Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
What is molal depression constant? Does it depend on nature of the solute ? |
|
Answer» Solution :`K _(f) =` molar FREEZING point DEPRESSION constant or cryoscopic constnat. `Delta T _(f) = K_(f).m,` where `Delta T_(f)=` depression in freezing point, m = molality of the solution `K_(f)=` cryoscopic constant If `m =1, Delta T_(f) = K _(f)` i.e., cryoscopic constant is equal to the depression in freezing point for 1 molal solution cryoscopic constant depends on the molar concentration of the solute PARTICLES. `K_(f)` is directly proportional to the molar concentration of the solute particles. `Delta T_(f) = K _(f) xx (W_(B) xx 1000)/( M _(B) xx W_(A))` `W_(B)=` mass of the solute, `W_(A)=` mass of solvent, `M _(B)=` molecular mass of the solute. |
|
| 2. |
Which one among the following does not have the hydrogen bond? |
| Answer» Answer :A | |
| 3. |
Thermally most stable oxide is |
|
Answer» `Na_(2)O` |
|
| 4. |
Which of the following is // are correct match (i) Photochemical smog: SO_(2) (ii)Nitrate in drinking water : Blue baby syndrome (iii) Phosphate fertilisers in water : BOD level of water increases. |
|
Answer» only (i) |
|
| 5. |
The sulphur content of cystine is 26.7%. Given that cystine contains two sulphur atoms, what is the molecular weight of cystine? |
