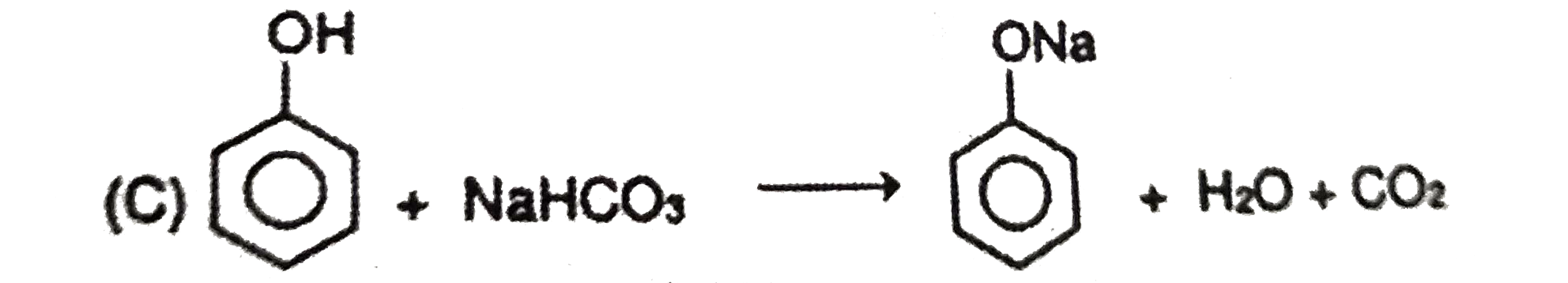Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which of the following salts will have the highest pH in water ? |
|
Answer» KCL |
|
| 2. |
Water softening by Clark's process uses |
| Answer» Answer :B | |
| 3. |
What kind of molecules show disproportionation reactions ? Give one example of a compound each of nitrogen and phosphorus which show disproportionation reactions. Write chemical equation in each case. |
|
Answer» Solution :Compounds in which one of elements exists in three different OXIDATION STATES can show disproportionation reactions. For example, `3Hoverset(+3)NO_(2)toHoverset(+5)NO_(3)+2overset(+2)NO+H_(2)O","4H_(3)OVERSET(+3)PO_(3)overset(Delta)to3H_(3)overset(+5)PO_(4)+overset(-3)PH_(3)` |
|
| 4. |
When potassium acetate is electrolysed, we get |
|
Answer» methane |
|
| 5. |
To study the decomposition of hydrogen iodide, a student fills an evacuated 3 litre flask with 0.3 mol of HI gas and allows the reaction to proceed at 500^(@)C. At eauilibrium he found the concentration of HI which is equal to 0.05 M. Calculate K_(C) and K_(P) for this reaction. |
|
Answer» Solution :`V=3L` `[HI]_("initial")=(0.3" mol")/(3L)=0.1M` `[HI]_(EQ)=0.05M` `2HI(g)hArrH_(2)(g)+I_(2)(g)`  `K_(C)=([H_(2)][I_(2)])/([HI]^(2))` `=(0.025xx0.025)/(0.05xx0.05)` `K_(C)=0.25` `K_(P)=K_(C)(RT)^(Deltang)` `Deltan_(g)=2-2=0` `K_(P)=0.25(RT)^(0)=0.25` |
|
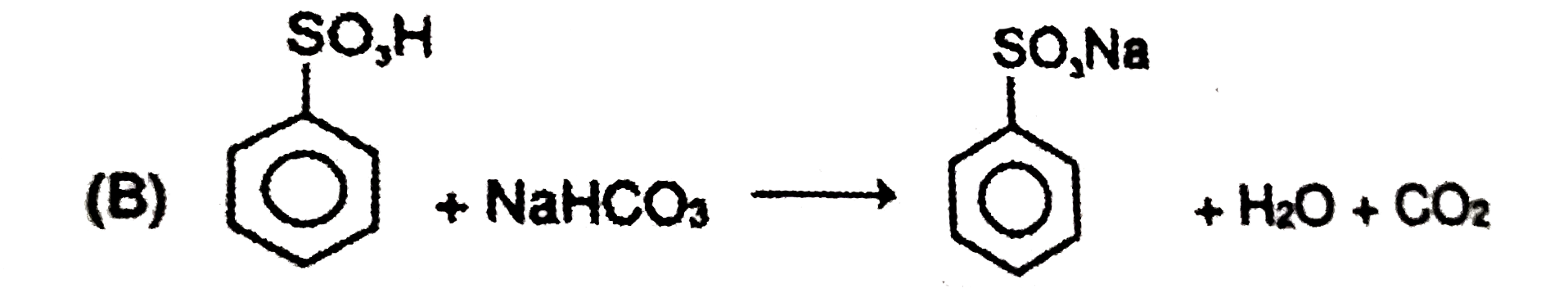
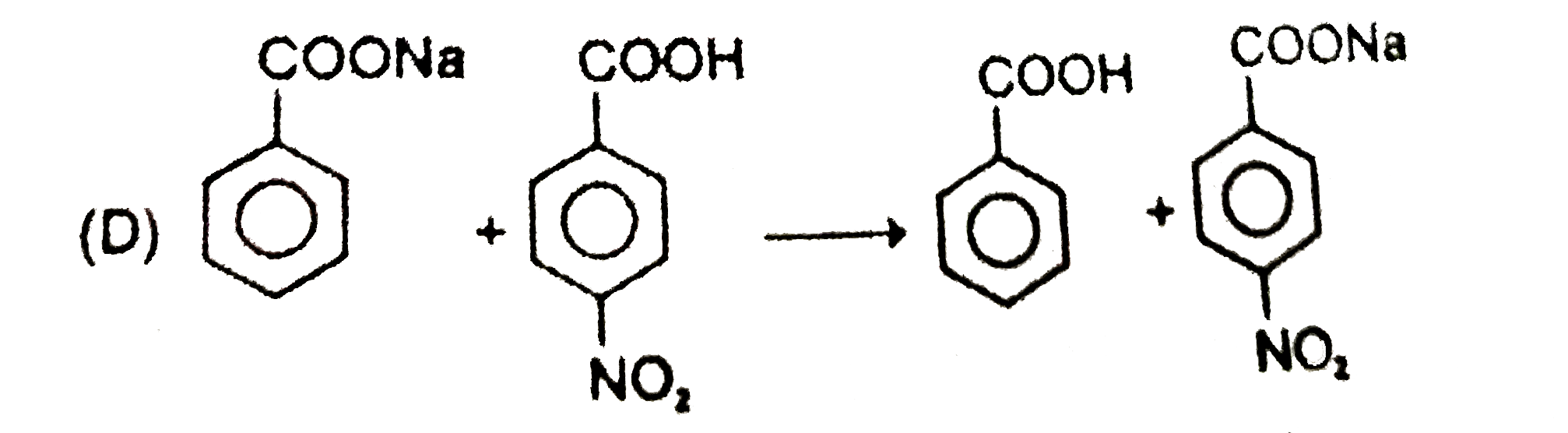
| 6. |
Which of the following reagents can react with phenol to produce phenolphthalein ? |
|
Answer» phthalic anhydride/`H_(2)SO_(4)` 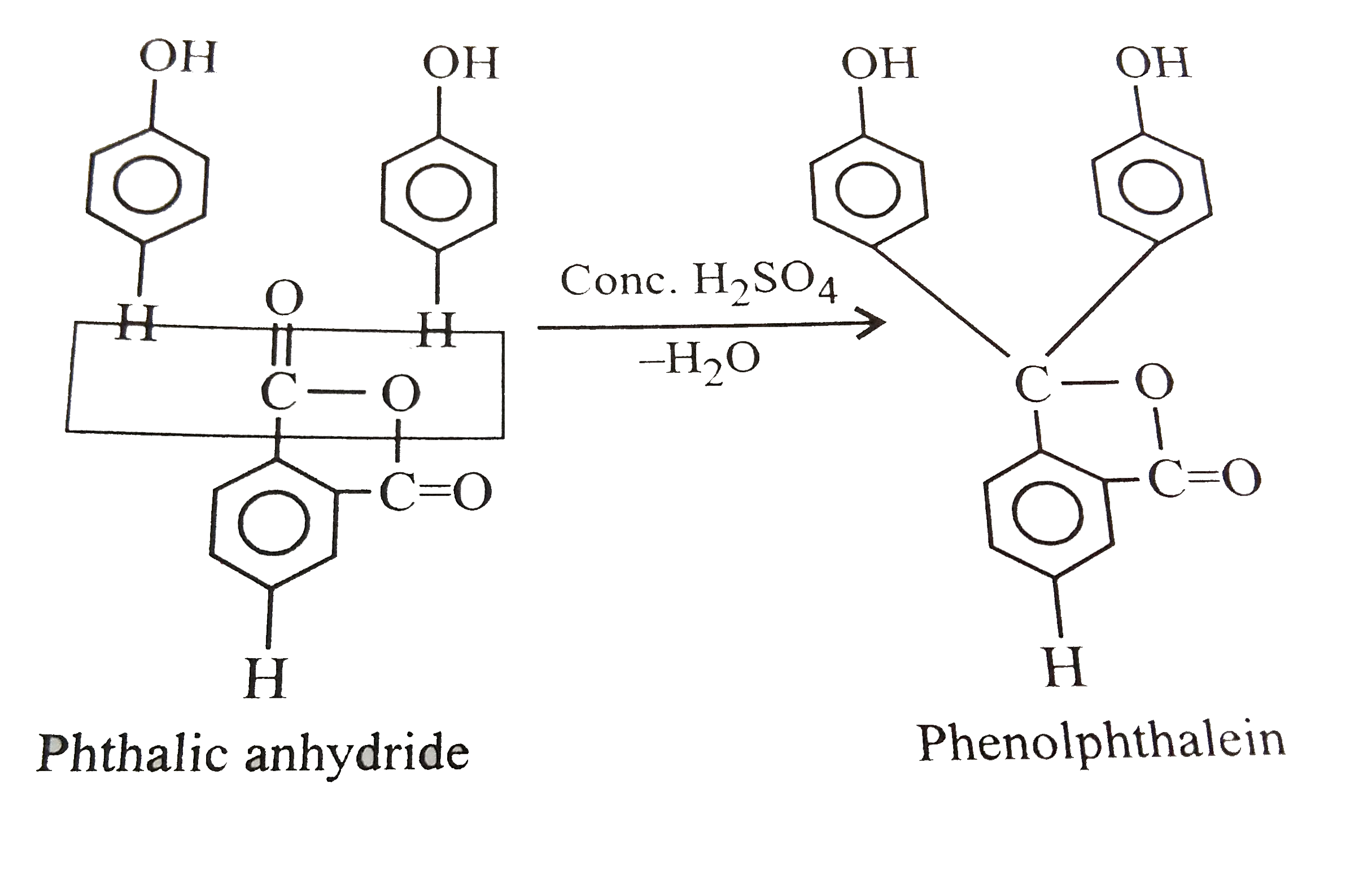
|
|
| 7. |
Which of the following is used as a source of O_(2) in space capsules, submarines etc? |
|
Answer» `K_(2)O` `2Na_(2)O_(2)+2CO_(2)rarr2Na_(2)CO_(3)+O_(2)` |
|
| 8. |
Whichobservatin arenotexplainby electronmagnetictheoryof physics ? |
|
