 6 g of carbon-12? |
|
Answer» 7.5 G ethane = 6/12 = 0.5 moles = `0.5 xx 6.022 xx 10^(23)` carbon atoms. No. of moles in 8 g of methane = 8 /16 = 0.5 moles ` = 0.5 xx 6.022 xx 10^(23)` carbon atoms. No. of moles in 7.5 g of ethane = 7.5 / 16 - 0.25 moles = `2 xx 0.25 xx 6.022 xx 10^(23)` carbon atoms |
|
| 40. |
The volume of the nucleus of an atom when compared to the extra nuclear part is ______. |
|
Answer» heavier |
|
| 41. |
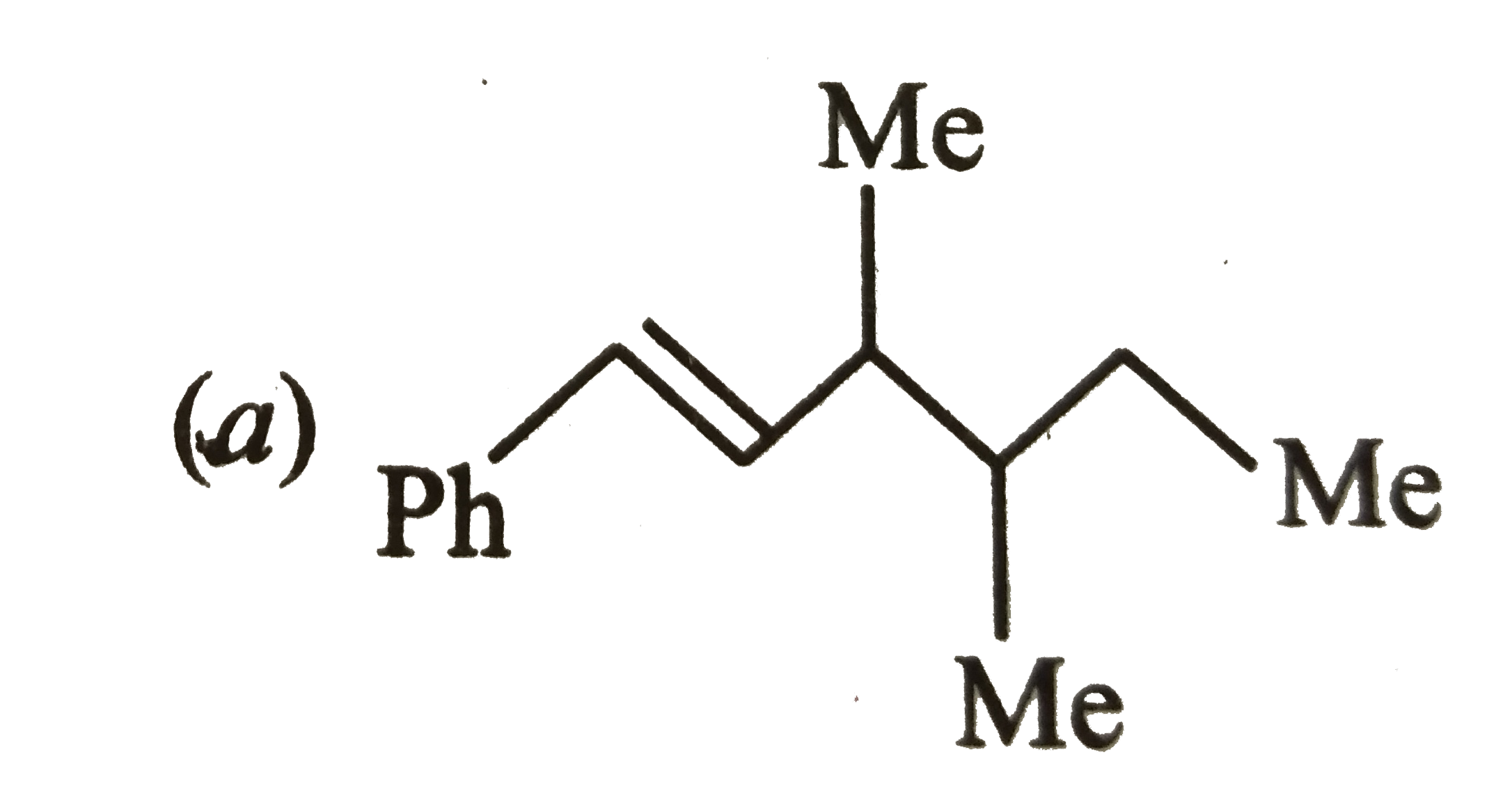
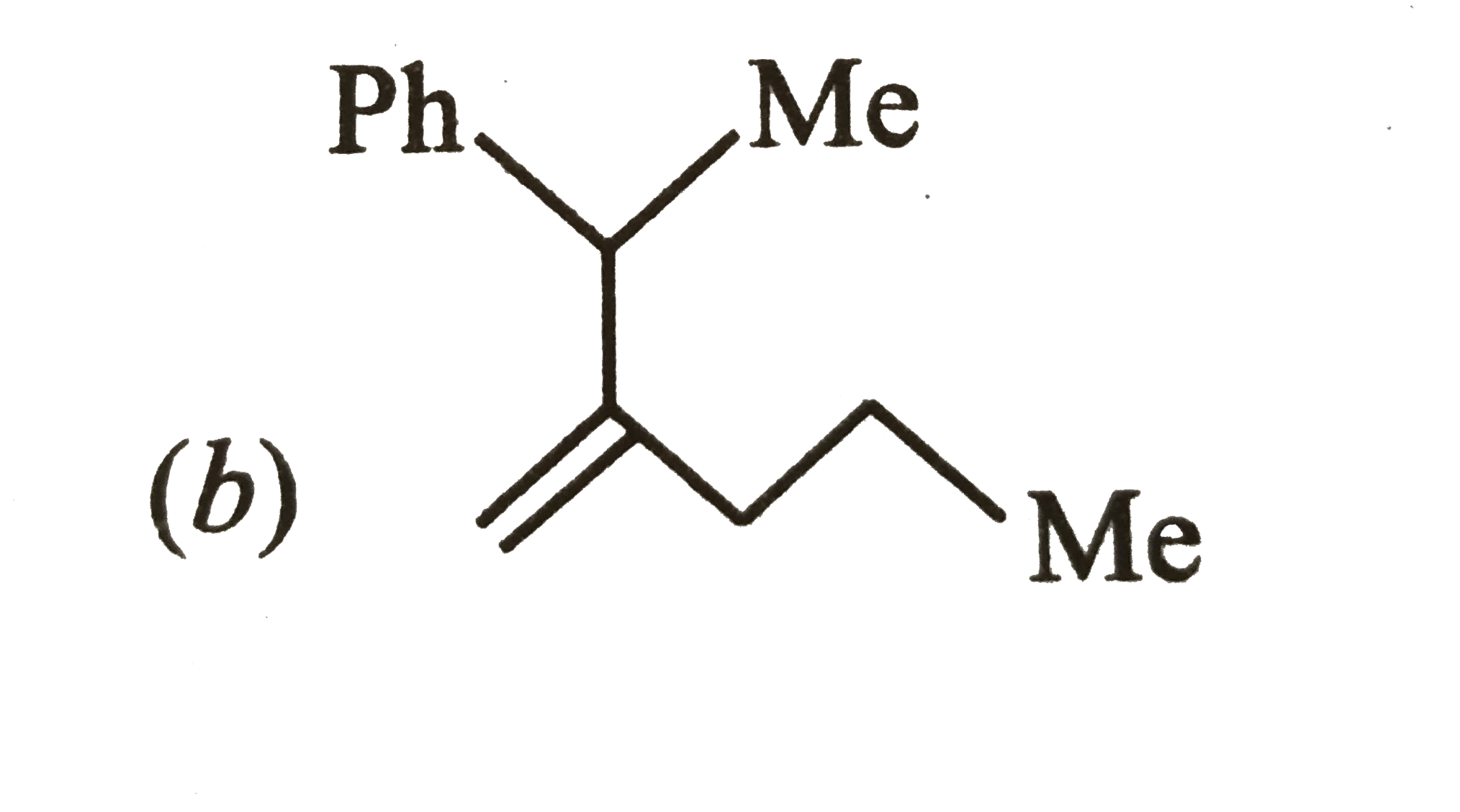
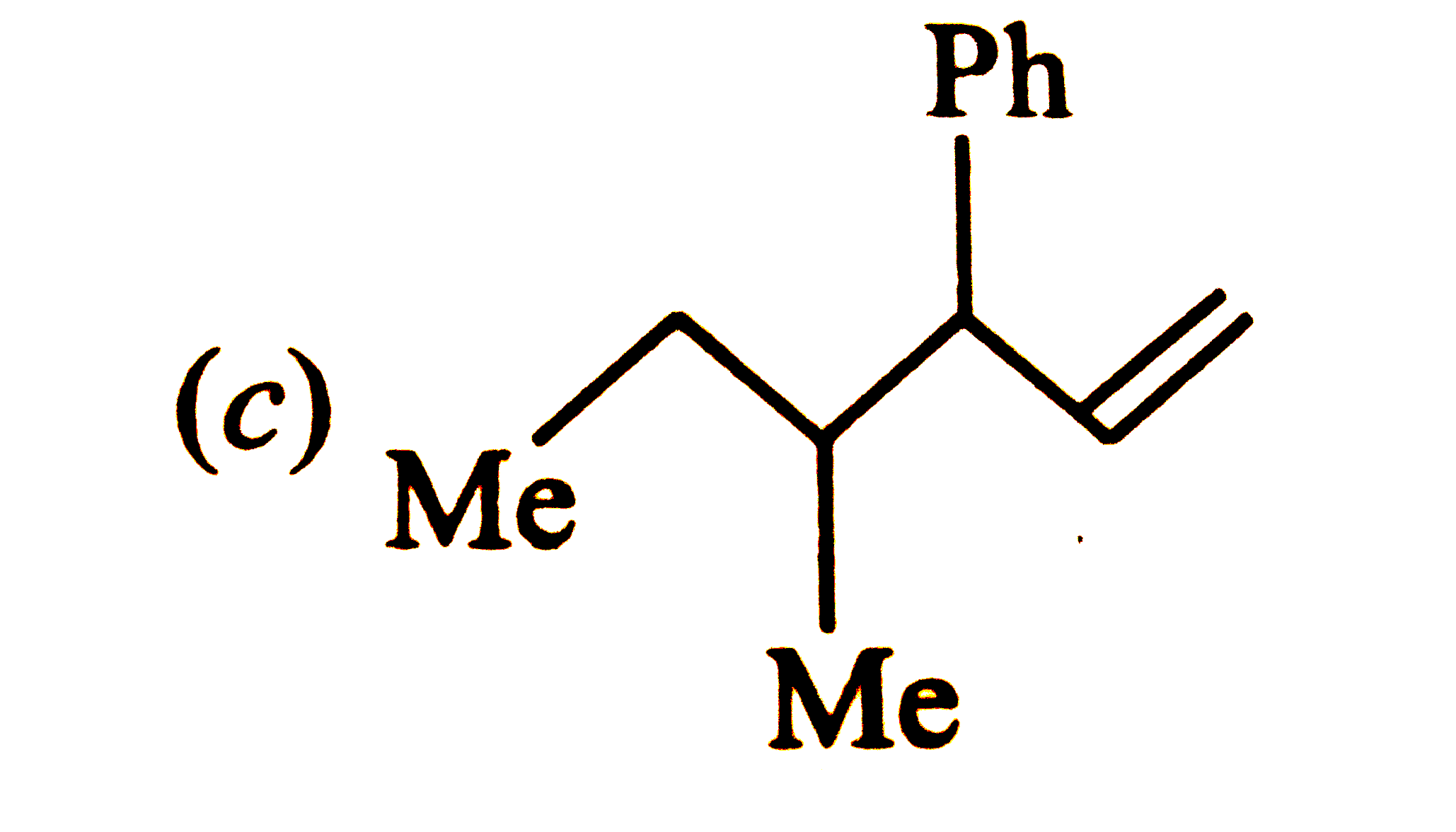
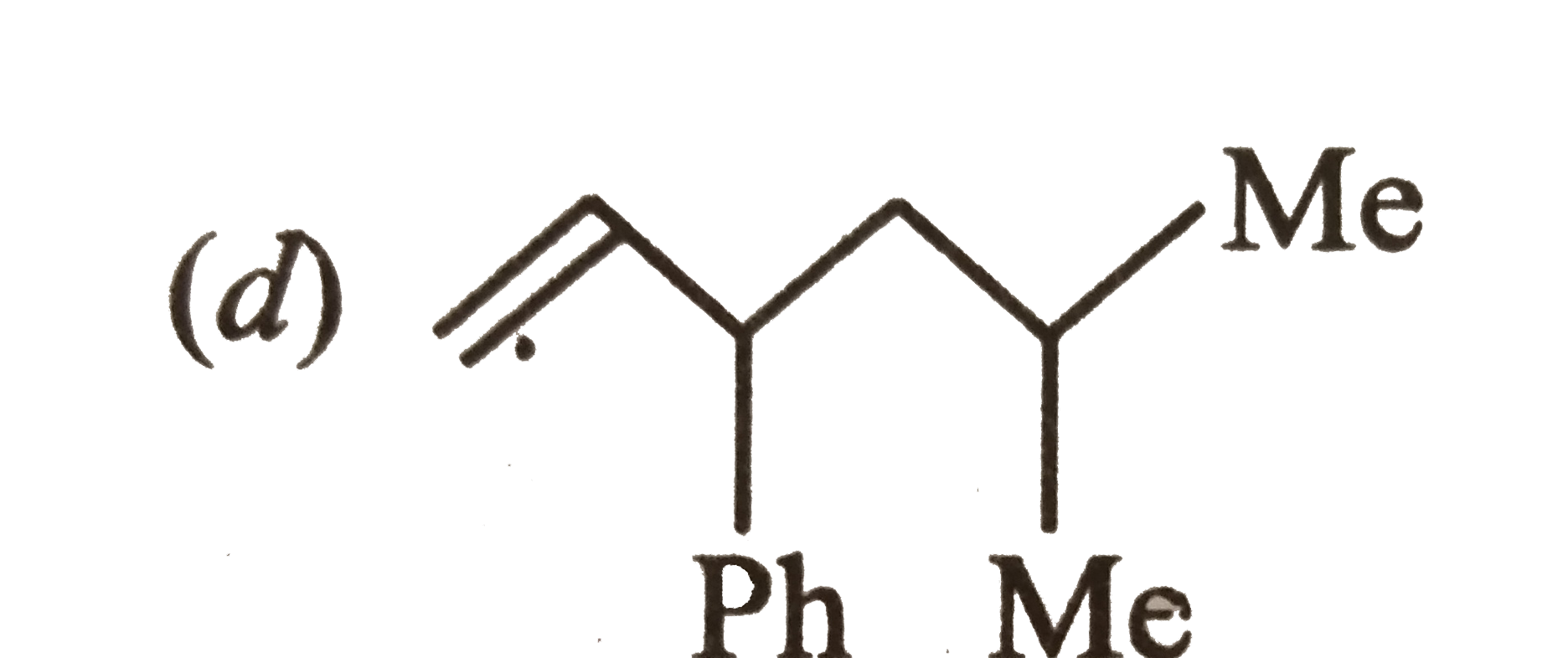
Which one of the following is s-butylphenylvinyl methane ? |
|
Answer»
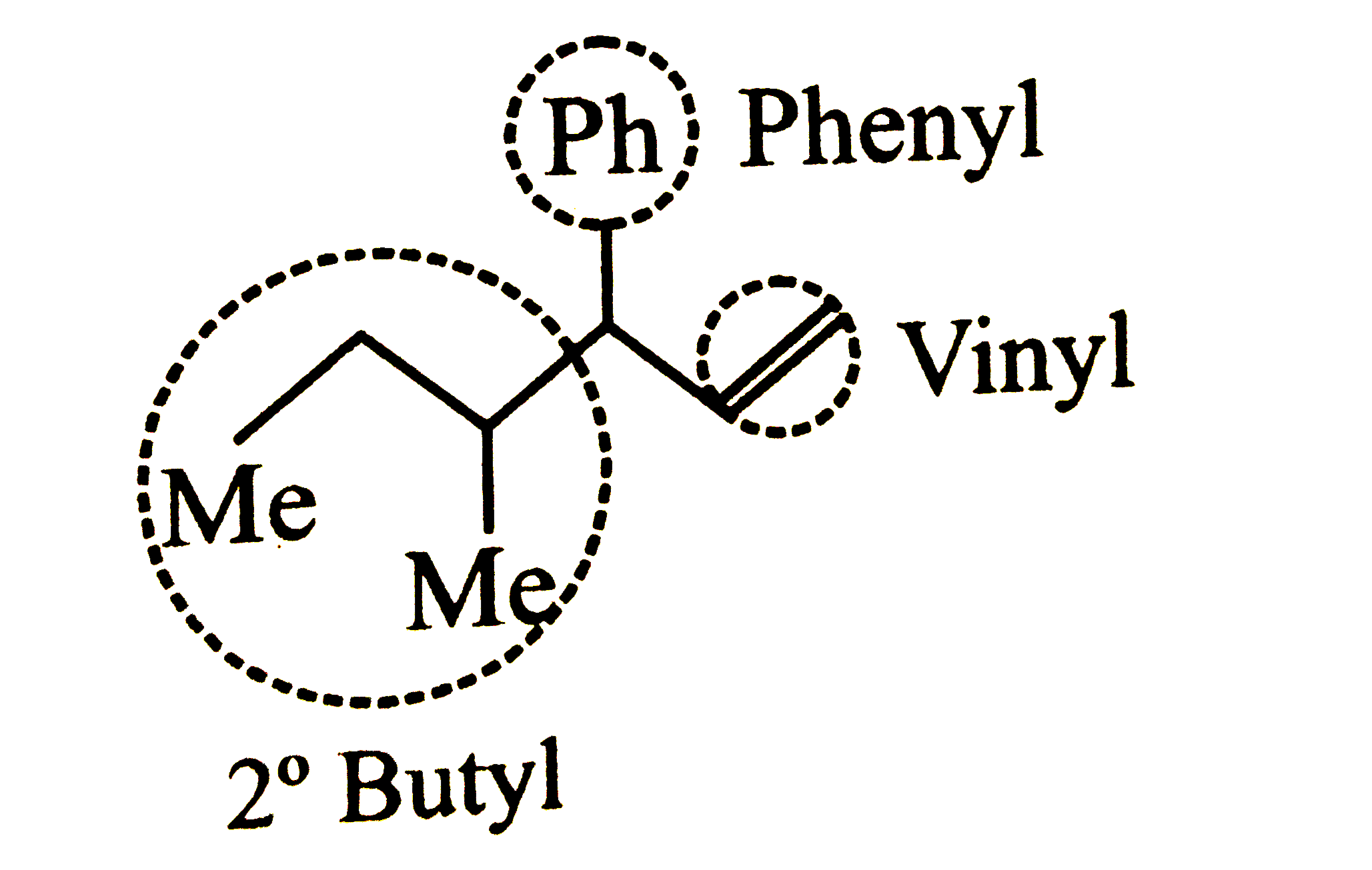
|
|
| 42. |
Which of the following gases is not a green house gas ? |
|
Answer» CO |
|
| 43. |