|
Answer» `= (100 xx 64)/(26.7) = 239.7 g mol^(-1)` |
|
| 6. |
What is Global warming? |
| Answer» SOLUTION :The HEATING up of the CARTH through the greenhouse effect is called Global WARMING. | |
| 7. |
The solution having minimum boiling points is |
|
Answer» ` 0.1 M C_6 H_12 O_6` |
|
| 8. |
When 10 g of anhydrous CaCl_(3) is dissolved in 200 g of water, the temperature of the solution rises by 7.7 ^(@) C. Calculate the heat of hydration of CaCl_(2) to CaCl_(2). 6 H_(2)O. Given that the heat of dissolution of CaCl_(2). 6 H_(2)O is 19.08 kJ mol^(-1) . Assume specific heat of the solution to be same as that of water, i.e., 4.184 J g^(-1) K^(-1) |
|
Answer» Solution :Step I. Calculationof heat of dissolution of anhydrous `CaCl_(2)` Molar mass of `CaCl_(2) = 40 + 2 xx 35.5 = 111g mol^(-1)` Heat EVOLVED in dissolved of 10g`CaCl_(2)` in 200 G WATER `=m xx c xx Detla t` `= 200 xx 4.184 xx 7.7J` `:. `Heat of dissolutionper mole `=( 200 xx 4.184 xx 7.7) /( 10 ) xx 111 J = 71.52kJ` i.e,`Delta_("disso.")H ( CaCl_(2)) = - 71.52kJ mol^(-1)` Step II. Calculation of heat of hydration . Aim `: CaCl_(2)(s) + 6H_(2)O(l)rarr CaCl_(2).6 H_(2)O(s)` We have `:` `Delta_("disso.") H(CaCl_(2))= 71.52kJ mol^(-1) ` ( Calculated above) `Delta_("disso.") H ( CaCl_(2). 