Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which of the following statements is correct for absolute entropy of a substance ? |
|
Answer» It is shown as `S^(@)`. |
|
| 2. |
Which of the following ions, has polarizing power close to that of Mg^(2+) |
|
Answer» `RB^(+)` |
|
| 3. |
Three closed vessels A,B and C are at the same temperature T and contains gases which obey the Mawellian distribution of velocities. Vessel A containe only O_(2). B only N_(2) and C a mixture of equal quantities of O_(2) and N_(2). If the average speed of the O_(2) molecules in vessel A is V_(1), that of the N_(2) molecule in vessel C is: |
|
Answer» `((V_(1)+V_(2)))/(2)` |
|
| 4. |
The values of K_(sp) of two sparingly soluble salts, Ni(OH)_(2) and AgCN are 2.0xx10^(-15) and 6.0xx10^(-17), respectively. Which salt is more soluble ? Explain. |
|
Answer» `K_(SP) = s (2s)^(2) = 4 s^(3 ) = 2.0 xx 10^(-15) `. This gives `s=5.8xx10^(-5)` mol `L^(-1)` `{:(AgCN,hArr,"Ag"^(+),+,CN^(-),,,),(s,,s,,s,,,):}` or `s=sqrt(K_(sp))=sqrt(6.0xx10^(-17))=7.8xx10^(-9) ` mol `L^(-1)` |
|
| 5. |
What volume of water should be added to 300 mL of 0.5 M NaOH solution so as to get a solution of 0.2 M ? |
|
Answer» `overset(("Dilute"))(M_(1)V_(1))-= overset(("Conc."))(M_(2)V_(2))` `0.2xxV_(1)=0.5xx300orV_(1)=(0.5xx300)/(0.2)=750` ML `:.` Volume of WATER to be ADDED `= 750-300 = 450 mL`. |
|
| 6. |
Which one of the following molecule (s) is (are) expected to exhibit diamagnetic behaviour ? |
|
Answer» `S_(2)` i.e., there is no UNPAIRED electron PRESENT . HENCE both of them are diamagnetc. |
|
| 7. |
The volume occupied by any ges at S.T.P. is ___. |
|
Answer» 22.4 LITRES |
|
| 8. |
Total number of species present in 1 mole of potash alum in terms of avagadro number, 'N' are |
|
Answer» 3N |
|
| 9. |
Wavelengths of different rad iatio n s are given below : lambda(A) - 300 nm lambda(B) = 300 mum lambda(C) = 3 nm lambda(D) = 30A Arrange the seradiations in th eincreasing order of their energies. |
|
Answer» SOLUTION :`LAMBDA(A) = 300 nm = 300 xx 10^(-9)` m `lambda(B) = 300 mu m = 300 xx 10^(-6)m` `lambda (C) = 3nm = 3 xx 10^(-9) m` `lambda(D) = 30A = 30 xx I10^(-10) m = 3 xx 10^(-9) m` he 1 Energy, `E = (hc)/(lambda)`So, `E prop (1)/(lambda)` -INCREASING order of energy is `B lt A lt C = D` |
|
| 10. |
The supply of oxygen to the tissues by blood (haemoglobin) can be examined by |
|
Answer» BOYLE's law |
|
| 11. |
The stability of lyophillic colloid is due to which of the following? |
|
Answer» CHARGE on their particles |
|
| 12. |
The RMS velocity of hydrogen is sqrt7 times the RMS velocity of nitrogen. If T is the temperature of the gas |
