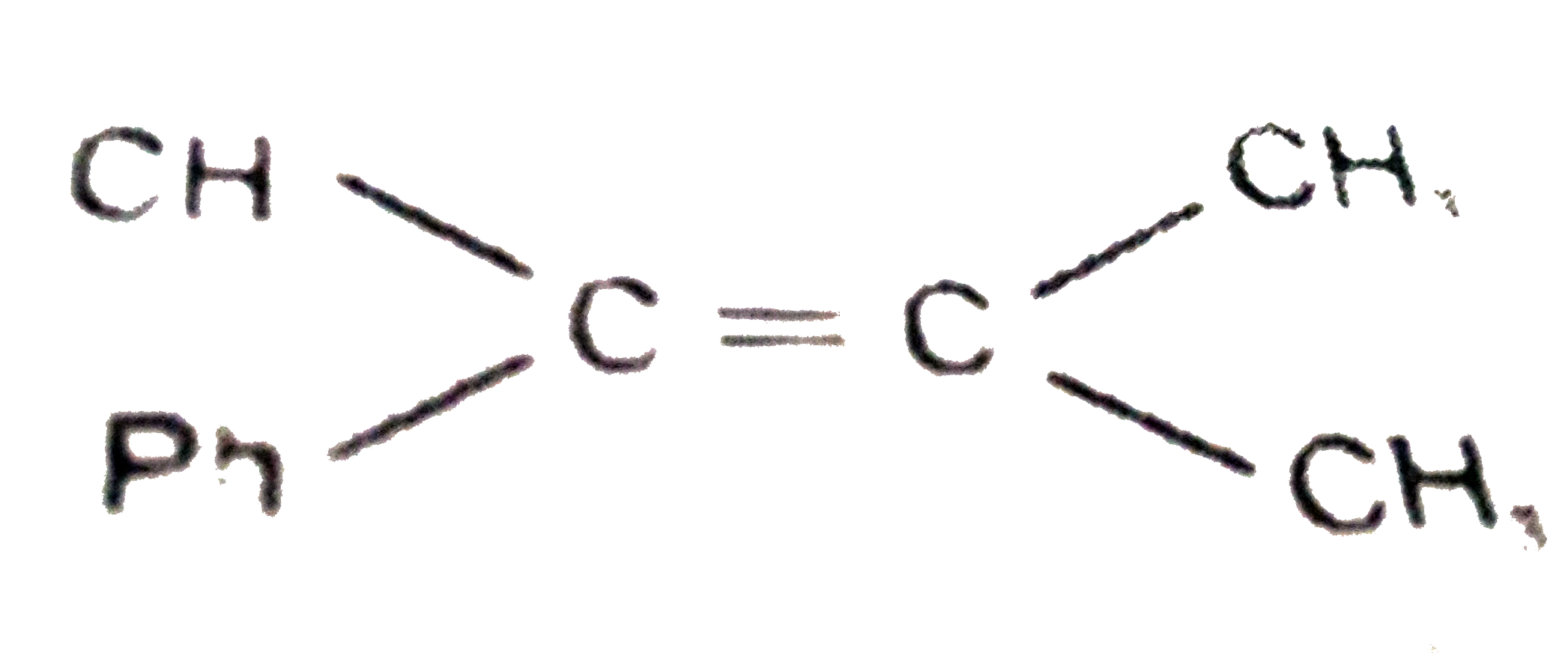Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which of the following is an example of a metal? |
|
Answer» BENZENE, Toluene |
|
| 2. |
Which bond is stronger between single bond of C-C of alkane and double bond C=C of alkene ? |
| Answer» SOLUTION :C=C of alkene is strong because it has BOND ENTHALPY of `681 KJ mol^(-1)` | |
| 3. |
Which of the following does not show electrical conduction ? |
|
Answer» Potassium |
|
| 4. |
Thermite' is a mixture of X parts of ferric oxide and Y parts of aluminium powder. X, Y respectively are |
|
Answer» 3,1 |
|
| 5. |
The work function (phi) of some metals is listed below. The number of metals which will show photoelectric effect when light of 300 nm wavelength falls metal is one the {:("Metal",Li,Na,K,Mg,Cu,Ag,Fe,Pt,W),(phi eV,2.4,2.3,2.2,3.7,4.8,4.3,4.7,6.3,4.75):} |
|
Answer» 3 |
|
| 6. |
Which of the following pairs are tautomers ? |
|
Answer» `CH_3CH_2CHO " and" CH_3COCH_3`  More delocalisation, more DISPERSAL of CHARGE and more STABILITY |
|
| 7. |
To what stable isotope ""_(103)^(257)Lw decay after having been produced by artificial means ? |
|
Answer» `""_(83)^(209)Bi` |
|
| 8. |
Which of the following mechanism is followed in the halogenation of alkanes in the presence of U-V light? |
|
Answer» Nucleophilie SUBSTITUTION |
|
| 9. |
Which of the following oxides shows electrical properties like metals ? |
|
Answer» `SiO_(2)` |
|
| 10. |
Which is the correct Lewis structure of Helium? |
|
Answer» `overset(.)He.` |
|
| 11. |
Whatis emissionspectrum ? |
| Answer» SOLUTION :Whenelectronabsorb the highand combackfromhigherenergylevelto aroundstatethanemitsradiation.It iscalledemission SPECTRA . | |
| 12. |
When Deltan_(g) is negative in chemical equilibrium reaction then : |
|
Answer» `K_(p)ltK_(p)` |
|
| 13. |
Which of the following statements regarding the resonance energy of benzene is correct ? |
|
Answer» It is the energy required to break the C-H bond in benzene |
|
| 14. |
Write the structures of the monomers of the following polymers: (i) Natural rubber (ii) Synthetic rubber(Buna-S) (iii) Nylon-6,6 (iv) Polythene (v) PVC (vi) Teflon (viii) Bakelite (ix) Polyropylene (x) Neoprene (x) Neoprene (xi) Plexiglass (xii) Buna-N |
