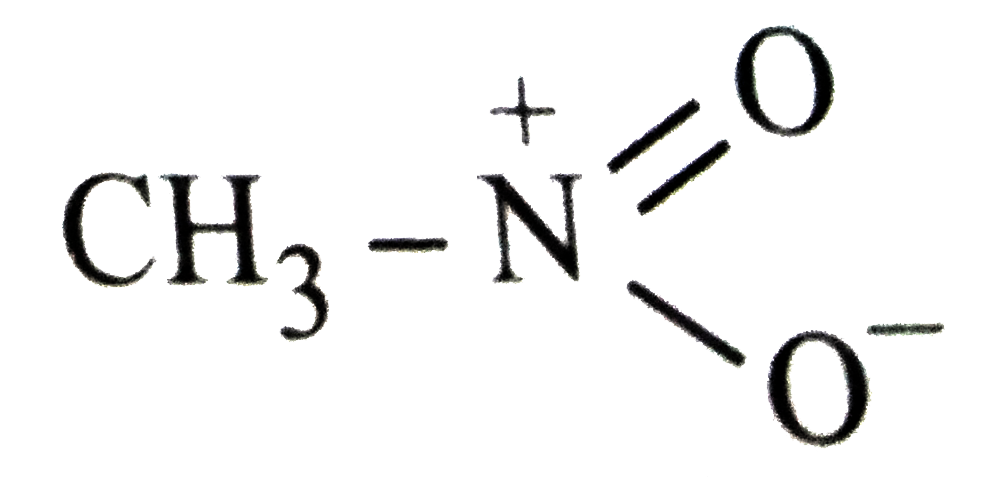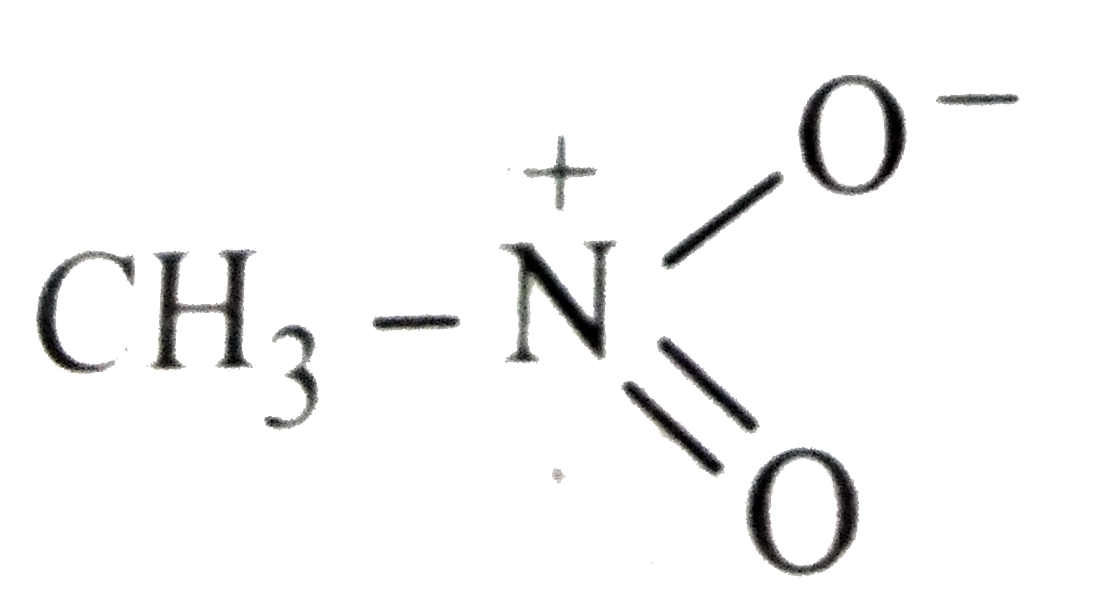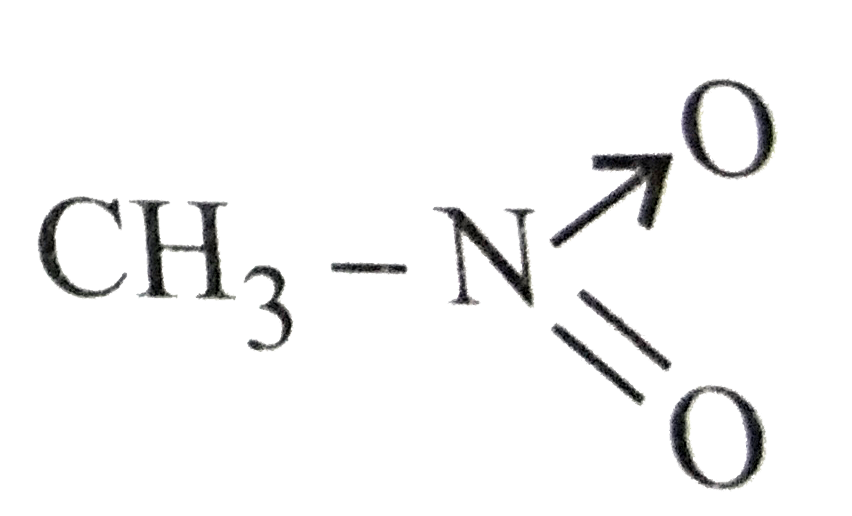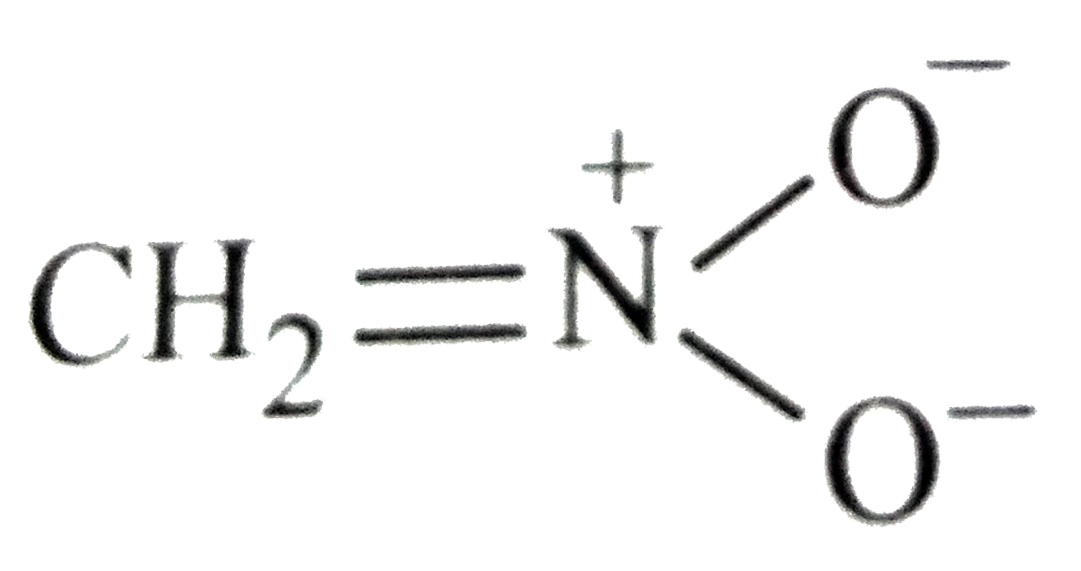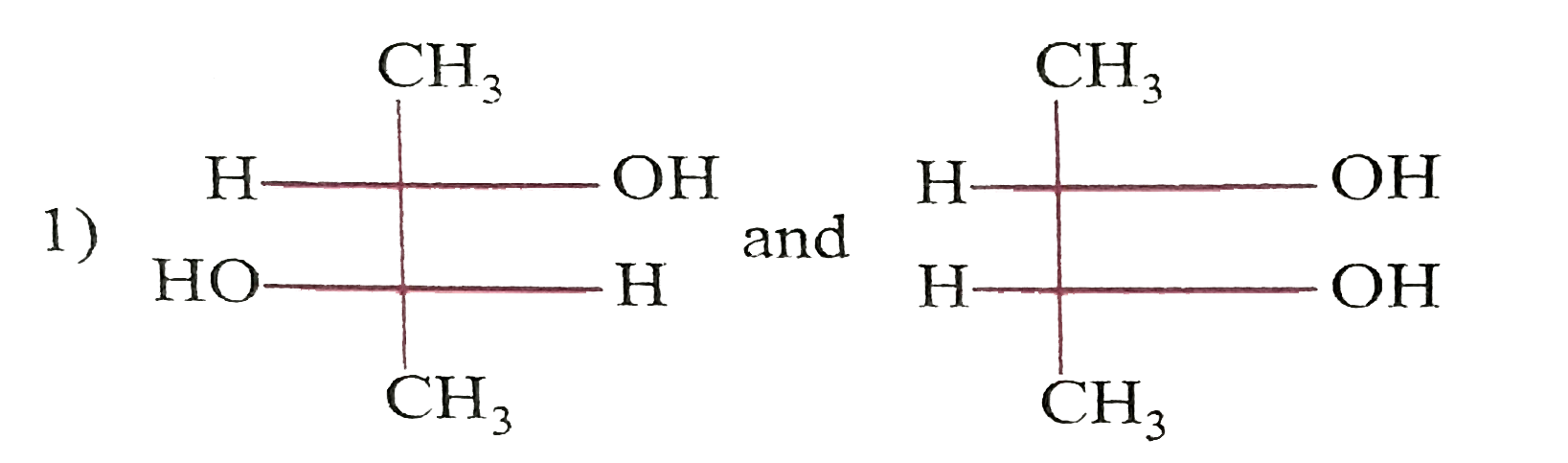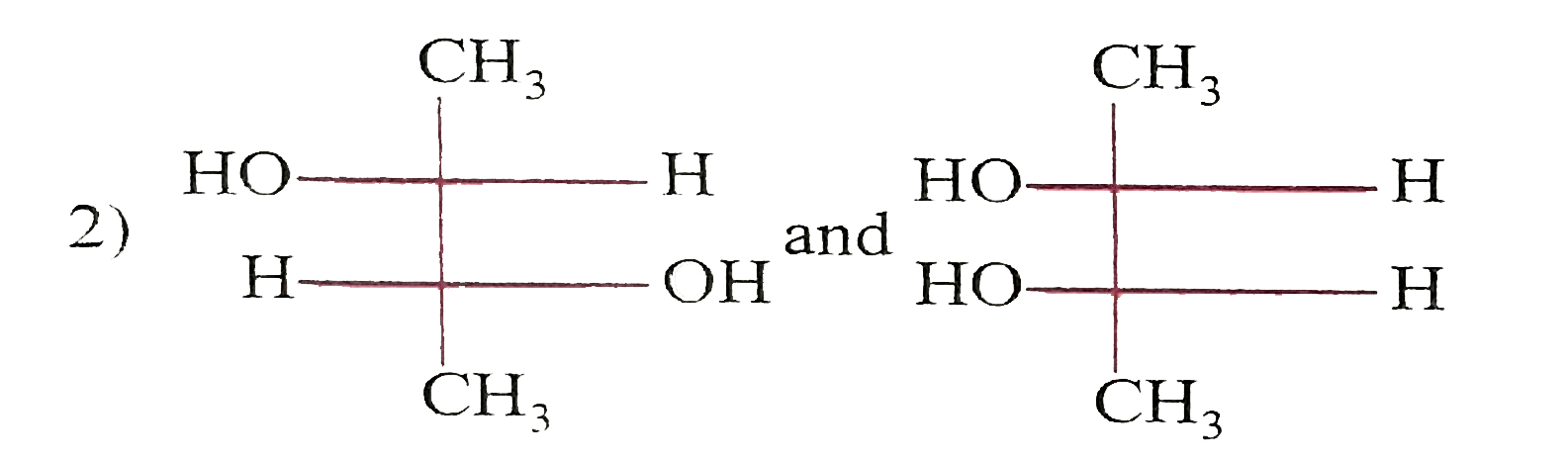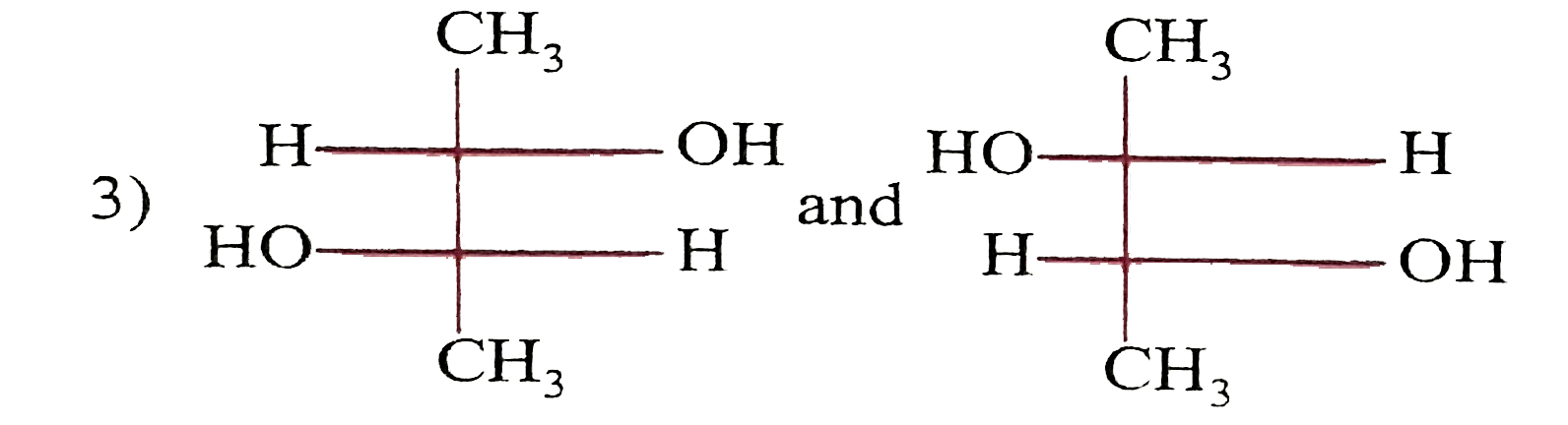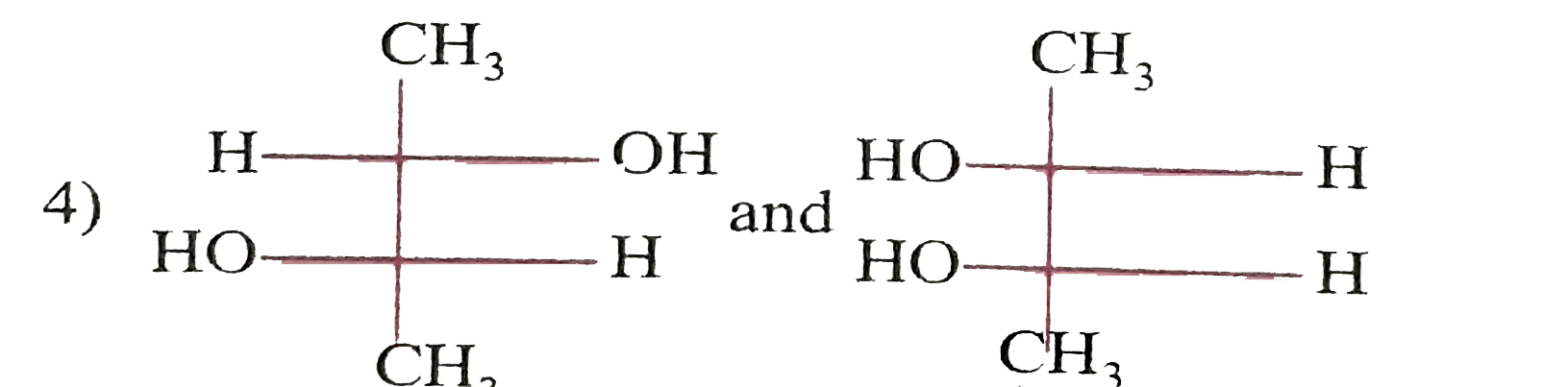Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Tyndall effect is more pronounced in |
|
Answer» hydrophilic sols |
|
| 2. |
Under what conditions is the heat of reaction equal to enthalpy change? |
|
Answer» SOLUTION :The heat of reaction is EQUAL to enthalpy change at CONSTANT PRESSURE i.e, `q_(p) = DeltaH`. |
|
| 3. |
Which of the following compunds are resolvable? |
|
Answer»
(B) Non-resolvable (c ) Resolvable (d) Resolvable |
|
| 4. |
which organic species has only one type of hybridized carbon? |
|
Answer» `CH_2=C=CH_2` |
|
| 5. |
Which of the following oxides turns yellow on heating and becomes white on cooling? |
| Answer» Answer :A::B | |
| 6. |
The treatment of ethyl formate with excess of RMgX gives |
|
Answer» `R-UNDERSET(O)underset(||)(C)-R` |
|
| 7. |
Which would be larger: an atomic mass unit based on the current standard or one based on the mass of a Be-9 atom set at exactly 9 amu? |
|
Answer» |
|
| 8. |
The term van der Waals forces refers to |
|
Answer» DIPOLE - Dipole INTERACTION |
