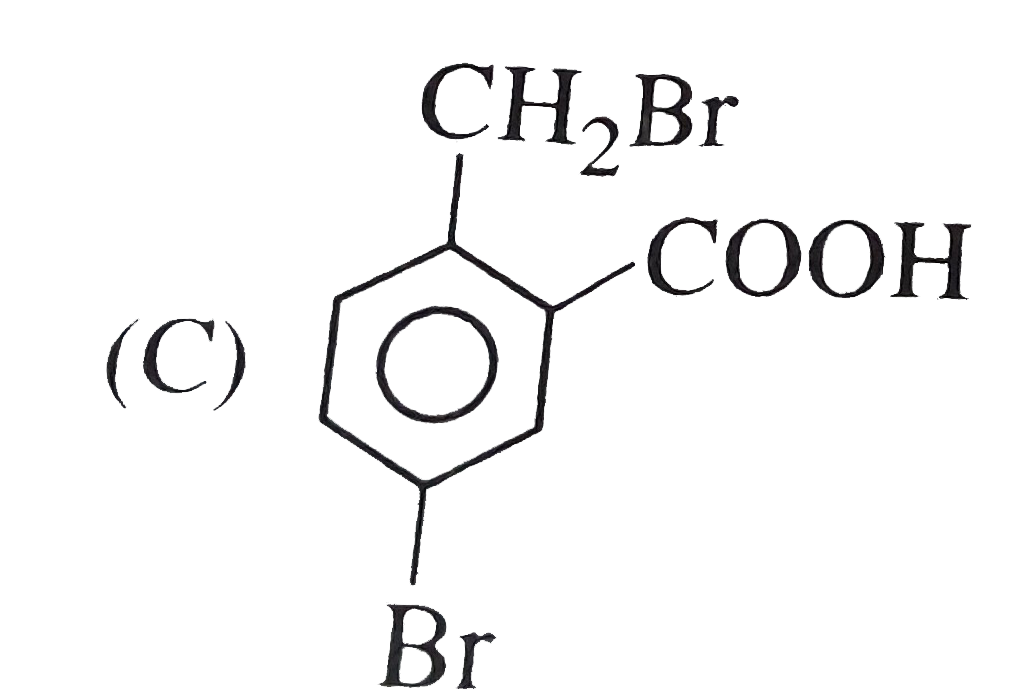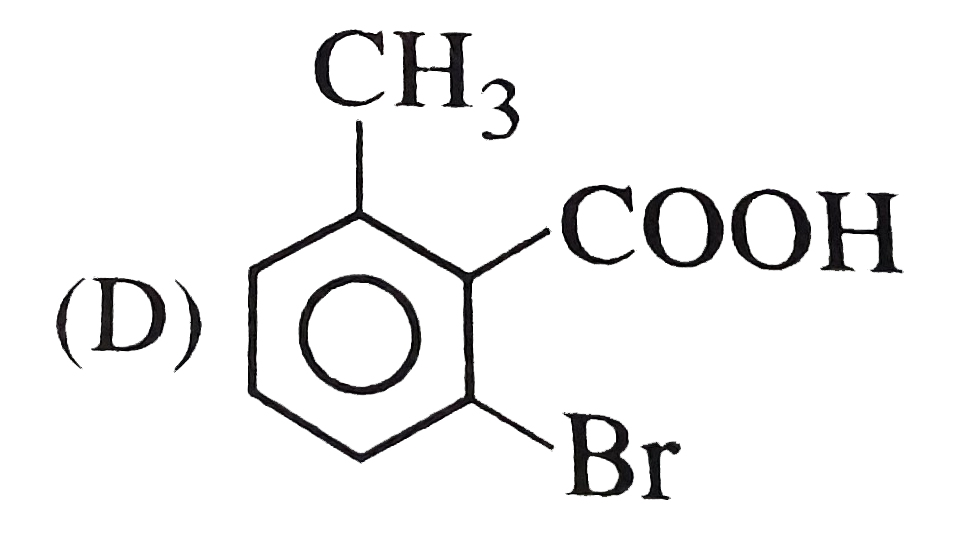Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Observe the following reaction :Ar-C-Hoverset(OH^(-))hArr[X]overset(Ar-overset(O)overset(||)C-H)hArr[X]+[Z] overset(H^(+)"Transfer")hArrY^(1)+Z^(1)The slowest step of the reaction is : |
|
Answer» 1 |
|
| 2. |
Observe the following reaction :Ar-C-Hoverset(OH^(-))hArr[X]overset(Ar-overset(O)overset(||)C-H)hArr[X]+[Z] overset(H^(+)"Transfer")hArrY^(1)+Z^(1)Rate of step -2 will be fastest with the combination of : |
|
Answer»
|
|
| 3. |
Observe the following reaction and answer the following questions : The structure of Q_(1) is : |
|
Answer» `Ph-UNDERSET(O^(Ө))underset(|)C=CH-underset(O)underset(||)C-H` |
|
| 4. |
Observe the following reaction and answer the following questions : The product 'R' is : |
|
Answer» `Ph-UNDERSET(OH)underset(|)C=C=underset(OH)underset(|)C-H` |
|
| 5. |
Observe the following graph for the formation of HI from H_(2) & I_(2) as H_(2)+I_(2) harr 2HI the above graph indicates what? |
|
Answer» I = old equilibrium II is `r_(F) gt r_(b)` I & III are equation |
|
| 6. |
Observe the following experiment: Which of the following reaction is/are correctly matched about set-I and set-II? |
|
Answer» `Na_(2)S_(2)O_(3)+2HCI rarr S (darr) +H_(2)SO_(3)+2NaCI` |
|
| 7. |
Observe the following experiment. The correct statement about set up (I) and (II) are: |
|
Answer» In SET up (I), initially no precipitate is formed but on adding of excess of reagent and followed by boiling gives greyish black PPT. |
|
| 8. |
Observe the following experiment: The incorrect statement about set-II is/are: |
|
Answer» The acidified liquid soon becomes TURBID owing to formation of sulphur and `H_(2)SO_(4)` |
|
| 9. |
Observe the following compound and select +ve & -ve test respectively |
|
Answer» `+ + + -` |
|
| 10. |
Observe the following compounds |
|
Answer» |
|
| 11. |
Observe the following compound and match the reagents of List-I and List-II |
|
Answer» <P> |
|
| 12. |
Observe the followig reaction and determine True statement |
|
Answer» If aromtic RING I have `-NO_(2)` GROUP then rate of REACTION will decrease. |
