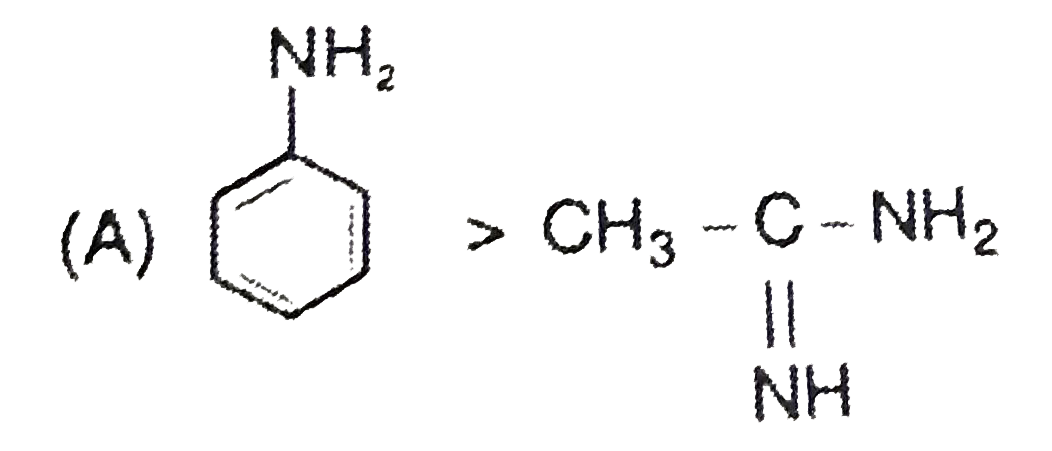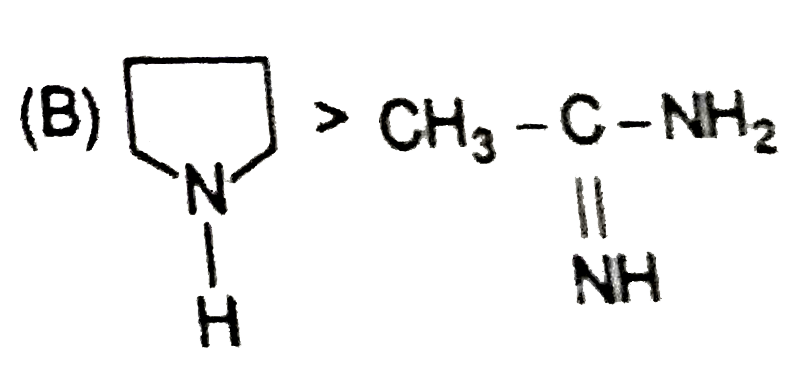Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
On a ship sailing in Indian ocean where the temperture is 26.1^@C, a ballon is filled with 20.18L of air. What will be the volume of the ballon when the ship reaches Pacific ocean, where temperature is 23.4^@C ? |
Answer» Solution :The CHARLES. law is given as,`(V_1)/(V_2) =(T_1)/(T_2)`  The volume of BALLOON at pacific ocean = ` V_2- (V_1 T_2 )/(T_(12)) = (20.18 xx 296 .4)/( 299.1 ) = 20 L` |
|
| 2. |
Oleum is mixture of H_(2)SO_(4) and SO_(3) i.e. H_(2)S_(2)O_(7) which is obtained by passing SO_(3) is solution of H_(2)SO_(4).In order to dissolve SO_(3) in oleum, dilution of oleum is done by water in which oleum is converted into pure H_(2)SO as shown below: H_(2)SO_(4)+SO_(3)+H_(2)Oto2H_(2)SO_(4) (pure) When 100 gm oleum is diluted with water then total mass of diluted oleum is known as percentage labelling in oleum. For example: 109% H_(2)SO_(4) labelling of oleum sample means that 109 gm pure H_(2)SO_(4) is obtained on diluting 100 gm oleum with 9 gm H_(2)O which dissolves al free SO_(3) in oleum. If the number of moles of free SO_(3), H_(2)SO_(4), and H_(2)O be x, y and z respectively in 118% H_(2)SO_(4) labelled oleum, the value of (x+y+z) is |
|
Answer» `2.2` 100G oleum + 18G water `underset("80 g")(SO_(3))+underset("18 g")(H_(2)O)rarrH_(2)SO_(4)` `18gH_(2)O` COMBINES with `80_(g)SO_(3)` So, weight of `SO_(3)` in 100g oleum = 80g `therefore` No. of moles of `SO_(3)=(80)/(80)=1` No. of moles of `H_(2)SO_(4)=(20)/(98)=0.2` No. of moles of `H_(2)O=(18)/(18)=1` Total no. of moles = 2.2 |
|
| 3. |
Oleum is mixture of H_(2)SO_(4) and SO_(3) i.e. H_(2)S_(2)O_(7) which is obtained by passing SO_(3) is solution of H_(2)SO_(4).In order to dissolve SO_(3) in oleum, dilution of oleum is done by water in which oleum is converted into pure H_(2)SO as shown below: H_(2)SO_(4)+SO_(3)+H_(2)Oto2H_(2)SO_(4) (pure) When 100 gm oleum is diluted with water then total mass of diluted oleum is known as percentage labelling in oleum. For example: 109% H_(2)SO_(4) labelling of oleum sample means that 109 gm pure H_(2)SO_(4) is obtained on diluting 100 gm oleum with 9 gm H_(2)O which dissolves al free SO_(3) in oleum. If 109% H_(2)SO_(4) labelled oleum, the percent of free SO_(3) and H_(2)SO_(4) are |