|
| 9. |
What will be the final product ? |
|
Answer»
|
|
| 10. |
Write of the following compounds will show cis-trans isomerism ? (i)(H_3C)_2C=CH-C_2H_5 , (ii)H_2C=CBr_2 , (iii)C_6H_5CH=CHCH_3 , (iv)H_3C-CH=C Cl(CH_3) |
| Answer» Solution :Alkenes (i) and (ii) have identical ATOMS or groups on one of the carbon atoms of the double BOND and HENCE do not show GEOMETRICAL isomerism. In contrast, alkenes (iii) and (iv) have different atoms or groups on each carbon atom of the double bond and hence exhibit geometrical isomerism. | |
| 11. |
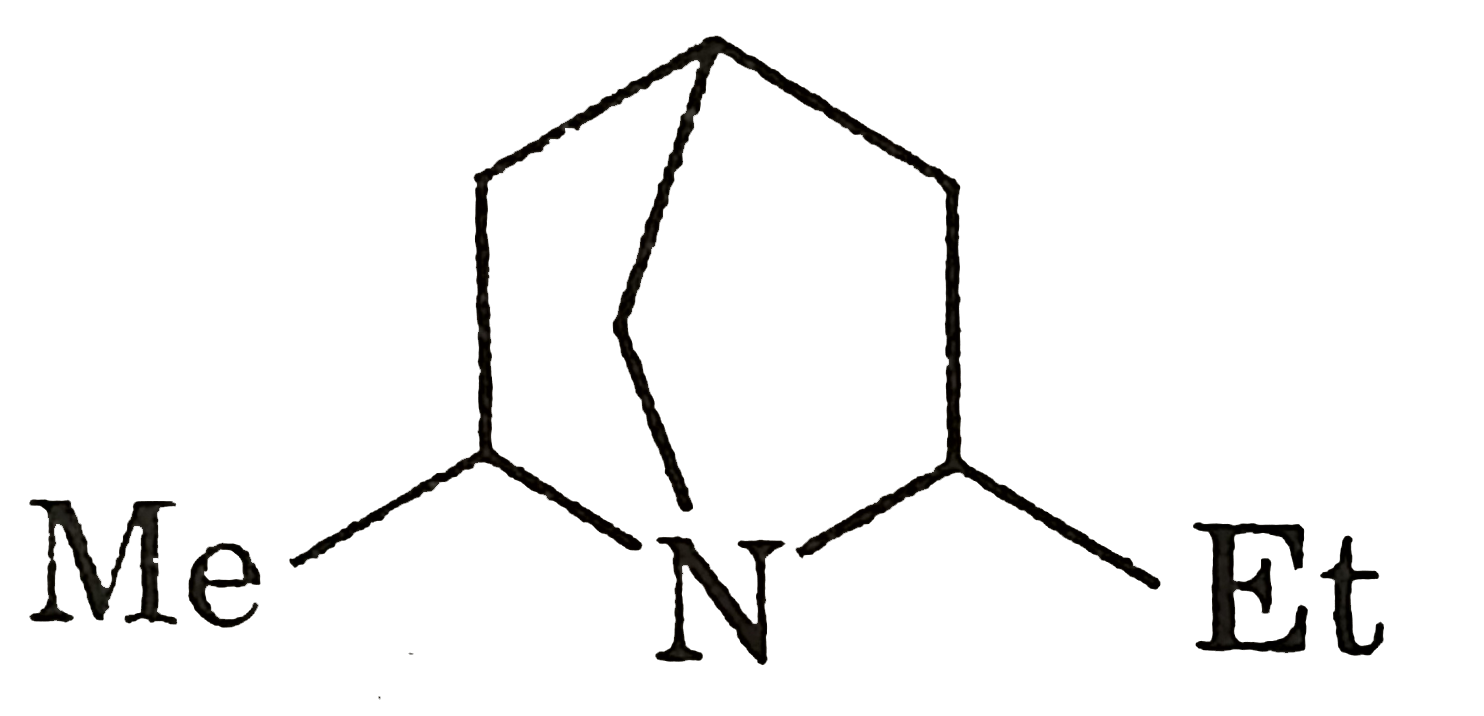
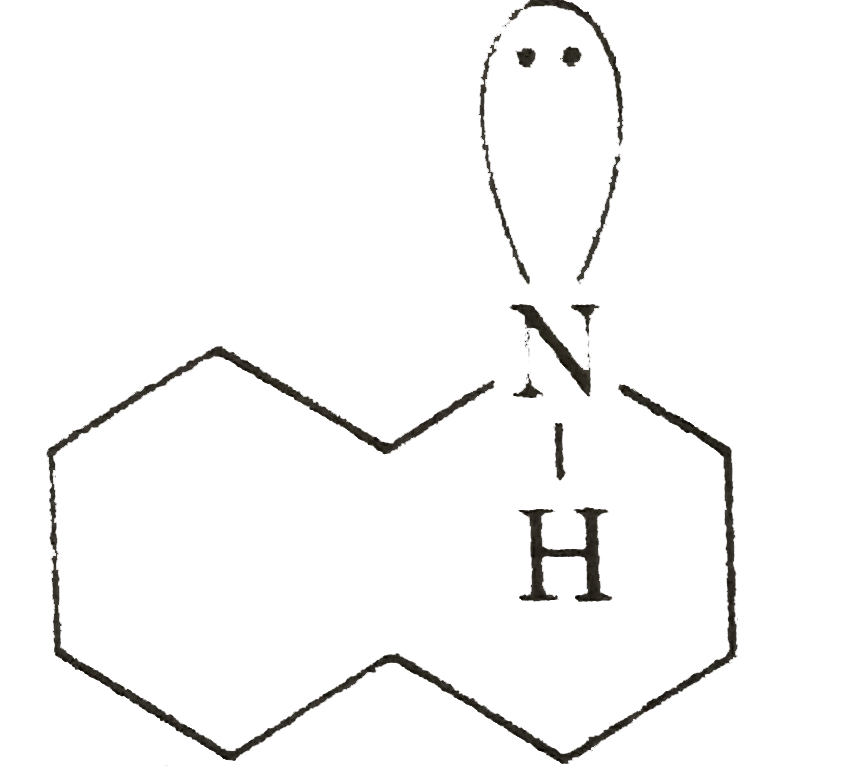
Which of the following is not structure of nitromethane molecule? |
|
Answer»
|
|
| 12. |
Which one of the following arrangements represents the correct order of electron gain enthalpy of the given atomic species? |
|
Answer» `OltSltFltCl` |
|
| 13. |
Which series, contain radioactive elements ? |
| Answer» SOLUTION :Actinoid series CONTAIN RADIOACTIVE elements. | |
| 14. |
What product will be obtained when magnesite is dissolved in hot dilute H_(2)SO_(4) |
|
Answer» `MgSO_(4),H_(2)O,CO_(2)` |
|
| 15. |
Which statement is true for meso compound? |
|
Answer» There are one or more planes of symmetry b) A single molecule is identical to its mirror image c)More than one stereogenic centre must be present |
|