|
| 13. |
(O.A=Oxidising agent, R.A= Reducing agent, M= Molecular weight) |
|
Answer» |
|
| 14. |
Objective question (single correct answer). i. The molarity of a aqueous solution of glucose (C_(6) H_(12) O_(6)) is 0.01 To 200 mL of the solution, which of the following should be carried out to make it 0.02 M? I. Evaporate 50 mL of solution III. Add 0.180 g of glucose and then evaporate 50 mL of solution III. Add 50 mL of water The correct option is: a. Ib. IIIc. IId. I, II, III ii. The atomic mass of Cu is 63.546. There are only two naturally occuring isotopes of copper Cu^(63) and Cu^(65). The percentage of natural abundance of Cu^(63) in nearly a. 30b. 10c. 50d. 73 iii. An aqueous solution of urea (NH_(2) COHN_(2)) is 3.0 molal. The mole fraction of urea is a. 0.33b. 0.25c. 0.66d. 0.05 iv 0.2 M H_(2) SO_(4) (1 mL) is diluted to 1000 times of its initial volume. the final normality of H_(2) SO_(4) is: a. 2 xx 10^(-3)b. 2 xx 10^(-4)c. 4 xx 10^(-4)d. 2 xx 10^(-2) v. Which of the following question are dependant on temperature? a. Molarlityb. Normalityc. Mole fractiond. Molality vi. A sample of H_(2) SO_(4) density 1.85 mL^(-1) is 90% by weight. What is the volume of the acid that has to be used to make 1 L f 0.2 M H_(2) SO_(4)? a. 16 mLb. 18 mLc. 12 mLd. 10 mL vii. The hydrated salt Na_(2)SO_(4). nH_(2)O undergoes 55.9% loss in weight on heating and becomes anhydrous. The value of n will be a. 5b. 7c. 3d. 10 viii. 0.2 mol of HCl and 0.1 mol of barium chloride is dissolved in water to produce a 500 mL solution. The molarity of Cl^(ɵ) is. a. 0.06 Mb. 0.12 Mc. 0.09 Md. 0.80 M ix. The density of 1 M solution of NaCl is 1.055 g mL^(-1). The molality of the solutions is. a. 1.0585b. 1.00c. 0.0585d. 0.10 x. Hydrochloric acid solution A and B have concentration of 0.5 N, and 0.1 N, respectively. The volume of solutions A and B required to make 2 L of 0.2 N hydrochloric acid are a. 0.5 L of A + 1.5 of B b. 1.0 L of A + 1.0 L of B c. 0.75 L of A + 1.25 L of B d. 1.5 L of A + 0.5 L of B |
|