|
Answer» `30%,70%` 100g oleum + `9G H_(2)O` `80g-18gimplies40g-9g` `therefore 100g` oleum contain `40G SO_(3) and 60g H_(2)SO_(4)` `%SO_(3)=(40)/(100)xx100=40` `%H_(2)SO_(4)=(60)/(100)xx100=60` |
|
| 4. |
Oil of Bitter almonds is |
|
Answer» Nitrobenzene |
|
| 5. |
Oidentify the incrrect statement ? |
|
Answer» The INTERMEDIATE PRODUCTS obtained during eletrolysis of 50% `H_(2)SO_(4)` are `H_(2)S_(2)O_(8)` and `H_(2)SO_(5)` |
|
| 6. |
OHC-(CHOH)_(4)-CH_(2)OHhas a primary alcoholic group at |
|
Answer» `1^(st)` CARBON |
|
| 7. |
OH^- ions are in acidic solution and H_3O^+ions an in basic solution available is it true ? Explain. |
|
Answer» Solution :Right there is `OH^-` in ACID solution and `H_3O^+`in base solution ALWAYS present. because, In acid on `[OH^-]=K_w/([H_3O^+])` In base solution `[H_3O^+]=K_w/[[OH^-]]` |
|
| 8. |
OH group in methyl carbinol is neutral while it is acidic in acetic acid because : |
|
Answer» methyl carbinol molecules get associated  the `+I` effect of `C_(2)H_(5)` decreases the polarity, of OH group while `C = O` group of acetic increases the polarity of `-OH` group the `+I` effect of `C_(2)H_(5)` decreases the polarity, of OH group while `C = O` group of acetic increases the polarity of `-OH` group
|
|
| 9. |
Of the two molecules OCS and CS_(2) which one has higher dipole moment value ? Why ? |
Answer» Solution :OCSand `CS_(2)` :  The dipole moment `H_("OCS") = 0.7149 pm 0.0003 " Debye " CS_(2) : S = C = S` In `CS_(2)`the bond dipole of 2C = S have same values and bond dipoles cancel each ether so dipoles moment of `CS_(2)` is zero . Among OCS and `CS_(2)` OCS has a HIGHER DIPOLEMOMENT because in OCS oxygen is more ELECTRONEGATIVE that sulphur and C = S and C = O bond in OCS molecules do not cancel each other. On other hand `CS_(2)` due to linear STRUCTURE the bond dipole to two `C = S` bonds cancel each other. On the other hand `CS_(2)` due to linear structure the bond dipoles of two C = S bonds cancel each other and the result and dipole moment value is zero . So OCS has a higher dipole moment than `CS_(2)`. |