| 16. |
Which of the following is not a redox reaction |
|
Answer» `KCl+K_(2)Cr_(2)O_(7)+con.H_(2)SO_(4)rarrCrO_(2)Cl_(2)+K_(2)SO_(4)+H_(2)O` OXIDATION state of Cr will remain same in reactant as well as product. |
|
| 17. |
Under identical conditions of pressure and temperature |
|
Answer» `(r_(H_2))/(r_(O_2)) = 4` `(gamma_(He))/(gamma_(CH_4)) = sqrt((16)/(4)) = 2 , (gamma_(He))/(gamma_(SO_2)) = sqrt((64)/4) = 4`. |
|
| 18. |
What is the pH of the resulting solution when equal volumes of 0.1 M NaOH and 0.01 M HCl are mixed ? |
|
Answer» `7.0` 0.1 X 1 - 0.01 x 1 = N x 2 `[OH^-]=N_R=0.09/2`=0.045 N pOH=-log (0.045) = 1.35 `therefore` pH=14-pOH=14-1.35 = 12.65 |
|
| 20. |
Whichof the following is least reactive towards electrophilic addition ? |
|
Answer»
 is least reactive towards ELECTROPHILIC ADDITION * Because `UNDERSET(NO_2)(C)H=CH_2` is electroedanating group equile is electrone group , So the electron density is less in this compound while. is least reactive towards ELECTROPHILIC ADDITION * Because `UNDERSET(NO_2)(C)H=CH_2` is electroedanating group equile is electrone group , So the electron density is less in this compound while.
|
|
| 21. |
Which of the following is not a characteristic of alcohol ? |
|
Answer» LOW m.p. |
|
| 22. |
Which among the following species have the same number of electrons in its outermost as well as penultimate shell |