Answer» So, the only possible answers is (c ). I. Evaporate `50 mL` of solution mmoles of glucose initially `= 0.01 xx 200 = 2` volume after evaporation `= 200 - 50` `= 150 mL` `M_("glucose") = (("mmoles")/(V_(mL))) = (2)/(150) = 0.013 M` II. mmoles of glucose added `= (0.180)/(180) xx 100 = 1` Total mmoles of glucose `= 2 + 1 = 3` Volumes `= 150 mL` (after evaporation of `50 mL` solution) `M_("glucose") = (3)/(150) = 0.02 M` III. Add `50 mL` of water New volume of solution `= 200 + 50 = 250 mL` `M_("glucose") = (2 mmol)/(250 mL) = 0.008 M` Hence, answer is (c ) ii. d. `63.546 = (a xx 63 + (100 - a) xx 65)/(100)` `a = 72.7% ~~ 73.%` iii. d. `m = (X_(2) xx 1000)/((1 - x_(2)) xx Mw_(1))` `3 = (X_(2) xx 1000)/((1 - x_(2)) xx 18)` Solve for `x_(2) = 0.05` iv. c. `M_(1) V_(1) = M_(2) V_(2)` `0.2 M xx 1 mL = M_(2) xx 1000 mL` `M_(2) = 2 xx 10^(-4)` `:. N = 2 xx 2 xx 10^(-4) = 4 xx 10^(-4)` vi. c. `M_(1) V_(1) = M_(1) V_(2) (M = (% "by weight" xx 10 xx d)/(Mw_(2)))` `V_(1) xx (90 xx 10 xx 1.8)/(98) = 0.2 xx 1 L` `V_(1) = 0.012 L = 12 mL` vii. d. Loss in weight is DUE to `nH_(2) O`. `:. (142 + 18 n) g of Na_(2) SO_(4). nH_(2) O = 18 n g` of loss in weight of `H_(2) O` `100 g of Na_(2) SO_(4) . nH_(2) O = (18 n xx 100)/(142 + 18 n)` `:. (18 n xx 100)/(142 + 18n) = 55.9` solve for `n implies n = 9.99 ~~ 10` viii. d. `0.02 "MOL" HCl = 0.02 of H^(o+) + 0.2 "mol" Cl^(ɵ)` `0.01 "mol" BaCl_(2) = 0.1 "mol" of Ba^(2+) + 0.1 xx 2 "mol" of Cl^(ɵ)` Total `Cl^(ɵ) = 0.4 "mol"`. Total volume `= 500 mL = (1)/(2) L` `:. [Cl^(ɵ)] = (0.4)/(1//2) = 0.8 M` ix. b. `d_(sol) = M ((Mw_(2))/(1000) + (1)/(m))` `1.0585 = 1 M ((58.5)/(1000) + (1)/(m))` solve for `m` `m = 1.0` x. a. `V_(1) + V_(2) = 2 L` `N_(1) V_(1) + N_(2) V_(2) = N_(3) V_(3)` `0.5 xx V_(1) + 0.1 xx V_(2) = 0.2 xx 2` `0.5 V_(2) + 0.1 V_(2) = 0.4` Solve equations (i) and (ii) `V_(1) = 0.5 L, V_(2) = 1.5 L` |
|
| 15. |
O_2^(2-)is the conjugate base of |
|
Answer» `OH^(-) ` |
|
| 17. |
O_(2)^(+),NO^(+) are present in |
| Answer» Solution :thermosphere | |
| 18. |
O_(2) molecule has ________ bonds . |
| Answer» Answer :a | |
| 19. |
O_(2-) ions are present in : |
|
Answer» BCC ARRANGEMENT |
|
| 20. |
O^(2-) and Si^(4+)are isoelectronic ions. If the ionic radius of O^(2-) is 1.4 oversetoA, the ionic radius of Si^(4+) will be |