6 H_(2)O) = 19.08 kJ mol^(-1) ` ( Given ) i.e.,`(i)CaCl_(2) (s) + aq rarr CaCl_(2)(aq), DeltaH_(1) = - 71.52kJ mol^(-1)` (ii) `CaCl_(2). 6 H_(2)O(s) + aq rarr CaCl_(2)(aq), DeltaH_(2) = 19.08kJ mol^(-1)` Reaction (i) can be written in two steps as `:` `CaCl_(2)(s) + 6 H_(2)O(l) rarrCaCl_(2). 6 H_(2)O(s) , Delta H - Delta H _(3)` `CaCl_(2). 6 H_(2) O (s) + aq rarr CaCl_(2)(aq), DeltaH = Delta H _(4)` `:. Delta H _(3) + Delta H _(4)DeltaH _( 1) = - 71.52 kJ mol^(-1)` But `DeltaH_(4) = DetlaH _(2) = 19.08 kJ mol^(-1)` `:. DeltaH _(3) + 19.08 -71.52kJ mol^(-1)` or `Delta H _(3) = - 90.6 kJ mol^(-1)` |
|
| 9. |
Which of the following is/are incorrect about keto-enol tautomerism? |
|
Answer» Acid catalysed enolisation INVOLVE the INTERMEDIATE carbanion 
|
|
| 10. |
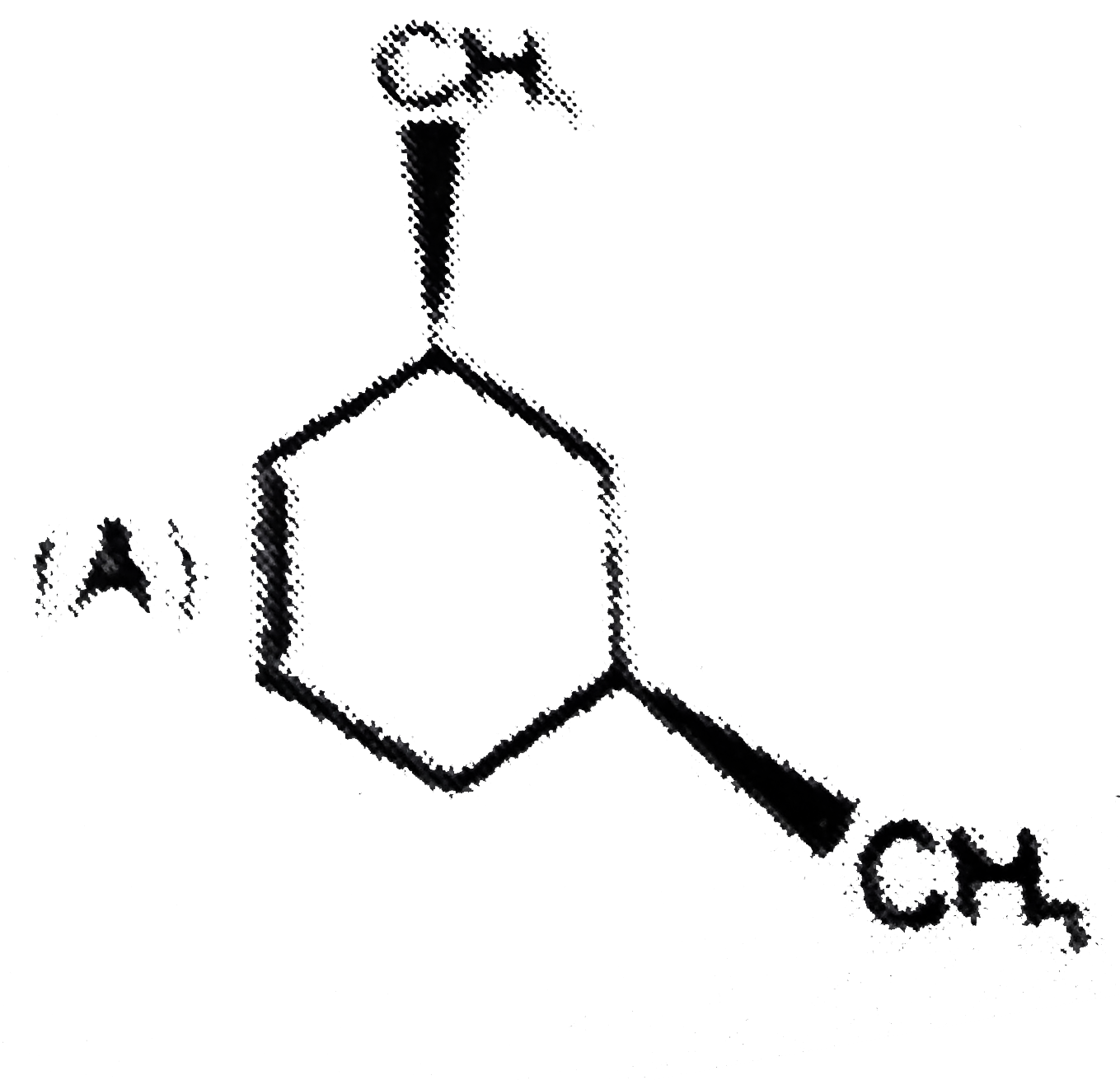
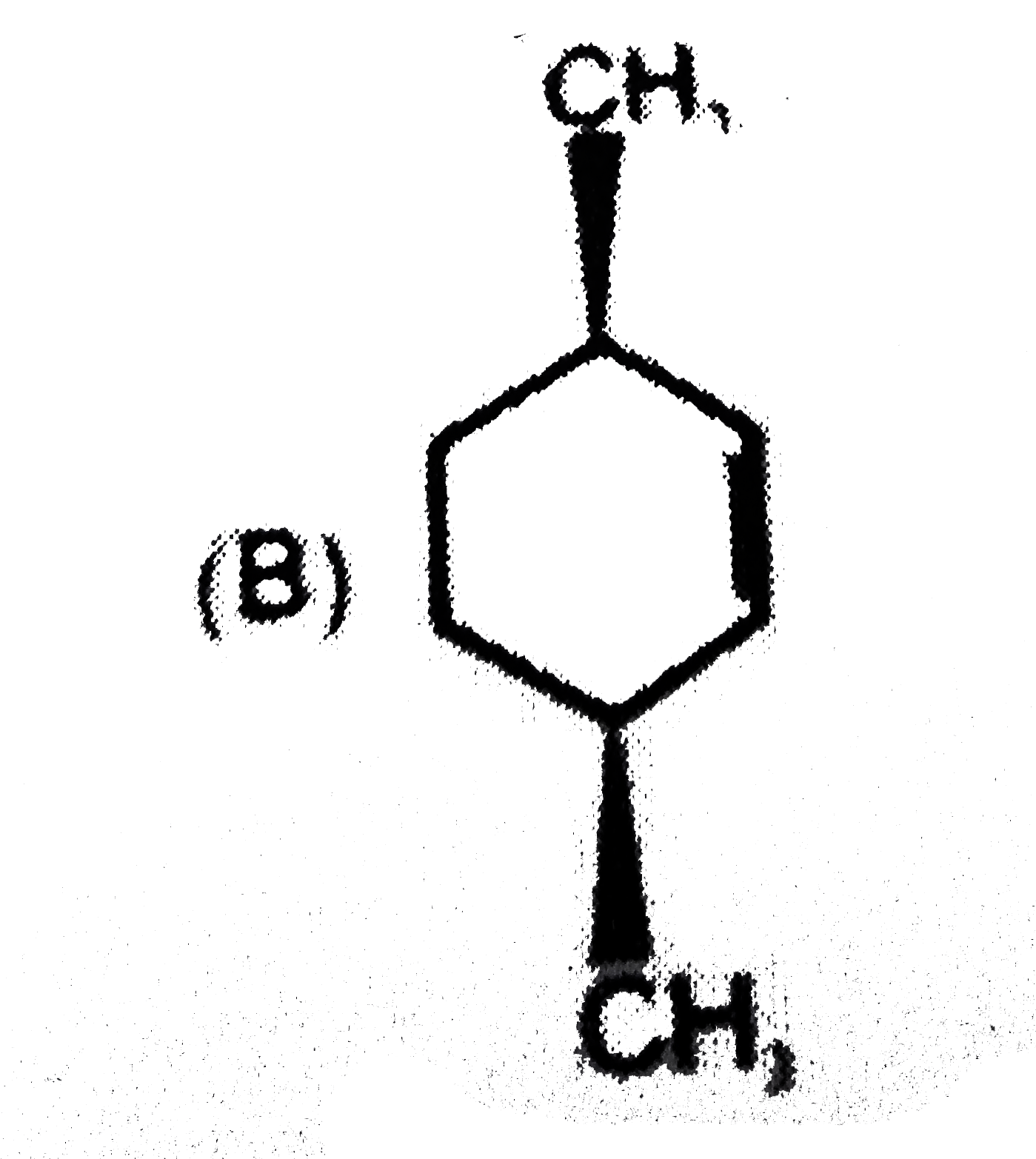
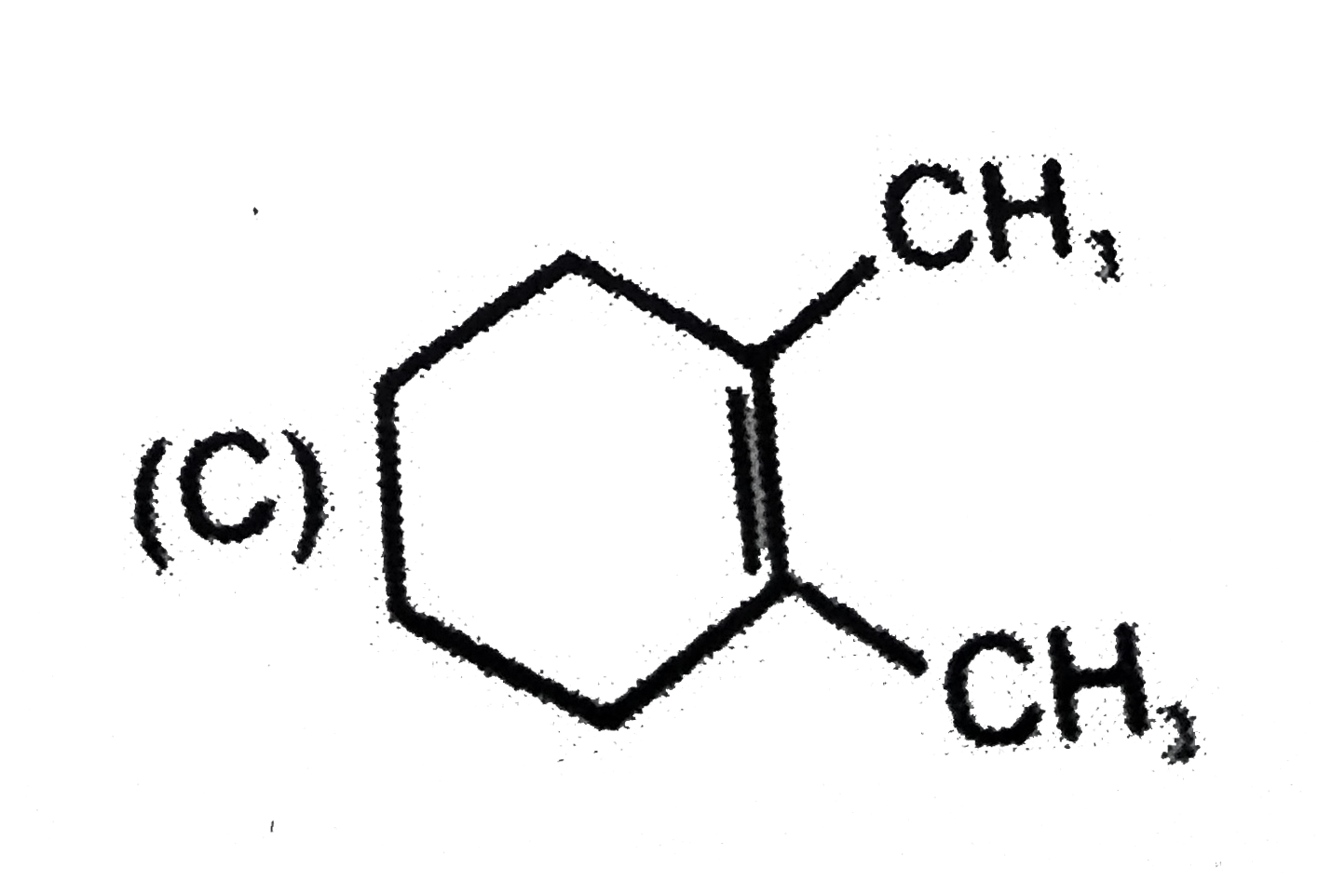
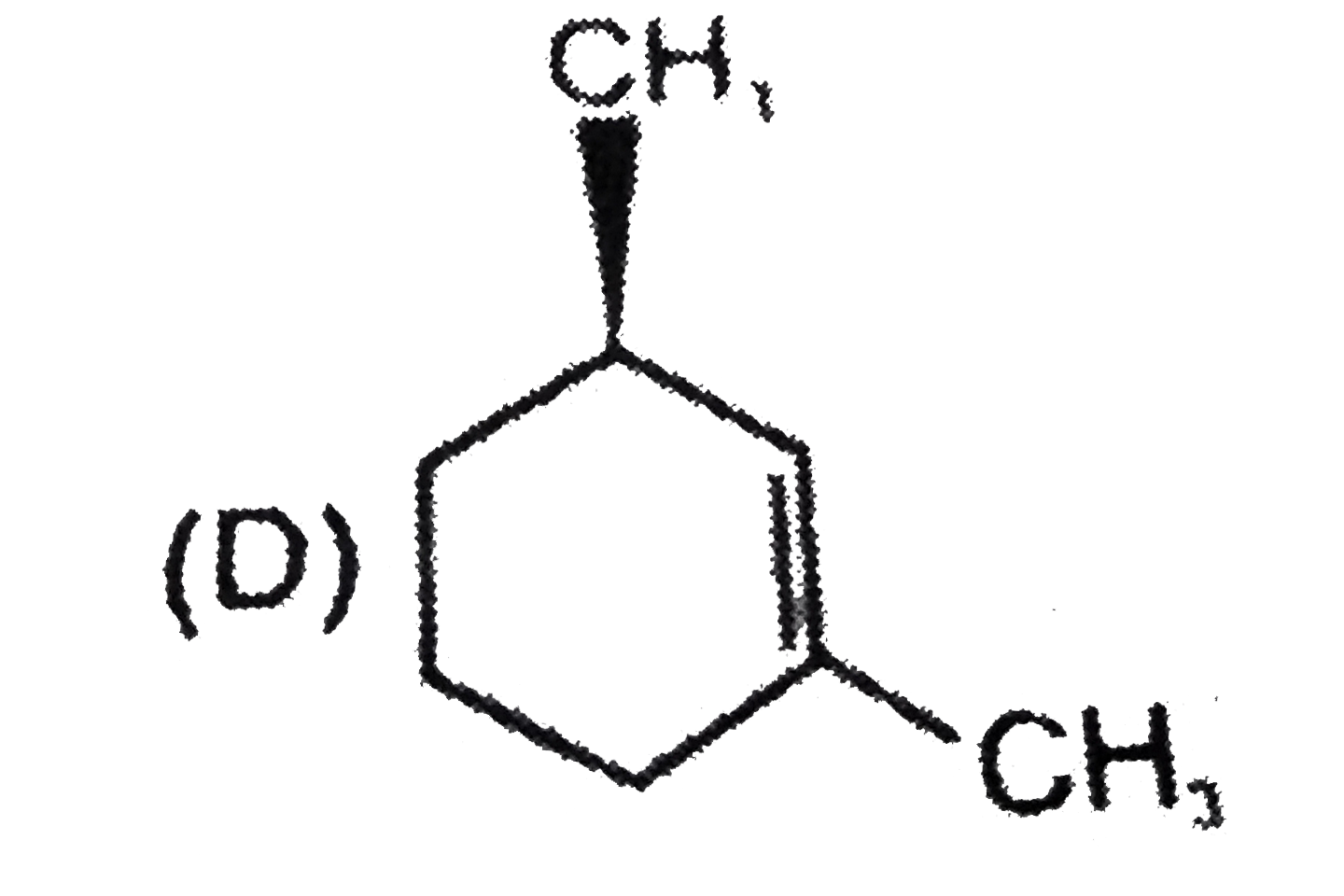
x grams of calcium carbonate was completely burnt in air. The weight of the solid residue formed is 28 g. What is the value of x (in grams)? |
|
Answer» 44 |
|
| 11. |
trans -Cyclohexane-1, 2-diol can be prepared by the reaction of cyclohexene with |
|
Answer» `KMnO_(4)` 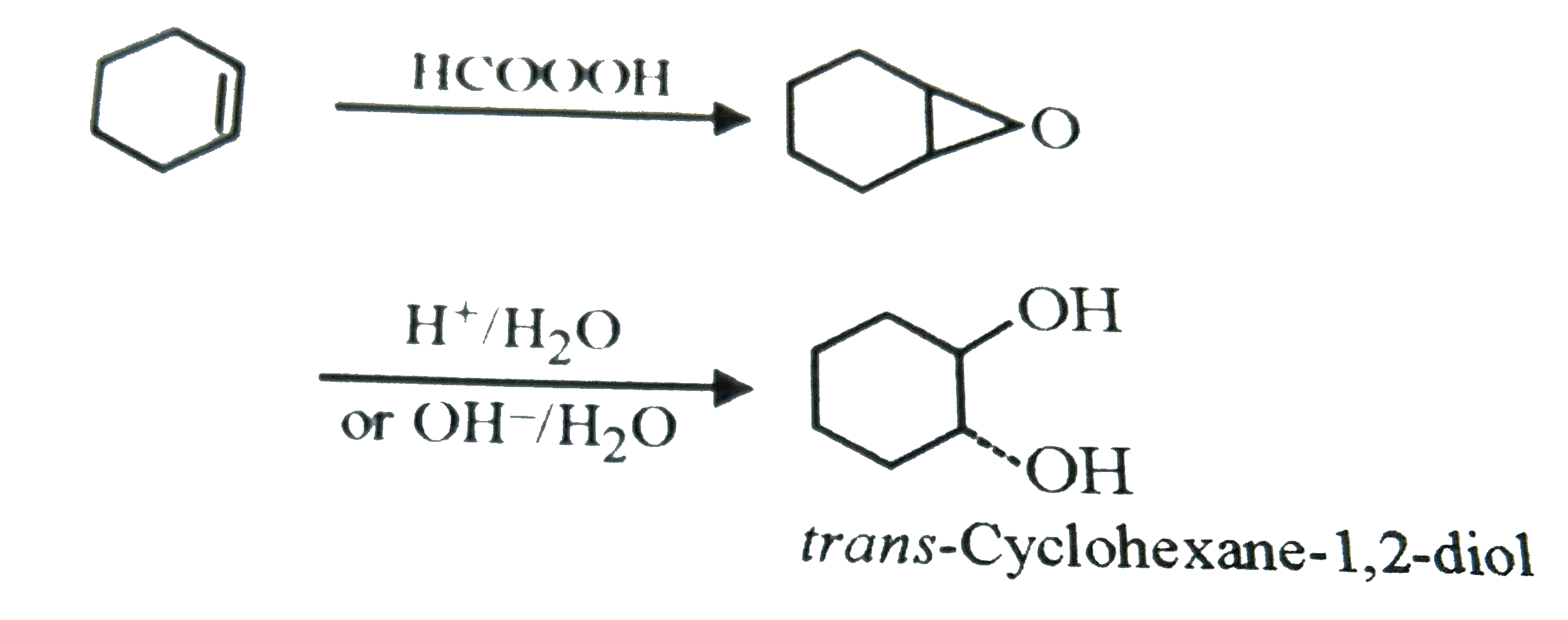
|
|
| 12. |
When CO_(2) is pass through the solution of Ca(OH)_(2), it produce……….. |