Answer» Solution :(i) Thenatureof emissionof radiationfrom hotbodies( black- bodyradiation ) (II) Photoelecticeffect: Ejectionof electronsfrommetalsurfacewhenradiationstrikes (III) Variationof heatcapacityof SOLIDS as afunctionof temperature . (iv) Linespectraof atomswithspecialreferenceto hydrogen. |
|
| 9. |
Whatis aromatisation |
Answer» Solution :Alkanes with sixto TEN carbon atoms are converted into homologous of benzen at higher TEMPERATURE and in the PRESENCE of a cataylst this process is known as aromatisationfor example n hexane passed over `Cr_(2)O_(3)`supported on alumina at 873 GIVES benzene 
|
|
| 10. |
The values of van der Waals' constant 'a' for O_2, N_2, NH_3 " and " CH_4 " are " 1.360, 1.390, 4.170 " and " 2.253 L^2 " atm " mol^(-2) respectively. The most easily liquefiable gas among these is |
|
Answer» `N_2` |
|
| 11. |
The two main chemicals responsible for the depletion of ozone layer are _________ and ____________ |
|
Answer» |
|
| 12. |
The relative order of basicity of conjugate bases is |
|
Answer» `OH^(-) lt OR^(-) lt HC -= C^(-)` Acidic charatcer of conjugate acids is `H_(2)O gt ROH gt HC -= CH` `:.` the order of basicity is `underset("weakest base")(OH^(-)) lt underset("STRONGEST base")( RO^(-)lt HC -= C^(-))` |
|
| 13. |
Which of the following is an example of carbocyclic alicyclic compound? |
|
Answer» Pyrrole |
|
| 14. |
When LiNO_(3) is heated, it gives oxide, Li_(2)O whereas otheralkali metals nitrates decompose to give corresponding |
|
Answer» NITRITE |
|
| 15. |
What is Resonance structure ? Why the resonances structure required explain with' suitable example.OR Give resonance of O_(3) , its requirement, delimitation and advantages |
|
Answer» SOLUTION :Resonance STRUCTURES : Different Lewi , structure of single molecule / ion is KNOWN as resonance structure. According to the concep of resonance, whenever a single Lewis structure cannot describe a molecule accurately so mor than one Lewis structure means resonance structure describe. Resonance structure of ozone `(O_(3))` : To draw resonance