Answer» Solution : (iii) Hexamethylene diamine `[H_(2)N(CH_(2))_(6)NH_(2)]` and adipic ACID `[HOOC(CH_(2))_(4)COOH]` (iv) Ethylene `(H_(2)C=CH_(2))` (v) VINYL chloride `(H_(2)C=CHCl)` (vi) Tetrafluoro ethylene `(F_(2)C=CF_(2))` (vii) Ethylene glycol `(HOCH_(2)-CH_(2)OH)` and phthalic acid (viii) PHENOL and formaldehyde (ix) Propylene `(CH_(3)CH=CH_(2))` (x) Chloroprene `(H_(2)C=overset(CL)overset(|)(C )-CH=CH_(2))` (xi) Methyl methylacrylate `(H_(2)C=overset(CH_(3))overset("|")("C")-COOCH_(3))` (xii) 1,3-Butadine and acrylonitrile `(H_(2)C=CH-CN)` |
|
| 15. |
Which of the following is the correct order of stability of free radicals ? |
|
Answer» benzyl `GT` ALLYL `gt 2^(UNDERSET(-)o) gt 1^(underset(-)o)` |
|
| 16. |
Which of the following statements correct ? |
|
Answer» Fullerenes have dangling bonds |
|
| 17. |
Which of the following property does not support anomalous behaviour of boron? |
|
Answer» Small size |
|
| 18. |
The total amount of oxygen consumed by microorganisms in decomposing the waste present in a certain volume of sample of water is called __________ of water. |
|
Answer» |
|
| 19. |
Which of the following compounds will give same major product on acid catalysed hydration ? |
|
Answer» `Ph-undersetunderset(CH)(|)oversetoverset(CH)(|)C-CH=CH` |
|
| 20. |
Which of the following is a lyophillic colloid? |
|
Answer» Milk |
|
| 21. |
Write IUPAC names of the following compounds |
Answer» SOLUTION : 
|
|
| 22. |
What are elimination reaction? Give an example. |
|
Answer» SOLUTION :In these reactions, TWO atoms or groups are removed from a MOLECULE without being substituted by other atoms or groups.A new `C-C` DOUBLE bond is formed between the carbon atoms to which are eliminated atoms or groups are previously attached.It is ALWAYS accompanied with change in hybridization. Example :`CH_(3) - underset"(Bromopropane)" (CH_(2)-CH_(2)) - CH_(2) - Br overset"(alc.KOH)"to underset "(Propene)"(CH_(3) -CH) = CH_(2) + KBr + H_(2)O` |
|
| 23. |
Which mixture is known as producer gas ? |
|
Answer» `O_2+N_2` |
|
| 24. |
Which of the following metals is present in chlorophyll ? |
|
Answer» MG |
|
| 25. |
Which of the following is/are correct about the solubility ? |