|
| 10. |
Of the two molecules OCS and CS_(2) which one has higher dipole moment value .Why? |
Answer» Solution :OCS and `CS_(2)`:  The DIPOLE moment `mu_(ocs) = 0. 7149 + 0. 0003` Debye. `Cs_(2):`S = C = S In `CS_(2),` the bond dipoles of 2 C = S have same values and bond dipoles cancel each other so dipole moment of `CS_(2)` is zero. Among OCS and `CS_(2),` OCS has a HIGHER dipole moment because in OCS OXYGEN is more electronegative than sulphur and C =S and C = O bonds in OCS molecules do not cancel each other. On the other HAND `CS_(2)` due to linear structure, the bond dipole of twoC = S bonds cancel each other. On the other hand, `CS_(2)` due to linear structure, the bond dipoles of two C =S bonds cancel each other and the RESULTANT dipole moment value is zero. So OCS has a higher dipole moment than `CS_(2)`. |
|
| 11. |
Of the two elements given in each of the following cases, choose the element having a more negative electron gain enthalpy. Explain. N andO |
| Answer» Solution :Elements with higher positive value of electron affinity have a more negative value of electron GAIN enthalpy. As NITROGEN has a half filled p-subshell so electron affinity of OXYGEN (O) SHALL be higher. | |
| 12. |
Of the isomeric hexanes , the isomers that give the minimum and number of monochloro derivatives are respectively |
|
Answer» 3-methylpentane and 2,3-dimethylbutane 2-Methylpentane has five different types of hydrogens and hence FORMS five MONOCHLORO derivatives. On the other hand , 2,3-dimethylbutane `underset"(1)"CH_3-underset(._((1))CH_3)underset|overset"(2)"CH-underset"(1)"underset(CH_3)underset|overset"(2)"CH-overset"(1)"CH_3`has TWO different types of hydrogens and hence forms two monochloro derivatives. |
|
| 13. |
Of the four oxyacids of chlorine the strongest oxidising agent in dilute aqueous solution is _____ |