structure the position of nucleus of atom is not change. Every reasons structure the position of bonding and non-bonding electron is change but Lewis structure is not seen different. ·The different resonance structure is represented by a double HEADED arrow. All resonance structure are similar energy. Two resonance structure of `O_(3)` molecule (I) and (II) and (III) is resonance hybrid.  Limitation of resonance : Any one is not seen right structure. The O- O bond length is 148 and O = O bond length is 121 pm. Thus single and double bond length is not experimentally present in `O_(3)` . `therefore ` Correct structure of `O_(3)` is not (I) and (II) Resonance : Correct structure is resonance hybrid. Experimentally determined oxygen - oxygen (0 - 0) bond length in the `O_(3)` molecule are same 128 pm. So, (III) is a real or accurate structure of `O_(3)` in which single and double bond is not stable. Resonance structure are necessary because it GIVES correct different Lewis structure. Orientation : Single assumption structure can.t give correct bond length & bond energy. Advantage : It gives assumption of correct structure. |
|
| 16. |
when the electrons move towards the substituent attached to the conjugated system_______effect occurs . |
|
Answer» `-R` |
|
| 18. |
Which of the following will have the lowest pH when one mole of each of the following is hydrolysed will water ? |
|
Answer» `(CH_(3)C)_(2)O` |
|
| 19. |
The temperature below which a gas obeys Joule- Thomoson effect is called ___________. |
|
Answer» BOYLE TEMPERATURE |
|
| 20. |
Which among the following is a set of nucleophiles ? |
|
Answer» `BF_3,H_2O,NH_2^-` |
|
| 21. |
What electronic transition in Li^(+2) produces the radiation of the same wave length as the first line in the Lyman series of hydrogen ? |
| Answer» Answer :D | |
| 22. |
Which element do you think would have been names by-Lawrence Berkely Laboratory & Sseaborg's group? |
|
Answer» SOLUTION :(1) LAWRENCIUM, LR and BERKELIUM, Bk. (2) Seaborglum, Sg. |