|
Answer» Solubility of `CaF_2 ` is more in buffer solution of pH = 3 than in pure water (b) ` S^(2-)` reacts with ` H^(+) ` ( C )` Zns hArr 2n^(+2) + S^(2-), KSP ` ` (H_2S hArr 2H^(+)+S^(2-) ,Ka)/( Zns +2H^(+)hArr Zn^(+2)+H_2S, K =(Ksp)/(Ka))` |
|
| 26. |
What is the most important application of de Broglie concept ? |
| Answer» SOLUTION :In the CONSTRUCTION of ELECTRON MICROSCOPE used for the MEASUREMENT of objects of very small size. | |
| 27. |
Which of the following statement is incorrect regarding the structure of borax? |
|
Answer» NUMBER of B - B bonds are zero |
|
| 28. |
What type of stoichiometric defect is show by : ( i) ZnS (ii) AgBr ? |
|
Answer» Solution : (i) ZNS SHOWS Frenkel defect because its have a LARGE difference in size. (ii)AgBr shows both frenkel and SCHOTTKY defects. |
|
| 29. |
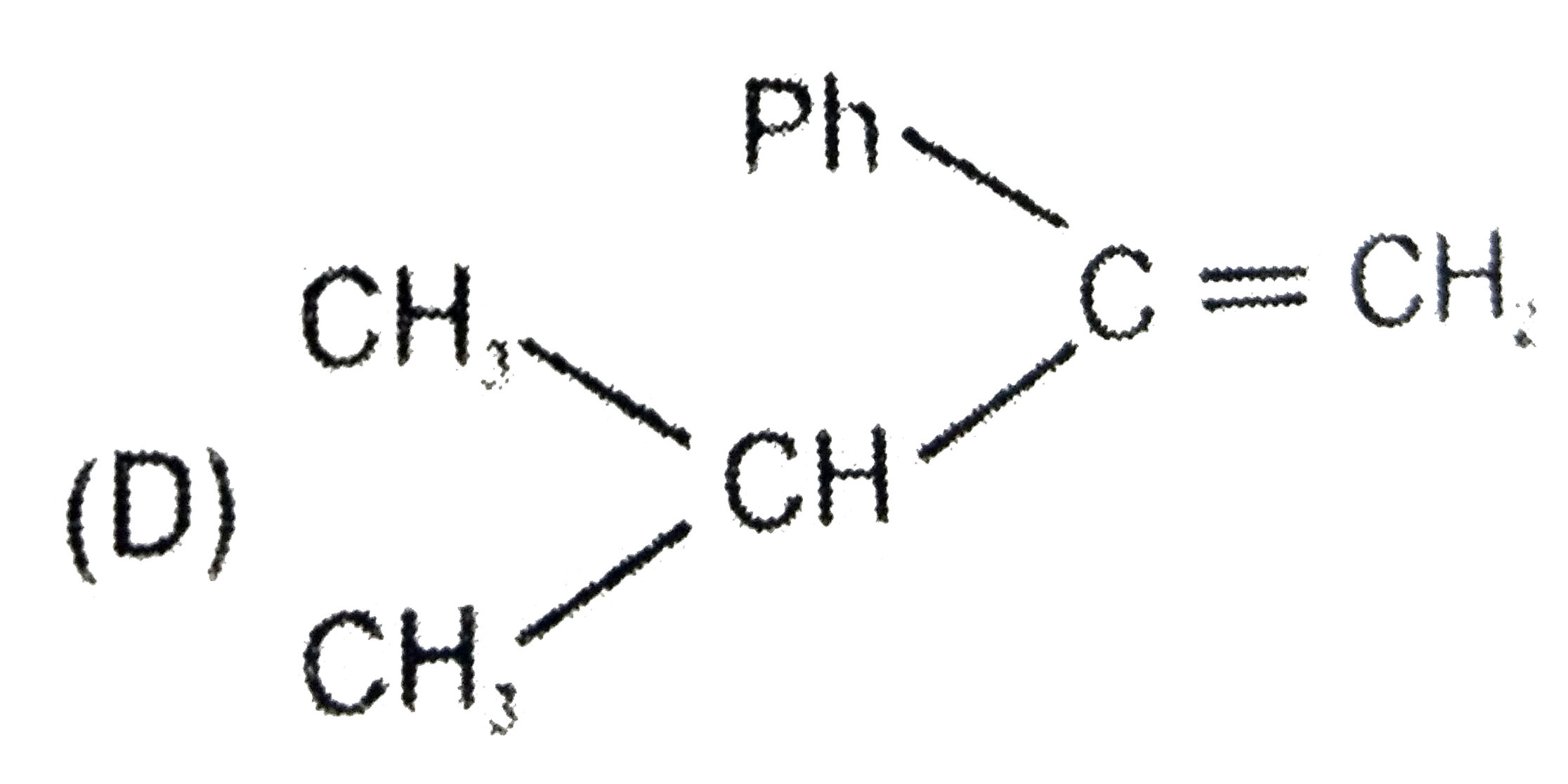
Write the IUPAC names of the compound: CH_3CH = C(CH_3)_2 |
Answer» SOLUTION :
|
|
| 30. |
While suphurdioxide and hydrogen peroxide can act as an oxidising as well as reducing agents in their reaction ozone and nitric acid act onlyu as oxidant why? |
|
Answer» Solution :`H_(2)O` are produced during the PROCESS (B)the purpose of writing `O_(2)` two times suggest that `O_(2)` is being obtained from each of the two reactant the path of reaction (a) and (b) can be DETERMINED by using `H_(2)O` or `D_(2)^(O)` in reaction (a) or by using `H_(2)O_(2)^(18)` or `O_(3)^(18)`in REACTOIN (b) |
|
| 31. |
Write the resonating structures for the following molecules. Benzene |
Answer» SOLUTION :
|
|
| 32. |
Which are produce during high temperature combustion processes ? |
|
Answer» OXIDES of hydrogen |
|
| 33. |
Which one is not a mineral of sodium ? |
|
Answer» ROCK salt |
|
| 34. |
The systematic name of PhCH_(2)COOH is |
|
Answer» benzeneacetic ACID |
|
| 35. |
Which of the following sequence is best suited to convert benzene to 3-chloro aniline? |
|
Answer» NITRATION, REDUCTION, CHLORINATION |
|
| 36. |
What happens to the internal energy of the system if : (a) Works is done on the system (b) Work is done by the system. |
| Answer» SOLUTION :(a) If work is DONE on the system INTERNAL energy will increase. (b) If work is done by the system, internal energy will decrease. | |