|
Answer» `T (H_2) = T (N_2)` `:. "" U_(H_2)/U_(N_2)=sqrt((T_(H_2)xx28)/(T_(N_2)xx2))=sqrt7` (GIVEN) `(T_(H_2)xx14)/(T_(N_2))=7` or `T_(H_2)=1/2 T_(N_2), i.e., T_(H_2)ltT_(N_2)` |
|
| 13. |
What type of salts are Na_(2)HPO_(3) and NaHS ? |
Answer» Solution : `Na_(2)HPO_(3)` is obtained by REACTION between NaOH and `H_(3)PO_(3)` (a dibasic acid), (i.e., Both displaceable hydrogens are replaced by NA. No acidic hydrogenis left. Hence, `Na_(2)HPO_(3)` is a normal salt. NaHS is obtained byreplacement of ONE acidic hydrogen of `H_(2)S` by Na (on reaction with NaOH). Hence, NaHS is an acidic salt. |
|
| 14. |
Which catalyst is used in Friedel-craft alkylation reaction ? And what is its function ? |
Answer» Solution :The anhydrous `AlCl_(3)` is USED as catalyst in Friedel-craft reaction. This catalyst is Lewis acids and hence. `:CL^(-)` is accepted by `CH_(3)-Cl` and form`AlCl_(4)^(-)` COMPLEX and form methyl of ethyl carbocations.  As MENTIONED aboe, with anhydrous `AlCl_(3)` the formation of alkyl carbocations `R^(+).(overset(+)(C)H_(3), CH_(3)overset(+)(C)H_(2)....)` etc., strong electrophilic reactants are start forming. |
|
| 16. |
Which of the following element has the greatest tendency to lose electrons ? |
|
Answer» F |
|
| 17. |
Which of the following reagent is used to distinguish gem-dihalides and vicinal dihalides? |
|
Answer» ALCOHOLIC KOH |
|
| 18. |
What happens when silver propionate reacts with Br_(2) in C Cl_(4) ? |
|
Answer» Solution :Silver salt of fatty ACIDS `(CH_(3) CH_(2) CO O AG)` , e.g., silver propionate TREATED with `Br_(2) C Cl_4` givesbromoalkane . This reaction is called Hunsdicker reaction . `CH_(3) - CH_(2) COO Ag + Br_(2) overset(C Cl_(4)) underset("REFLUX") (to) CH_(3) - CH_(2) Br + CO_(2) uarr + Ag Br` |
|
| 19. |
What is responsible for soil pollution ? |
| Answer» Solution :Insecticides, pesticides and HERBICIDES which is USE to prevent the CORPS are responseble for the soil POLLUTION. | |
| 20. |
Write the structural formula for the following compounds. (i) cyclohexa-1, 3-diene (ii) methyl cyclopentane |
Answer» SOLUTION :(i) 
|
|
| 21. |
What is the most important condition for a process to be reversible in thermodynamics ? |
| Answer» SOLUTION :The process should be carried out infinitesimally slowly, or the DRIVING FORCE should be infinitensimally greater than the opposing force. | |
| 22. |
What is entropy ? |
| Answer» SOLUTION :ENTROPY is the MEASUREMENT of RANDOMNESS of the SUBSTANCE. | |
| 23. |
Which one the following is an extensive property ? |
| Answer» Answer :C | |
| 24. |
The solubility of methanol in water is due to the fact that |
|