|
Answer» `1.4 A^(@)` |
|
| 21. |
O_2 and He are taken in equal weights in a vessel. The pressure exerted by Helium in the mixture is |
|
Answer» `O_3` |
|
| 22. |
o-Xylene on reductive ozonolysis give ______ , _________ and ______ |
|
Answer» |
|
| 23. |
o-xylene, m-xylene and p-xylene are isomers of each other. What type of isomerism they possess ? |
|
Answer» FUNCTIONAL GROUP isomerism |
|
| 24. |
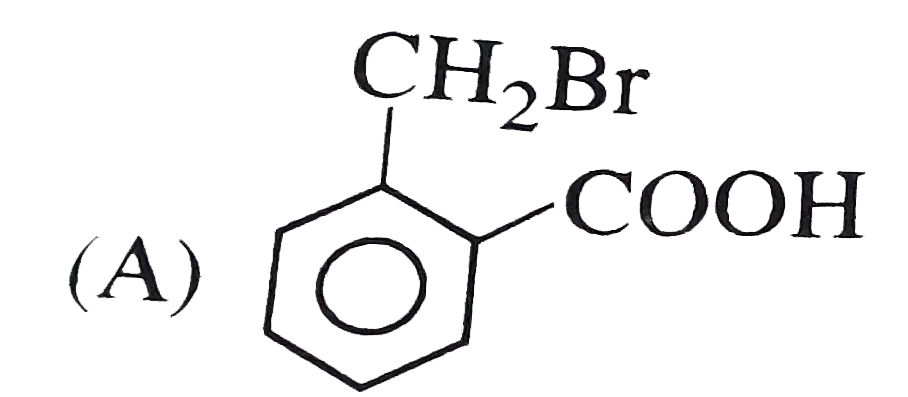
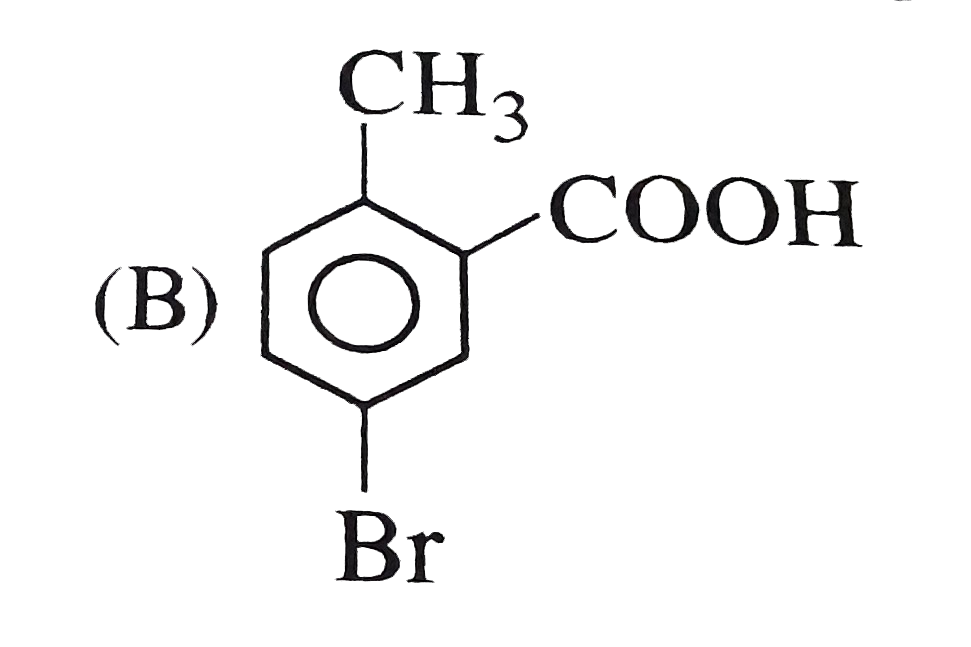
o-toluic acid on reaction with Br_(2) +Fe gives |
|
Answer»
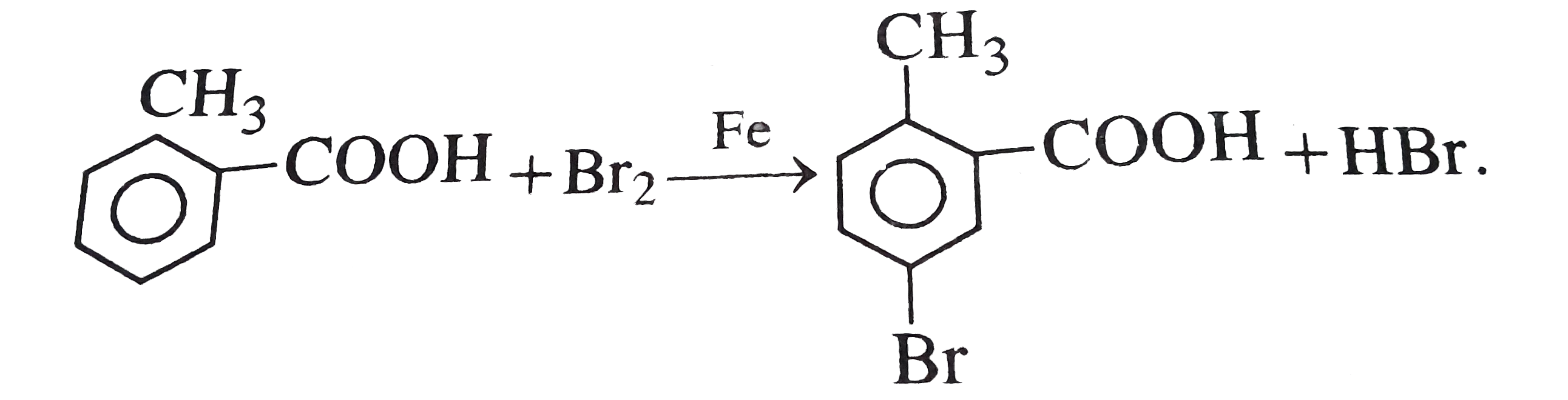
|
|
| 25. |
o-nitrophenol (x) and p-nitrophenol (y) are two compounds. The melting point relationship between the two is |
|
Answer» `X GT y` |
|
| 26. |
O-Nitrophenol is more volatile than p-Nitrophenot (Or) boling point of p-aitriphenol is greater than O-nitrophenol) |
| Answer» Solution :In O-nitrophenol there exists intra MOLECULAR HYDROGEN BONDING WITHIN the molecule where as in-nitrophenol there exists inter molecular hydrogen bonding between the molecule. | |
| 27. |
O + 2e^(-) to O^(-) , DeltaH = 639 kJ/mole O + e^(-)to O^(-) DeltaH o= -141 kJ/mole O^(-) + e^(-) to O^(-) , DeltaH = xkJ/mole What is the value of x ? |
|
Answer» `– 780 ` |
|
| 28. |
o- and p-directing substituents are ..... substituents, whereas m-directing substituents are ......... substituents. |
| Answer» SOLUTION :ACTIVATING, DEACTIVATING | |
| 29. |
How does an o- and p-directing group activate the ring? Illustrate with an example. |
|
Answer» |
|
| 30. |
Numerous forms of the periodic table have been devised from time to time. A modern version which is most convenient and widely used is the long or extended from of periodic table. The aufbau principle and the electronic configuration of atoms provide a theoretical foundation for the periodic classification. The horizontal rows are called periods. There are altogether seven periods. The first period consists of 2 elements. The subsequent periods consists of 8, 8,18, 18 and 32 elements respectively. The seventh period is incomplete and like