|
Answer» `HClO_(4)` `OVERSET(+1)(HClO)ltoverset(+3)(HClO_(2))ltoverset(+5)(HClO_(3))ltoverset(+7)(HClO_(4))` |
|
| 14. |
Of the following, which change will shift the reaction towards the productI_(2)(g) hArr 2I(g) Delta H^(@) ""_(f) (298K) = + 150 kJ |
|
Answer» INCREASE in CONCENTRATION of I |
|
| 15. |
Of the following pairs, the one containing examples of metalloid elements in the periodic table is: |
|
Answer» NA and K |
|
| 16. |
Of the following molecules, which have shape similar to carbon dioxide ? |
|
Answer» `SnCl_(2)` |
|
| 17. |
Of the following molecules, which have shape similar to carbon dioxide? |
|
Answer» `SnCl_(2)` `C_(2)H_(2)` - Linear |
|
| 18. |
Of the following compounds, which has the highest boiling point? |
|
Answer» n-Butyl CHLORIDE |
|
| 19. |
Of the following compounds, the most acidic is |
|
Answer» `As_(2)O_(3)` |
|
| 20. |
Of the following acids, the one which has the capability to form complex compound and also possesses oxidising and reducing properties is |
| Answer» Solution :`HNO_(2)` acts as reducing oxidising and COMPLEXING AGENT. | |
| 21. |
Of the five isomeric hexanes , the isomer which can give two monochlorinated compounds is |
|
Answer» n-hexane |
|
| 22. |
Of the five isomeric hexanes, the isomer which can give two mono chlorinated compounds is |
|
Answer» n-Hexane `underset("2, 3-dimethylbutane")(H_(3)C-underset(H)underset(|)OVERSET(CH_(3))overset(|)C-underset(H)underset(|)overset(CH_(3))overset(|)C-CH_(3))` 2, 3-dimethylbutane have TWO types of hydrogen atoms. So, on monochlorination gives only two monochlorinated compounds. `H_(3)C-overset(CH_(3))overset(|)underset(H)underset(|)C-overset(CH_(3))overset(|)underset(H)underset(|)C-CH_(3)overset(Cl_(2))toH_(3)C-underset(H)underset(|)overset(CH_(3))overset(|)C-underset(H)underset(|)overset(CH_(3))overset(|)C-CH_(3)` `H_(3)C-overset(CH_(3))overset(|)underset(Cl)underset(|)C-underset(H)underset(|)overset(CH_(3))overset(|)C-CH_(3)` |
|
| 23. |
Of all the Group 13 elements, the element with lowest electronegatively is |
|
Answer» B |
|
| 24. |
ODS are used in ........ |