Answer» both have BENT shape |
|
| 25. |
Which of the following statements is most applicable to hydrogen ? It can act |
|
Answer» ASA REDUCING agent |
|
| 26. |
What is the percent weight composition of water in plaster of paris? |
|
Answer» SOLUTION :Chemical formula of plaster of paris is ` CaSO_(4). (1)/(2) H_(2)O` Formula weight of plaster of paris = 136 + 9 = 145 145 grams of plaster of paris = 9 grams water 100 grams of plaster of paris = ? % weight composition of water `= (9xx100)/(145) = 6.2` |
|
| 27. |
What do you mean by olefin ? Give example ? |
| Answer» Solution :Alkenes are also known as olefins (OIL forming) since the first MEMBER, ethylene or ethene `(C_(2)H_(4))` was found to FORM an oily LIQUID on reaction with CHLORINE. | |
| 28. |
Which of the following reaction(s) produces propene as one of the important organic product? |
|
Answer» `CH_3- OVERSET(O)overset(||)C-CH_3 + Zn (Hg) overset(HCl)to ` (b) gives alkene, Hoffmann.s elimination. (c) also gives propene as 
|
|
| 29. |
Which products are obtained on reaction of but-1-ene with HBr in presence of peroxide ? |
| Answer» Solution :`underset("But-1-ene")(CH_(3)CH_(2)CH=CH_(2))overset(+HBr)RARR underset("2-Bromobutane")underset("(MAIN product)")(CH_(3)CH_(2)underset(Br)underset(|)(CH)-CH_(3))+underset("Minor amount")(CH_(3)CH_(2)CH_(2)CH_(2)Br)` | |
| 30. |
Which set of conditions represent the easiest way to cool a gas ? |
|
Answer» LOW TEMPERATURE and HIGH PRESSURE |
|
| 31. |
Which of the following enzymes converts starch into maltose? |
|
Answer» Zymase |
|
| 32. |
Which of the following does not give yellow ppt. with I_(2)//NaOH? |
| Answer» Solution :HCHO does not give iodoform test. | |
| 33. |
Which mixture of water and H_(2)SO_(4) represents a soltion with a concentration that is closest to 30% by mass H_(2)SO_(4)? |
|
Answer» 30G `H_(2)SO_(4)+100gH_(2)O` |
|
| 34. |
Which of the following has maximum bond angle ? H_(2)O, CO_(2), NH_(3), CH_(4) |
| Answer» Solution :`CO_(2)` due to its LINEAR STRUCTURE. | |
| 35. |
Write the IUPAC names of the following compounds : (i) C_(6)H_(5)CH_(2)CH_(2)OH (ii) (CH_(3))_(2)CHC equiv CH (iii) (CH_(3))_(2)CHCH_(2)CHO (iv) BrCH_(2)CH_(2)CH_(2)Cl (v) CH_(3)-CH_(2)-underset(Br)underset(|)overset(CH_(3))overset(|)(C)-underset(Br)underset(|)(CH)-CH_(2)-Cl (vi) CH_(2)=CH-C equiv N (vii) CH_(3)CH=CHCH_(2)NH_(2) (viii) CH_(3)CH=CHCOOH (ix) CH_(3)O-CH(CH_(3))_(2) (x) (C_(2)H_(5))_(2)CH.CH_(2)OH |
|
Answer» Solution :(i) 2-Phenylethan-1-ol (II) 3-Methylbut-1-yne (III) 3-Methybutanal (iv) 1-Bromo-3-chloropropane (v) 2, 3-Dibromo-1-chloro-3-methylpentane. (VI) Prop-2-enenitrile (vii) But-2-en-1-amine (viii) But-2-enoic acid (IX) 2-Methoxypropane (x) 2-Ethylbutan-1-ol. |