|
Answer» `Mg^(2+)` `Ca^(2+)` =2,8,8 Hence, `Ca^(2+)` has 8 electrons each in outermost and PENULTIMATE SHELL. |
|
| 23. |
When electron make transition to gound state, the largest wavelenght (in nm) in then emitted radiattions is [Given :"E" repersents energy levelin ev] |
|
Answer» `(1240)/|(E_(6)-E_(5))|` |
|
| 24. |
Which of the sets of reagents below should be used toeffect the following transformation ? |
|
Answer» `CH_(3)CH_(2)Br,PPh_(3)//C_(4)H_(9)Li//THF//-78^(@)C.` 
|
|
| 25. |
What are the sources of water pollution? Give examples. |
|
Answer» Solution :(i) The water pollutants originate form both natural as well as human activities. The source of water pollution are classified as point and non-point sources. (II) Easily identified source of water pollution is called a point source. For example Municipal and industrial DISCHARGE pipes. (iii) Non-point source cannot be identificd easily. For example, agricultural runoffs, mining wastes, acid RAIN, STORM water drainage and CONSTRUCTION sediment |
|
| 26. |
Which one of the following statements is incorrect with regard to ortho and para dihydrogen? (1) They are nuclear spin isomers. (2) The ortho isomer has zero nucolear spin whereas the para isomer has one nuclear spin. (3) The para isomer is favoured at low temperatures. (4) The thermal conductivity of the para isomer is 50% greater than that of the ortho isomer. (5) It is never possible to obtain 100% pure ortho isomer. |