|
Answer» REFRIGERATOR |
|
| 25. |
Odd one out the following among elements in the triad. |
| Answer» Answer :a | |
| 26. |
Odd electron bond is not present in |
|
Answer» `NO` |
|
| 27. |
Octet rule mostly violated in the compounds formed by |
|
Answer» ALKALI metals |
|
| 28. |
Octet rule is mostly violated in the compounds formed by |
|
Answer» ALKALI METALS |
|
| 29. |
What is producer gas ? |
| Answer» | |
| 30. |
Octet rule is followed for the centrali atom in the formation of |
|
Answer» `SF_(6)` |
|
| 31. |
Octet rule in not followed in |
|
Answer» `SF_(6)` |
|
| 32. |
Octane number is related to |
|
Answer» Gasoline |
|
| 33. |
Octahedral shape is due to the hybridisation |
|
Answer» `SP^(3)d` |
|
| 34. |
Octahedral molecule among the following is |
|
Answer» `SO_(3)` |
|
| 35. |
____________occurs in large amounts in sea water. |
|
Answer» NaCl |
|
| 36. |
Obtain the relation between equilibrium constant K and K' forward and reverse reaction. |
|
Answer» Solution :We write reaction of HI synthesis. Its equilibrium is as under. `H_(2(g)) + I_(2(g)) hArr 2HI_((g))`…(Eq. -i) For this reaction as PER law of chemical equilibrium `K_c` is as follow. `K_c="[HI]"^2/([H_2][I_2])=X` ….(Eq.-ii) At same temperature the reverse reaction-(i) occurs the equilibrium of decomposition of HI reaction is as under. `2HI_((g)) hArr H_(2(g)) +I_(2(g))`.....(Eq.-iii) The equilibrium constant `K._c` at same temp. is , `K._c=([H_2][I_2])/([HI]^2)=1/X`....(Eq.-iv) So, `(K_c)(K._c)=([HI]^2)/([H_2][I_2])xx([H_2][I_2])/([HI]^2)=1` and `K_c=1/(K._c)` ....(RESULT -1) "Equilibrium constant for the reverse reaction is the inverse of the equilibrium constant for the reaction in the forward direction." |
|
| 37. |
Observed the following reaction sequence and choose the correct options. |
|
Answer» (A) and (B) are diastereomer's of each other. 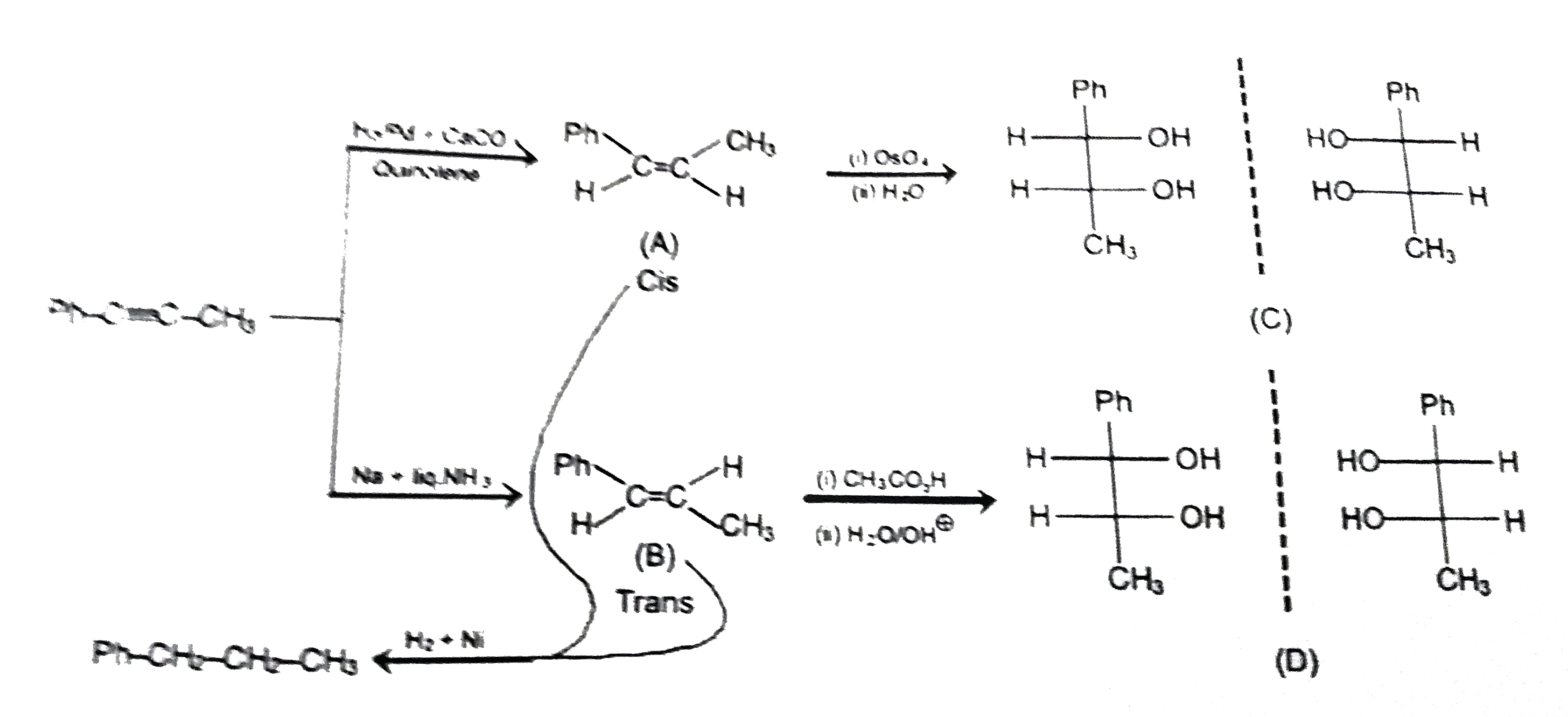
|
|
| 38. |
Observe these compound and give answer of following questions. Q. A carbohydrate undergoes the following conversion "D-Lyxose"underset(HCl)overset(NaCN)to'A'overset(H_(2)//Pd-BaSO_(4))to'B'overset(H_(3)O^(o+))to'C' 'C' can be |