|
| 36. |
What is the similarity and difference in sigma and pi orbitals ? |
|
Answer» Solution :Slmllarity : `sigma and pi` both are BMO (bonding molecular orbitals) and ELECTRON DENSITY is present between two atomic NUCLEUS in both. Difference : The electron density between two CENTRE in `sigma` MO is symmetrical while in `pi` MO electron density is above or below the PLANES and divided in two parts. |
|
| 37. |
What is a bond ? |
| Answer» Solution :A BOND formed between TWO ATOMS by the sideways overlapping of two half FILLED p-orbitals is called as a PI bond. | |
| 39. |
Which of the following products is formed when n-heptane is passed over (Al_(2)O_(3)+Cr_(2)O_(3)) catalyst at 773K? |
|
Answer» Benzene 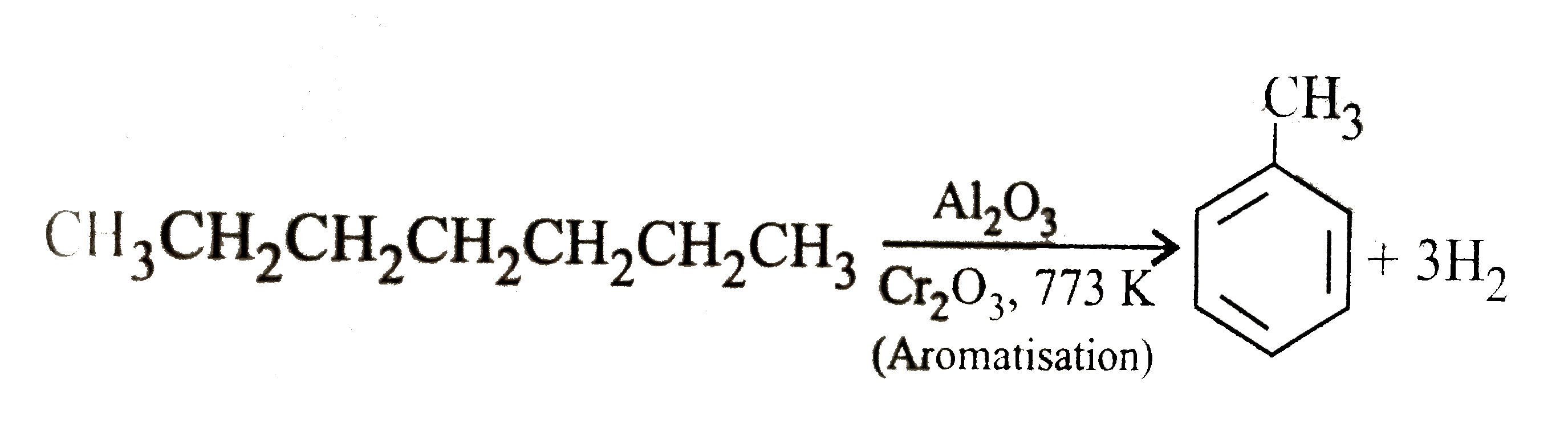
|
|
| 40. |
When NaCl is added to aqueous solution of glucose |
|
Answer» FREEZING point is LOWERED |
|
| 42. |
Write the possible resonance structures for SO_(3). |
Answer» SOLUTION :
|
|
| 43. |
Write the Newman's projections of ethane. |
Answer» SOLUTION :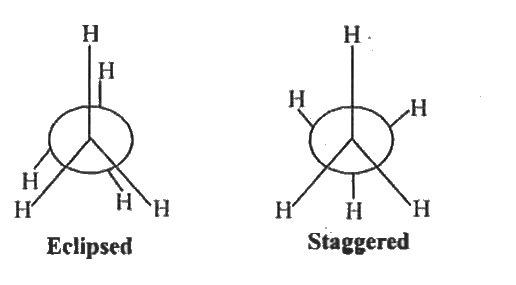
|
|
| 44. |
Which isotope of hydrogen (i) does not contain neutrons, (ii) contains equal number of protons and neutrons , (iii) is radioactive. |
| Answer» SOLUTION :(i) Protium(II) DEUTERIUM(III) TRITIUM | |
| 46. |
Which is more stable among Tl^(+3) and Tl^(+1). Why? |
|
Answer» Solution :`Tl^(1+)` is more stable than `Tl^(3+)` . It is DUE to inert pair effect. With increases in SIZE of atom in a GROUP. The s electrons present in the outermost orbit are reluctant in involving bond FORMATION. Hence, in thallium, only p ELECTRON involves in bond formation. |
|
| 47. |
Which one of these is most reactive? |
|
Answer» Na |
|
| 48. |
What will be the change in pH by adding of 0.1 M CH_3COONa in 0.1M CH_3COOH at 298K temperature ? (pK_a of CH_3COOH = 4.74 ) |
| Answer» SOLUTION :`CH_3COONa`= 2.87 PH of MIXTURE =4.81, THUS INCREASE in pH 1.14 | |
| 49. |
Which of the following compounds will have the smallest pK_(a) value ? |
|
Answer» BENZOIC acid |
|
| 50. |
What do you mean by Biochemical Oxygen Demand (BOD)? |
|
Answer» Solution :In a CERTAIN volume of water sample the AMOUNT of oxygen required by beacteria to break down the organic matter is called Biochemical Oxygen Demand (BOD). The amount of BOD in the water is a measure of the amount of organic material in the water, in terms of how much oxygen will be required to break it down biologically. Clean water would have BOD value of less than 5 ppm whereas HIGHLY polluted water COULD have a BOD value of 17 ppm or more. |
|