the sixth period would have maximum of 32 elements. Elements having similar outer electronic configurations in their atoms are grouped in vertical columns. These are referred to as groups or families. According to the recommendations of IUPAC, the groups are numbered 1 to 18 replacing the older notation of groups 0, IA, IIA, ....VIIA, VIII, IB.....VIIB. Each successive period in the periodic table is associated with the filling up next higher principal energy level following aufbau sequence. The number of elements in each period is twice the number of atomic orbitals available in the energy level that is being filled. All the elements are classified into four blocks, i.e., s-block, p-block, d-block, and f-block depending on the type of atomic orbitals that are being filled with electrons. Elements A, B, C, D and E have the following electronic configuration: (A)1s^2, 2s^2 2p^1 (B)1s^2, 2s^2 2p^6 , 3s^2 3p^1 (C ) 1s^2, 2s^2 2p^6 , 3s^2 3p^3 (D)1s^2 , 2s^2 2p^6 , 3s^2 3p^5 (E)1s^2 , 2s^2 2p^6 , 3s^2 3p^6 Which among these will belong to same group in the periodic table ? |
|
Answer» A and C |
|
| 31. |
Numerous forms of the periodic table have been devised from time to time. A modern version which is most convenient and widely used is the long or extended from of periodic table. The aufbau principle and the electronic configuration of atoms provide a theoretical foundation for the periodic classification. The horizontal rows are called periods. There are altogether seven periods. The first period consists of 2 elements. The subsequent periods consists of 8, 8,18, 18 and 32 elements respectively. The seventh period is incomplete and like the sixth period would have maximum of 32 elements. Elements having similar outer electronic configurations in their atoms are grouped in vertical columns. These are referred to as groups or families. According to the recommendations of IUPAC, the groups are numbered 1 to 18 replacing the older notation of groups 0, IA, IIA, ....VIIA, VIII, IB.....VIIB. Each successive period in the periodic table is associated with the filling up next higher principal energy level following