|
| 23. |
Which of the following is white ? |
|
Answer» `KMnO_(4)` |
|
| 24. |
Which iscorrect statement about sigma - and po- molecular orbitals? Statementsare : pi- bonding orbitals are ungerade pi-antibonding orbitals are ungerade sigma-antibondding orbitals are gerade |
|
Answer» 1 only ungerade, `PI` are ungerade whereas `pi^(**)` are gerade . |
|
| 25. |
Which of the following describes the following titration curve? |
|
Answer» A STRONG BASE is ADDED to a strong acid |
|
| 26. |
What are the raw materials needed for the manufacture of washing soda by Solvay process ? |
| Answer» SOLUTION :`NACL , CaCO_(3) and NH_(3)`. | |
| 27. |
The reagent used for converting acetylene to oxalic acid is |
|
Answer» `HgSO_(4)`/aqueous `H_(2)SO_(4)` |
|
| 28. |
What is peroxide effect |
| Answer» SOLUTION :The ADDITIONOF HBrto an alkanein the PRESENCEOF organicperoxidegivetheanti- Markovnikoffproduct.Thiscalledas PEROXIDEEFFECT | |
| 29. |
What happens when ? (i) Quick lime is heated with coke ? (ii) Carbon monoxide reacts with CI_(5) (iii) Plants absorb CO_(2) ? |
|
Answer» SOLUTION :(i) `CaO +3C RARR underset (("CALCIUM Carbide")) (CaC_(2)+CO)` (ii) `CO+CI_(2) rarr underset (("phosgene")) (COCI_(2))` (III) `6CO_(2) +6H_(2)O rarr underset ("glucose") (C_(6)H_(12)O_(6))+6O_(2)`. |
|
| 30. |
Which of the following pair is expected to have the same bond order ? |
|
Answer» `O_(2), N_(2) ` On the base of molecular orbital THEORY we can calculate bond order of molecules ions as BO = `(1)/(2) (N_(B) - N_(a))` Molecular orbital electronic configuration (MOEC) of `N_(2)` is : `sigma 1s^(2), sigma(8*) 1s^(2), sigma 2S^(2), sigma^(**) 2s^(2), pi 2p_(X)^(2) = pi 2p_(y)^(2) , sigma 2p_(x)^(2) ` Bond order of `N_(2)^(+) = (1)/(2) (10 - 4) =3 ` MOEC of `N_(2)^(+) = sigma 1s^(2) , sigma^(**) 1s^(2) , sigma 2s^(2) , sigma^(**) 2s^(2), pi 2p_(x)^(2) = pi 2p_(y)^(2) , sigma 2p_(z)` BO of `N_(2)^(+) = (1)/(2) (9 - 4) = 2.5 ` MOEC of `N_(2)^(-) = sigma 