|
Answer» D-Galatose |
|
| 39. |
Observe these compound and give answer of following questions A carbohydrate undergoes the following conversion D-arabinose underset(HCl)overset(NaCN)to'A'overset(H_(2)//Pd-BaSO_(4))to'B'overset(H_(3)O^(o+))'C' 'C' can be |
|
Answer» D-Glucose 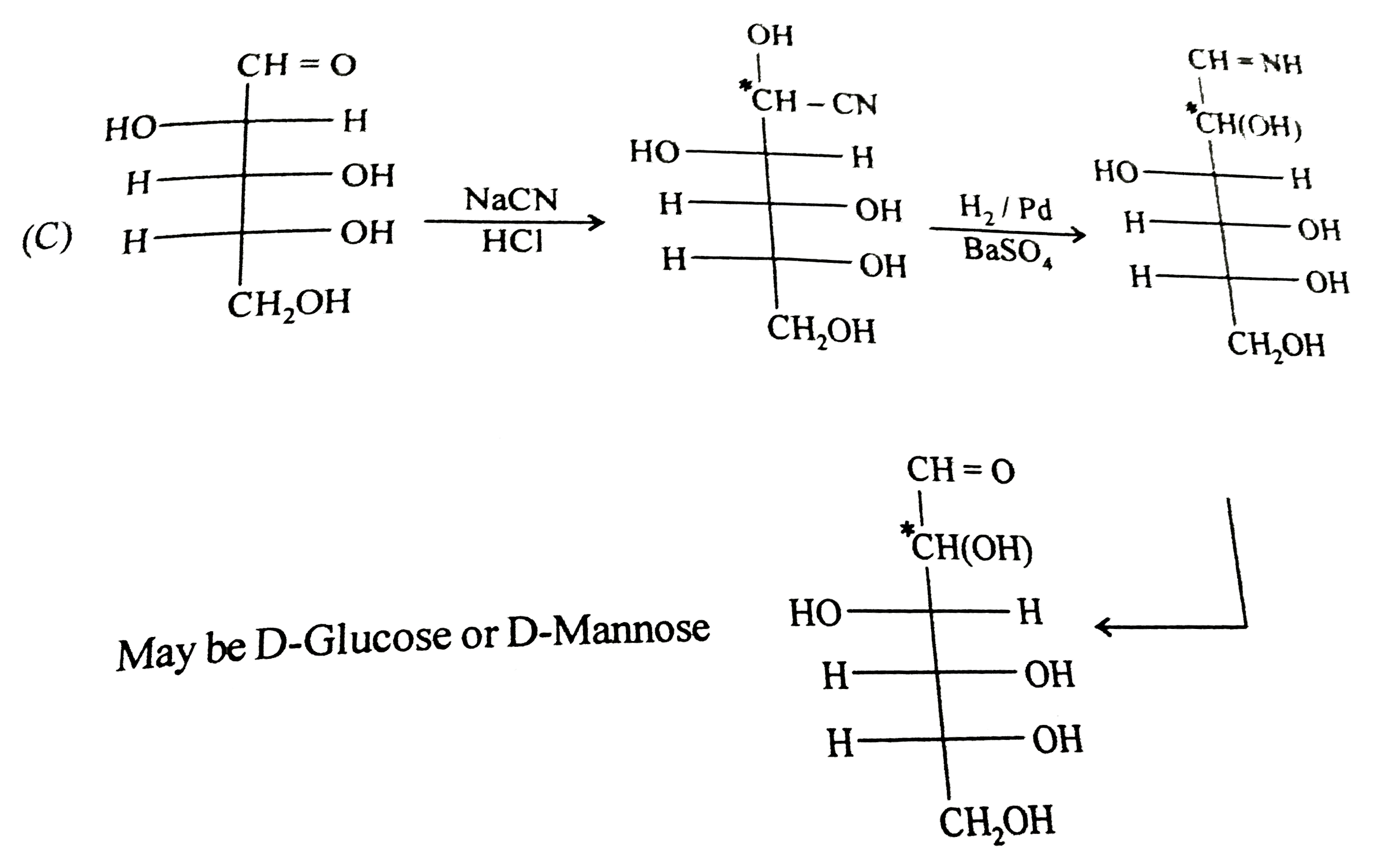
|
|
| 40. |
Observe the following two reaction of benzene. A) C_6H_6 + CH_3Cl underset(K_1)overset(AlCl_3)to C_6H_5CH_3 B) C_6H_5D + CH_3Cl underset(K_2)overset(AlCl_3)to C_6H_5CH_3 The relation between rate constant of the two reactions will be. |
|
Answer» `K_1 GT K_2` |
|
| 41. |
Observe the following statements regarding purification of bauxite I. During Hall's process, silicon is removed as Si(vapour) II. Bauxite ore contaminated with Fe_(2)O_(3) is purified in Baeyer's process III. During Serpeck's process AIN is formed. The correct answer is |
|
Answer» I, II and III are CORRECT |
|
| 42. |
Observe the following statements.Q I. The physical and chemical properties of elements are periodic functions of their electronic confirguration.Q II. Electronegativity of fluorine is less than the electronegativity of chlorine.Q III. Electropositive nature decreases from top to bottom in a group. The correct answer is |
|
Answer» I, II and III are CORRECT |
|
| 43. |
Observe the following statement : (I) Heavy water is harmful for the growth of animals (II) Heavy water reacts with Al_(4)C_(3) and forms deuterated acetylene (III)BaCl_(2).2D_(2)O is an example of interstitial deuterate |
|
Answer» `1 & 3 ` |
|
| 44. |
Observe the following sequence of reactions : Select the correct option regarding the relative basic strength (K_(b)): |
|
Answer»
|
|
| 45. |
Observe the following reaction sequence carefully and answer the question that follows. "Boric acid"overset("strong")underset("heat")rarrXoverset(C//Cl_(2))underset(Delta)rarrYoverset(LiAlH_(4))rarrAoverset("excess"NH_(3))underset("low temp")rarrBoverset(200^(@)C)underset(Delta)rarrC overset(NH_(4)Cl)underset("heat")rarrDoverset(NaBH_(4))rarr E overset(3HCl)rarr Z Pick out the incorrect statement from the following with respectto the above paragraph ? |