|
Answer» `CaCO_(3)` |
|
| 13. |
which of the following salt is present in teth? |
|
Answer» MILK of magnesia |
|
| 14. |
Two liquids A and B have vapour pressures 400 mm Hg and 450 mm Hg respectively at a given temperature. Which liquid has higher boiling point? |
| Answer» SOLUTION :LIQUID A | |
| 15. |
Which of the following suppresses the decomposition of H_(2)O_(2) |
|
Answer» `MnO_(2)` |
|
| 16. |
Which of the two is paramagnetic : V (IV) or V(V) and why ? |
| Answer» Solution :Electronic configuration of V(Z = 23) is `[Ar]^(18) 3d^(3) 4s^(2) " " :. V(IV) " is " [Ar]^(18) 3d^(1) and V(V) " is " [Ar]^(18) 3d^(0)` As V (IV) has one unpaired electron, THEREFORE, it is paramagnetic. | |
| 17. |
Which of the following statement(s) are correct? 1. Electronic configuration of Cr is [Ar]3d^(5)4s^(1) (At. No of Cr=24) 2. The magnetic quantum number may have negative value. 3. In silver atom, 23 electron have a spin of one type and 24 of the opposite type (At. No. of Ag=47) 4. The oxidation state of nitrogen in HN_(3) is -3. |
|
Answer» 1,2,3 |
|
| 18. |
What is the conc. of H_(2)O_(2) obtained by auto oxidation process? |
|
Answer» `50%` |
|
| 19. |
Which elements were discovered by scientist Ramsen and Glenn T Seaborg? |
|