|
Answer» While in para hydrogen, Spins of protons are in opposite direction, so the nuclear spin = `=1/2+(-1/2)=0` |
|
| 27. |
Who studied the proportion of elements in CuCO_(3) ? |
|
Answer» JOSEPH proust |
|
| 28. |
ThespeciesAr, K^(+) and Ca^(2+) containthe samenumberof electronic. In whichorder totheirradiiincrease. |
|
Answer» `CA^(2+) , LT K^(+) lt Ar` |
|
| 29. |
Which salt gives crimson colour in flame |
|
Answer» `SrCI_(2)` |
|
| 30. |
Which of the following has standard Enthalpy value not equal to zero ? |
|
Answer» RHOMBIC Sulphur |
|
| 31. |
X-ray study of ice shows that each oxygen atom is surrounded tetrahedrally by four other hydrogen atom at a distance of __________. |
|
Answer» 256 pm |
|
| 32. |
You are provided with a solid 'A' and three solutions of A dissolved in water -one saturated one unsaturated ,and one super saturated. How would you determine which solution is which? |
|
Answer» Solution :When maximum amount of solute is dissolved in a SOLVENT, any more addition of solute will result in precipitation at a given temperature and pressure Such solution is called SATURATED solution ` (##SUR_CHE_XI_V02_C09_E01_039_S01.png" width="80%"> Saturated