1s^(2) , sigma^(**) 1s^(2), sigma 2s^(2), sigma^(**) 2s^(2),pi 2p_(x)^(2) = pi 2p_(y)^(2), sigma 2p_(z)^(2) , pi 2p_(x)^(1) = pi^(**) 2p_(y)` BO of `N_(2)^(-) = (1)/(2) ( 10 - 5)= 2.5` MOEC of `O_(2) =sigma 1s^(2), sigma^(**) 1s^(2) , sigma^(**) 2s^(2) , sigma 2p_(z)^(2) , pi 2p_(x)^(2) = pi 2p_(y)^(2) , pi^(**) 2p_(x)^(2) = pi^(**)2p_(y)^(1)` BO of `O_(2) = (1)/(2) (10 - 6) = 2 ` MOEC of `O_(2)^(-) = sigma 1s^(2), sigma^(**)1s^(2), sigma 2s^(2), sigma^(**) 2s^(2) , sigma 2p_(z)^(2) , pi 2p_(x)^(2) = pi 2p_(y)^(2) , pi^(**) 2p_(x)^(2) = pi^(**) 2p_(y)^(1)` BO of `O_(2)^(-) = (1)/(2) (10 - 7) =1.5` MOEC of `O_(2)^(+) = sigma^(**) 1s^(2), sigma 2s^(2), sigma^(**) 2s^(2), sigma 2p_(z)^(2), pi 2p_(x)^(2) = pi 2p_(y)^(2) pi^(**) 2p_(x)^(2) = pi^(**) 2p_(y)` BO of `O_(2)^(+) = (1)/(2) (10 - 5) = 2.5 ` (A) Bond order of `O_(2) and N_(2)` are 2 and 3, respectively. (B) Bond order of both `O_(2)^(+) and N_(2)^(-) ` and 1.5 and 2.5 (C) Bond order of `O_(2)^(-) and N_(2)^(+) ` are 1.5 and 2.5, respectively. Bond order of `O_(2)^(-) and N_(2)^(-)` are 1.5 and 2.5 respectively. |
|
| 31. |
Which of the following reaction is an example for homogenous catalysis:- |
|
Answer» `2H_(2)O_(2(l))overset(MnO_(2)(s))to2H_(2)O_((l))+O_(2(G))` |
|
| 32. |
Which among the following elements is paramagnetic |
|
Answer» `KO_(2)` |
|
| 33. |
Write the structures of different isomers correspondingto the 5^(th) member of alkyne series. Write their IUPAC names. |
|
Answer» Solution :`5^(th)` Member of ALKYNE series is `C_(6)H_(10)`. Since first member of series is `C_(2)H_(2)`. The possible isomers and their IUPAC names are : 
|
|
| 35. |
The soldiers of Napolean army while at Alps during freezing winter suffered a serious problem as regards to the tin buttons of their uniforms. White metallic tin buttons get converted to grey powder, This transformation is related to |
|
Answer» an interaction with water vapour CONTAINED in humid AIR |
|
| 37. |
What temperature is 75^(@) F on the Kelvin scale? |