Answer» Solution :Ramsen : He GIVE same HINT to DISCOVER elements Kr and Xe SUCCESSFULLY. Glenn T. SEABORG : He discovered element mendelevium having atomic number 101. |
|
| 20. |
Water has a higher boiling point than the corresponding hydrides H_(2)S,H_(2)Se and H_(2) Te. This is because water has |
|
Answer» IONIC BONDS |
|
| 22. |
Which of the following give only meso compound on catalytic reduction ? |
|
Answer»
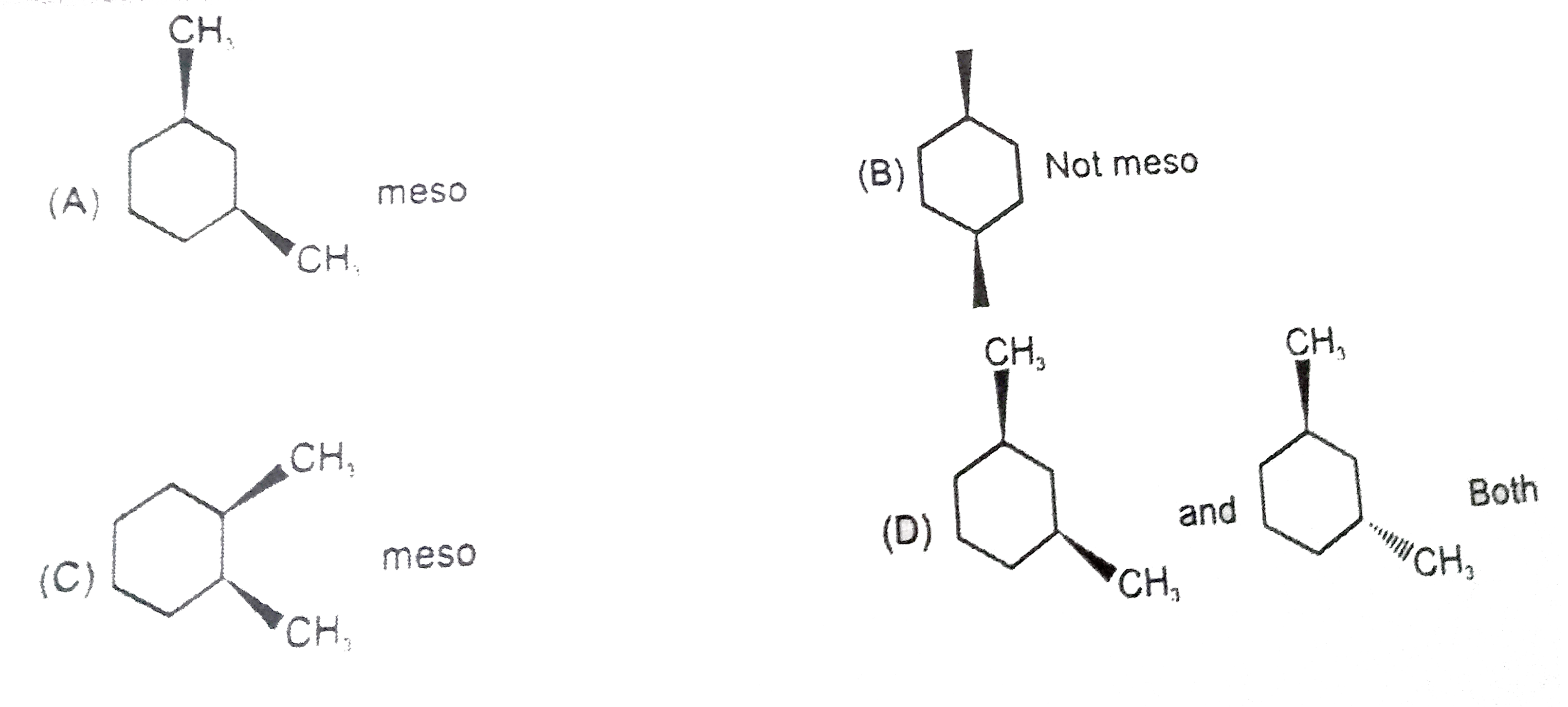
|
|
| 23. |
What is bon d energy? Give example. |
|
Answer» Solution :It is defined as..the amount of energy REQUIRED to break one mole of bonds of a particular type in the gasecous state. ..It is also CALLED bond dissociation enthalpy. Example : C-C bond enthatpy is `348kJmol^(-1)` |
|
| 24. |
Transition element exhibit variable valency on account of …… d- orbitals . |
|
Answer» NORMAL ELEMENTS |
|
| 25. |
Which is not correct statement |
|
Answer» s-block ELEMENTS, except Be and Mg, FORM ionic hydride |
|
| 26. |
Which of the following will notshow geometrical isomerism ? |
|
Answer»
|
|
| 27. |
The volume of gas is reduced to half from its original volume. The specific heat will be |
|
Answer» reduce to HALF Specific heat is an intensive property which is DEPENDING only on the nature of the gas. |
|
| 28. |
The spin quantum number has a value of |
|
Answer» `1//2` |
|