solution : A solution in which no more solute can be dissolved in a definite amount of solvent at given temperature is called a saturated solution Unsaturated solution : A solution which contains LESS amount of solute at a given temperature is called unsaturated solution . Super saturated solution : A solution which has more of solute than the saturated solution at a given temperature is called super saturated solution. |
|
| 33. |
What are the oxidation products of the following |
Answer» SOLUTION :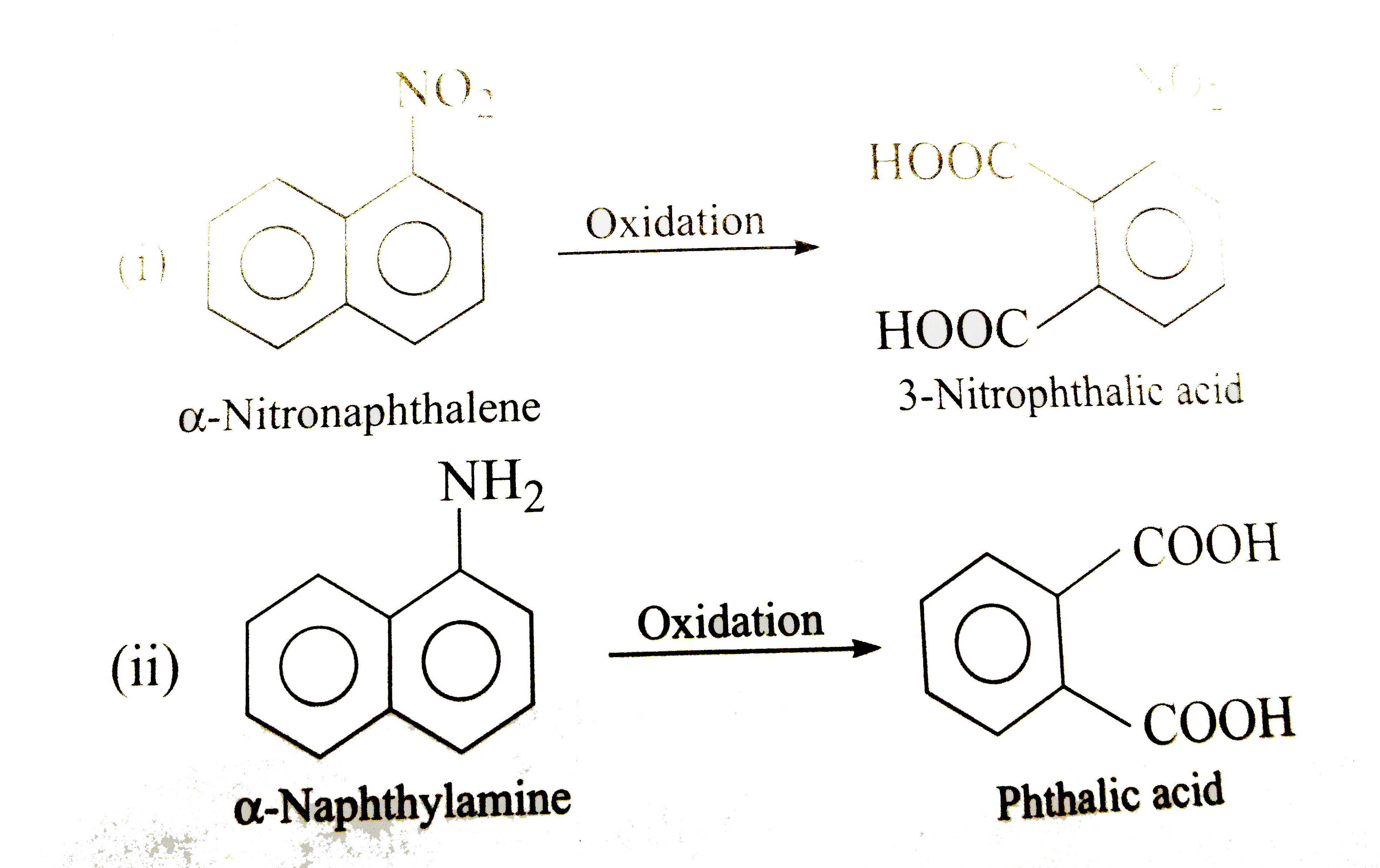
|
|
| 34. |
Which temperature and pressure will be to obtain more products in the following reaction ? Explain by Le-Chatelier principle. (i)N_(2(g)) + 3H_(2(g)) hArr 2NH_(3(g)) , DeltaH =-93.8 "kJ mol"^(-1) (ii)H_(2(g)) + CO_(2(g)) hArr CO_((g)) +H_2O_((g)) , DeltaH=+41 "kJ mol"^(-1) (iii)N_(2(g)) + O_(2(g)) hArr 2NO_((g)) , DeltaH=175 kJ (iv)CO_((g))+ 2H_(2(g)) hArr CH_3OH_((g)), DeltaH=-92.0 "kJ mol"^(-1) |
|
Answer» Solution :(i) High pressure and low temperature gives more PRODUCTS. (ii) And (iii) there is no effect of pressure, but high temperature give more products. (IV) High pressure and low temperature gives more products. |
|
| 35. |
Which of the following paris of compounds are diasteremeres? |
|
Answer»
|
|
| 36. |
Which of the followings are Lewis acids ? H_2O , BF_3 , H^+ and NH_4^+ |