The splitting of spectral lines by a magnetic field is called the zeeman effect after the Dutch physicist Pieter Zeeman. The Zeeman effect is a vivid confirmation of of space quantization. Magnetic quantum number 'm' was introduced during the study of zeeman effect. 'm' can have the (2l+1) values (-l,0,+l). Magnetic quantum number represents the orientation of atomic orbitals in three-dimensional space. the normal zeeman effect consists of the splitting of a spectral line of frequency v_(0) into three components i.e., v_(1)=v_(0)-(e)/(4pim)B,v_(2)=v_(0),V_(3)=v_(0)+(e)/(4pim)B Here, B is magnetic field. Q. Which of the following statements is incorrect with reference to the Zeeman effect? |
|
Answer» In a MAGNETIC field, the energy of a paritcular ATOMIC state depends on the values of 'm' and 'n' |
|
| 44. |
The splitting of spectral lines by a magnetic field is called the zeeman effect after the Dutch physicist Pieter Zeeman. The Zeeman effect is a vivid confirmation of of space quantization. Magnetic quantum number 'm' was introduced during the study of zeeman effect. 'm' can have the (2l+1) values (-l,0,+l). Magnetic quantum number represents the orientation of atomic orbitals in three-dimensional space. the normal zeeman effect consists of the splitting of a spectral line of frequency v_(0) into three components i.e., v_(1)=v_(0)-(e)/(4pim)B,v_(2)=v_(0),V_(3)=v_(0)+(e)/(4pim)B Here, B is magnetic field. Q. A d-subshell in an atom in the presence and absence of magnetic field is: |
|
Answer» five-fold DEGENERATE, non-degenerate |
|
| 45. |