| 29. |
Write the relationship between dipole movement and charge. |
| Answer» SOLUTION :m=exd (where m= DIPOLE MOMENT, c=charge d=distance) | |
| 30. |
Write reactions to justify amphoteric nature of aluminium. |
|
Answer» SOLUTION :A substance is called amphoteric if it DISPLAYS CHARACTERISTICS of both acids and BASES. Aluminium dissolves in both acids and bases, showing amphoteric behaviour. `2Al+6HCl to 2Al^(+3) + 6Cl^(-) + 3H_2` `2Al + 2NAOH + 6H_2O to 2Na^(+) [Al(OH)_4]^(-) + 3H_2` |
|
| 32. |
Which combination of the compounds and their geometry are correct ? |
|
Answer» `HgCI_(2)` - linear |
|
| 33. |
Which of the following is a semi conductor ? |
|
Answer» C |
|
| 34. |
Which of the following occurs reaction intermediate? |
|
Answer» Free radicals |
|
| 35. |
What are the shapes of ethyne and methane? |
|
Answer» SQUARE PLANAR and linear |
|
| 37. |
The total number of protons in 10 gCaCO_(3) are...... |
|
Answer» `3.011xx10^(23)` |
|
| 38. |
Using Dalton's law how will you determine the pressure of a dry gas. |
|
Answer» Solution :The pressure of dry VAPOR can be calculated unsing DALTON's law `P_("dry GAS collected")=P_("total")-P_("water vapour")` `P_("water vapour")` is generally referred as aquesous tension and its values are available for air atvairous temperatures. Letus understand Dalton's law by SOLVING this Ne, `0.74` mole of AR and `2.5` mole of Xe. Calculate the partial pressure of gases, if the total pressure is 2 atm. at a fixed temperature. |