| Answer» SOLUTION :LEWIS ACID : `H_2O , BF_3` and `H^+` | |
| 37. |
Which of the following shows ferrimagnetism, |
| Answer» Solution :`Fe_(3)O_(4)`is ferrimagnetic. | |
| 38. |
What is/are correct statement (s)? |
|
Answer» `CH_3cOONH_4` have greater degree of hydrolysis in 0.2M solution in comparison of 0.4 M solution (b)anions come fromacids which are more acidic than `H_2O` undergo hydrolysis. (c ) AMONG two acids , anioncomes fromweaker acid (less `K_a) ` undergo more hydrolysis. (d)` HSO_4^(-) `comes fromstronger acid `H_2SO_4` . so it does not undergo hydrolysis. |
|
| 39. |
Which statement (s)is/are wrong about the interconversion of trans - and cis- isomeric alkenes by ultraviiolet radiation with a suitable photosensitiser ? |
|
Answer» `U.V.` radiation provides energy to excit `pi bar(e)` into an anti`-` bonding `pi` molecular orbital `(pi ^(**) ABMO)` MOMENTARILY breaking the `pi-` bond. |
|
| 40. |
Which is more metallic among the elements of IIIA group? Why? |
| Answer» Solution :Aluminium has least ELECTRONEGATIVITY value and ALSO least reduction potantial. HENCE it is more ELECTROPOSITIVE and so is more METALLIC. | |
| 41. |
Which phrase would be incorrect to use |
|
Answer» A MOLECULE of a compound |
|
| 42. |
Which of the following statements is not true about H_(3)PO_(2) ? |
|