aufbau sequence. The number of elements in each period is twice the number of atomic orbitals available in the energy level that is being filled. All the elements are classified into four blocks, i.e., s-block, p-block, d-block, and f-block depending on the type of atomic orbitals that are being filled with electrons. What is the position of the element in the periodic table satisfying the electronic configuration (n-1)d^1 ns^2 for n=4 ? |
|
Answer» 3RD PERIOD and 3rd GROUP |
|
| 32. |
Numerous forms of the periodic table have been devised from time to time. A modern version which is most convenient and widely used is the long or extended from of periodic table. The aufbau principle and the electronic configuration of atoms provide a theoretical foundation for the periodic classification. The horizontal rows are called periods. There are altogether seven periods. The first period consists of 2 elements. The subsequent periods consists of 8, 8,18, 18 and 32 elements respectively. The seventh period is incomplete and like the sixth period would have maximum of 32 elements. Elements having similar outer electronic configurations in their atoms are grouped in vertical columns. These are referred to as groups or families. According to the recommendations of IUPAC, the groups are numbered 1 to 18 replacing the older notation of groups 0, IA, IIA, ....VIIA, VIII, IB.....VIIB. Each successive period in the periodic table is associated with the filling up next higher principal energy level following aufbau sequence. The number of elements in each period is twice the number of atomic orbitals available in the energy level that is being filled. All the elements are classified into four blocks, i.e., s-block, p-block, d-block, and f-block depending on the type of atomic orbitals that are being filled with electrons. An element belongs to group 17 with atomic number is 17. What is the atomic number of the element belonging to same group and present in fifth period? |
|
Answer» 25 |
|
| 33. |
The solubility product of PbS at 25^@ C is 9 xx 10^(-28). What is its solubility in pure water and in 2 xx 10^(-12) M Na_2S solution ? |