|
| 40. |
Which of the following statement(s) is/are correct about benzene? |
|
Answer» It BURNS with SOOTY flame |
|
| 41. |
Which of the following element(s) belongs to halogen group ? |
|
Answer» `1s^2 , 2s^2 , 2p^6 , 3s^23p^5` |
|
| 42. |
What is the relation between the value of K_c for the forward reaction and value of K'_c for the reverse reaction in the reaction H_(2(g)) + I_(2(g)) hArr 2HI_((g))at equilibrium ? |
|
Answer» `K'_c=2K_c` |
|
| 43. |
Which of the following is a Deuterolysis reaction |
|
Answer» `SO_(3)+D_(2)OtoD_(2)SO_(4)` |
|
| 44. |
Which of the following is used to remove lignin from woodpulp? |
|
Answer» Slakedlime |
|
| 45. |
Which of the following show optical isomerism and the isomers can be separated into dextro and laevo forms? |
|
Answer»
|
|
| 46. |
Write the sulphonation reactions of benzene. |
Answer» Solution :Various products OBTAINED by sulphonation of benzene which depends on CONDITION.  At LOWER TEMPERATURE benzene sulphonic acid and at high temperature BDS (Benzene Disulphonic Acid) is obtained. |
|
| 47. |
When excess carbon dioxide gas is passed through an aqueous solution of potassium super oxide a product (X) is formed . The correct statement regarding X is |
|
Answer» The ANION present in crystalline (X) exist as a dimer due to hydrogen bonds |
|
| 48. |
Boiling point of water at 750 mm Hg is 99.63^(@)C. How much sucrose is to be added to 500 gor water such that it boils at 100^(@) C ? |
|
Answer» Solution :Elevation in boiling point required,` Delta T _(b) = 100- 99.63^(@)=0.37^(@)` Mass of solvent (water)` W_(1)=500g` Mass of SOULTE, `C_(12) H _(22) O _(11) =342 g mol^(-1)` Molar mass of solvent `M _(1) = 18 g mol^(-1)` APPLYING the formula, `M _(2) = (1000 K _(b) W_(2))/(W_(1) XX Delta T_(b))` `W _(2) = (M_(2) xx Delta T_(b) xx W_(1))/(1000 xx K _(1))` `= ( 342 mol ^(-1) xx 0.37k xx 500 g )/( 1000 xx 0.52 kg mol ^(-1))= 121. 67g` |
|
| 49. |
What is a vapour pressure of liquid ? What is relative lowering of vapour pressure? |
|
Answer» Solution :The pressure of the vapour in equilibriumwith its liquid is called vapour pressure of the liquid at the given TEMPERATURE Relative LOWERING in vapour pressure : On addition of a non volatile solute it is observed that the vapour pressure of the solution is LESSER than the solvant ACCORDING to Raolut's LAW relative lowering of vapour pressure is equal to the mole fraction of the solute. |
|