Answer» It is a TRIBASIC acid |
|
| 43. |
Write down the products of ozonolysis of 1, 2-dimethylbenzene (o-xylene). How does the result support Kekule structure for benzene ? |
Answer» SOLUTION :
|
|
| 44. |
Which factors the solubility of substance is depends on ? |
|
Answer» Solution :(i) Latticce enthalpy of SALT (II) HYDRATION enthalpy of IONS in solution |
|
| 45. |
Name the species that will be isoelectronic with Rb^+ |
| Answer» SOLUTION :`Rb^+ (36 ELEMENTS) Kr,BR,Sr^(2+)` | |
| 46. |
What is electro valency of ion ? Give example . |
|
Answer» Solution :Electro valency of ion means NUMBER of unit CHARGE on ion. Example : In NaCl one unit positive charge in `NA^(+) and " in " CaF_(2)` two unit positive charge in `Ca^(2+)` and its elector velency is +2. In `F^(-) ,Cl^(-)` one unit NEGATIVE charge and valency is - 1 . 
|
|
| 47. |
What is a Bond angle? Give example. |
|
Answer» Solution :It is defined as the ..angle between the oribitals containing bonding ELECTRON pairs AROUND the CENTRAL atom in a molecule or in a complex ion... It is expressed in degress. Example: In `CO_(2)` the bond angle is `180^(@)`. So `CO_(2)` has linear shape. |
|
| 49. |
Which among CH_(4),SiH_(4),GeH_(4) and SnH_(4) is the most volatile ? |
|
Answer» `CH_(4)` |
|
| 50. |
Which method is used to prepare metal sols? |
|
Answer» Crystallization |
|