| 37. |
The reductive ozonolysis of an alkene gives a mixture of acetone and formaldehyde. What is the structural formula of the alkene? |
Answer» SOLUTION :
|
|
| 38. |
Which of the following alcohols will give iodoform test? |
|
Answer» Methanol |
|
| 39. |
Whichpairis truefromthe following ? |
|
Answer» AUFBAU(i) MVR |
|
| 40. |
What is conjugate acid and conjugate base ? Give example. |
Answer» Solution :In CONJUGATE acid one PROTEN is more  In conjugate acid one H and the charge is more upto +1 unit. In conjugate BASE one proten is LESS  In conjugate base one H and the charge is less upto +1 unit. |
|
| 41. |
Which of the following does not give allyl halide as major product ? |
|
Answer» `CH_2 = CH - CH_3 UNDERSET(HV)overset(SO_2Cl_2)to ` |
|
| 42. |
Total number of orbitals associated with third shell will be... |
|
Answer» 2 |
|
| 43. |
When the temperature of a reversible rection is increased from 327 to 427^(@)C, the equilibrium constant K_(p) is decreased by four times. Find the enthalpy of the reaction in this temperature range. |
|
Answer» Solution :`"log"((K_(p))_(2))/((K_(p))_(1))=(DELTAH^(@))/(2.303R)[(T_(2)-T_(1))/(T_(1)*T_(2))]` `"log"1/4=(DeltaH^(@))/(2.3xx8.3)[(700-600)/(600xx700)]` `-0.6201=(DeltaH^(@))/19.09xx100/420000` `thereforeDeltaH^(@)=-(0.6201xx19.09xx420000)/100` = -49734 J = -49.734 kJ |
|
| 44. |
Which of the following reaction is a redox reaction ? |
|
Answer» `4H_(3)PO_(4)+H^(+)toPH_(4)^(+)+3H_(2)PO_(3)` 
|
|
| 45. |
Which of the following cannot be considered as a step of mechanism in chain reaction of methane with Cl_2 ? |
|
Answer» `Cl_2 to CL^(**)` |
|
| 46. |
Which one of the following is a primary standard ? |
|
Answer» `KMnO_(4) ` |
|
| 47. |
Whichmetalplaysan importantroleinneuromuscularfunctions, internuronaltransimission cellmembraneintegrityandbloodcoagulation? |
| Answer» ANSWER :C | |
| 48. |
Which of the following structure is similar to graphite ? |
|
Answer» BN |
|
| 49. |
Which of the followng act both as oxidising as well as reducing agents ? |
|
Answer» `HNO_(2)` |
|
| 50. |
Which one of the following binary liquid mixtures exhibits positive deviation from Raoults law ? |
|
Answer» ACETONE + CHLOROFORM |
|