The splitting of spectral lines by a magnetic field is called the zeeman effect after the Dutch physicist Pieter Zeeman. The Zeeman effect is a vivid confirmation of of space quantization. Magnetic quantum number 'm' was introduced during the study of zeeman effect. 'm' can have the (2l+1) values (-l,0,+l). Magnetic quantum number represents the orientation of atomic orbitals in three-dimensional space. the normal zeeman effect consists of the splitting of a spectral line of frequency v_(0) into three components i.e., v_(1)=v_(0)-(e)/(4pim)B,v_(2)=v_(0),V_(3)=v_(0)+(e)/(4pim)B Here, B is magnetic field. Q. Which among the following is/are correct about the orientation of atomic orbitals in space? |
|
Answer» s-orbitals has single orientation |
|
| 46. |
The splitting of spectral lines by a magnetic field is called the zeeman effect after the Dutch physicist Pieter Zeeman. The Zeeman effect is a vivid confirmation of of space quantization. Magnetic quantum number 'm' was introduced during the study of zeeman effect. 'm' can have the (2l+1) values (-l,0,+l). Magnetic quantum number represents the orientation of atomic orbitals in three-dimensional space. the normal zeeman effect consists of the splitting of a spectral line of frequency v_(0) into three components i.e., v_(1)=v_(0)-(e)/(4pim)B,v_(2)=v_(0),V_(3)=v_(0)+(e)/(4pim)B Here, B is magnetic field. Q. In presence of magnetic field, d-suborbit is: |
|
Answer» five-fold degenerate |
|
| 47. |
The splitting of spectral lines by a magnetic field is called the zeeman effect after the Dutch physicist Pieter Zeeman. The Zeeman effect is a vivid confirmation of of space quantization. Magnetic quantum number 'm' was introduced during the study of zeeman effect. 'm' can have the (2l+1) values (-l,0,+l). Magnetic quantum number represents the orientation of atomic orbitals in three-dimensional space. the normal zeeman effect consists of the splitting of a spectral line of frequency v_(0) into three components i.e., v_(1)=v_(0)-(e)/(4pim)B,v_(2)=v_(0),V_(3)=v_(0)+(e)/(4pim)B Here, B is magnetic field. Q. Zeeman effect explains splitting of spectral lines is: |
| Answer» Answer :A | |
| 48. |
Which of the following property increases as we go down the group in the periodic property? |
|
Answer» IONIZATION energy |
|
| 49. |
What is the colour of the flame obtained on burning the vapours of ethyl borate? |
| Answer» SOLUTION :GREEN EDGED FLAME | |
| 50. |
Which one is pesticides? |
|
Answer» INSECTICIDES |
|