|
Answer» Conversion of E to Z is an example of addition reaction |
|
| 46. |
Observe the following reaction: [Ne(en)_(3)](NO_(3))_(2)+S_(2)O_(3)^(2-)(aq) overset("Slightly")underset("alk.medium")rarr'Y' (darr) violet ppt. Among the following, how many compound decompose the nickel ethylenediamine nitrate reagent with the precipitation of NiS, in the above test. Na_(2)SO_(3),Na_(2)SO_(4),Na_(2)S_(2)O_(3),Na_(2)S_(4)O_(6),H_(2)S, (NH_(4))_(2)S,NaSCN |
|
Answer» |
|
| 47. |
Observe the following reaction sequence carefully and answer the question that follows. "Boric acid"overset("strong")underset("heat")rarrXoverset(C//Cl_(2))underset(Delta)rarrYoverset(LiAlH_(4))rarrAoverset("excess"NH_(3))underset("low temp")rarrBoverset(200^(@)C)underset(Delta)rarrC overset(NH_(4)Cl)underset("heat")rarrDoverset(NaBH_(4))rarr E overset(3HCl)rarr Z Identify the incorrect statement form the followings about Boric acid |
|
Answer» Boric ACID crustallizes in a layer structure in which `H_(3)BO_(3)` UNITS are BONDED togethr by Vander Waals forces. |
|
| 48. |
Observe the following reaction sequence carefully and answer the question that follows. "Boric acid"overset("strong")underset("heat")rarrXoverset(C//Cl_(2))underset(Delta)rarrYoverset(LiAlH_(4))rarrAoverset("excess"NH_(3))underset("low temp")rarrBoverset(200^(@)C)underset(Delta)rarrC overset(NH_(4)Cl)underset("heat")rarrDoverset(NaBH_(4))rarr E overset(3HCl)rarr Z Which of the following statement with respect to the paragraph is incorrect? |
|
Answer» `"X"` on REACTION with metal oxide, GIVES metal metaborates |
|
| 49. |
Observe the following reaction sequence Calculate molecular mass [W] of product I and report your answer as N, where N=W-:3. |
| Answer» | |
| 50. |
Observe thefollowing reaction : Ar-C-Hoverset(OH^(-))hArr[X]overset(Ar-overset(O)overset(||)C-H)hArr[Y]+[Z] overset(H^(+)hArrY^(1)+Z^(1)In step -3 if Y transfers H^(-) ion to Z, then Y' andZ' are respectively : |
|
Answer» `PhCOO^(-)+PhCH_(2)OH` |
|