|
Answer» Solution :C) `75^(@)`F= 9/5^(@)`C + 32 = ((43 xx 5)/9)^(@)` C = `23.9^(@)`C = `(23.9 + 273)`K = 296.9 -~ 297 K |
|
| 38. |
What arethe atomicnumber of theelementswhichconstitutef- block ( lanthanoidsand actinoids ) ? |
| Answer» SOLUTION :LANTHANOIDS `=58 ` to 71, ACTINOIDS =90 to 103 . | |
| 39. |
Which of the following FCC structure contains cations in alternatetetrahedral voids? |
| Answer» Answer :B | |
| 40. |
When B is reacted with conc. H_2 SO_4, the gaseous product is |
|
Answer» `B_2 O_3` |
|
| 41. |
Which of the following reagents would be the best reactants for the following synthesis? |
|
Answer»
|
|
| 42. |
Which type of electrolytes are used in a salt bridge? |
| Answer» Solution :Only those electrolytes for which CATIONS and anions have NEARLY the same IONIC mobilities (i.e distance travelled by an ion PER second under a POTENTIAL gradient one volt) are used as electrolytes in the salt bridge. Thus KCl, `KNO_3,K_2SO_4 and NH_4NO_3` are preferred over NaCl, `NaNO_3` and `Na_2 SO_4`. | |
| 44. |
Two elements P and Q react seperately with highely electropositive metal to form binary compounds, which upon hydrolysis yield mixtures of boranes and silanes. P and Q respectively are |
|
Answer» <P>B,Al |
|
| 45. |
underset((X))("Salt of iron") overset(Na_(2)S_(2)O_(3))rarr Violet complex overset("On standing")rarr underset((Z))("Green solution") Wite oxidation number of Fe in (Z). |
|
Answer» |
|
| 46. |
The term that corrects for the attractive forces present in a real gas in the van der Waal's equation is: |
|
Answer» NB |
|
| 47. |
What is amphoteric oxide ? |
| Answer» Solution :Which oxide act as BASE with ACID and act as acid with base is KNOWN as AMPHOTERIC OXIDES. | |
| 48. |
Which of the following oxidation states are the most characteristic for lead and tin respectively : |
|
Answer» `+2,+2` |
|
| 49. |
Which of the following statement(s) is/are incorrect? |
|
Answer» `O_(2)` is paramagnetic, `O_(3)` is also paramagnetic |
|