| Answer» SOLUTION :`1.4 XX 10^(-9)M ` | |
| 34. |
Numbers of chain isomers shown by Nonane are |
|
Answer» 5 |
|
| 35. |
Number of years it would take to spend Avogadro.s number of rupee at the rate of 10 lakhs rupee per second is x xx10^(10). Find the value of x. |
|
Answer» `6XX10^(23)rarr?` `=(6xx10^(23))/(10^(16))sec=2xx10^(10)` years |
|
| 36. |
Number of water molecules present in the hydrated state of MgSO_(4) |
|
Answer» |
|
| 37. |
Number of water molecule in 2 mole of plaster of pairs |
|
Answer» `CaSO_(4),(1)/(2)H_(2)O(or)2CaSO_(4).H_(2)O` So in two moles of plaster of paris one mole of `II_(2) O is PRESENT |
|
| 38. |
Number of valence electrons in carbon is |
|
Answer» 3 |
|
| 39. |
Number of unpaired electron(s) present in an element having atom ic num ber Z = 24 is ......... |
|
Answer» 6 `Cr(Z=24) [AR]^(18)3d^(5)4S^(1)`  thus it has 6 unparied ELECTRONS |
|
| 40. |
Number of unpaired electrons in Gd(Z =64) and the net electrons spin are |
|
Answer» 7.3,5 |
|
| 41. |
Number of unpaired electrons in 1s^2 2s^2 2p^3 is |
|
Answer» 2 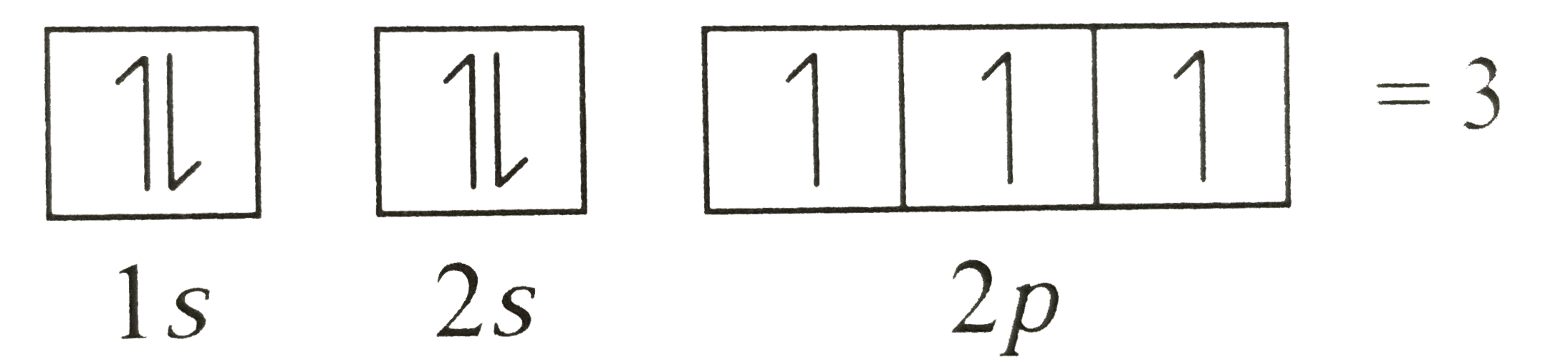
|
|
| 43. |
Number of total electrons in an orbit of - angular M omentum Quantum Number (I) is ........ |
|
Answer» `l^(2)` |
|
| 44. |
Number of the following elements which are block elements Pb, Al, B,K,S,Cd, Zn,Th,Sr |
|
Answer» |
|
| 45. |
Number of tetrahydral boran atoms in peroxoborate. |
Answer» 
|
|
| 46. |
The number of structurally isomeric primary amines for C_(4)H_(11)N |
|
Answer» 2 |
|
| 47. |
Number of t 3c - 2e bonds (hydrogen bridges) in Be(BeH_(4))_(2) is |
Answer» 
|
|
| 48. |
Number of structural isomers with the formula C_(4)H_(11)N |
|
Answer» 2 `C-C-UNDERSET( C )underset(|)N-C""C-C-underset(NH_(2))underset(|)C-C` `C-C-NH-C""C-underset(NH_(2))underset(|)OVERSET( C )overset(|)C-C` `C-underset( C )underset(|)C-C-NH_(2)""C-underset( C )underset(|)C-C-NHC` |
|
| 49. |
Number of structural isomers which can be obtained theoretically on monochlorination of 2-methylbutane is |
|
Answer» 1 
|
|
| 50. |
Number of stereoisomers possible for BrCH_2 CH Cl CH_2 CH Cl CH_3Br |
| Answer» Solution :Four | |