Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Number of sp^(2)-sp sigma bonds in the given compound is |
|
Answer» 1 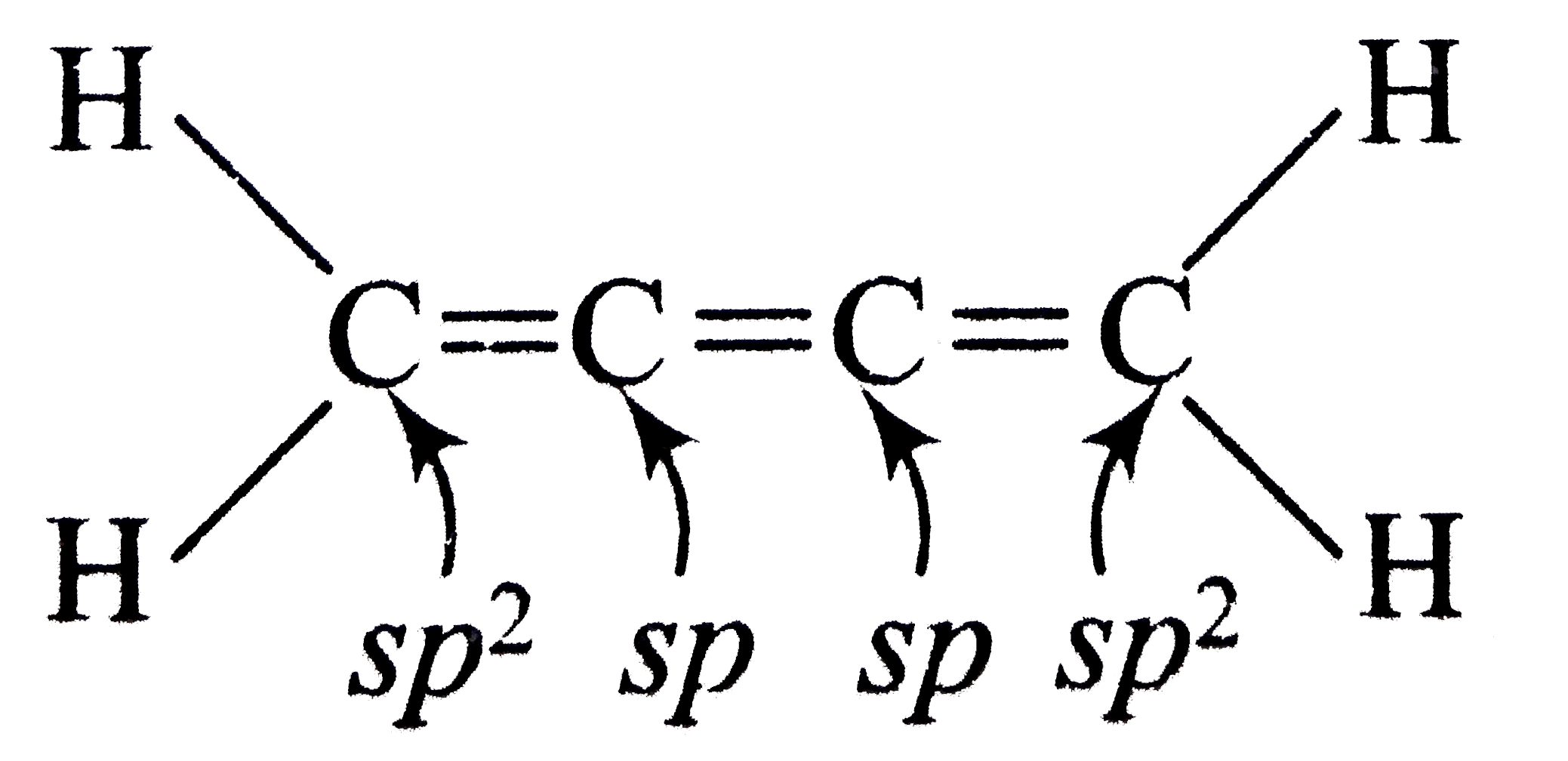
|
|
| 2. |
The no. of spherical nodes in a 4s orbital is |
|
Answer» Zero |
|
| 3. |
Number of six membered cyclic conjugated dieņes possible for C_(6)H_(8) is |
Answer» 
|
|
| 4. |
Number of significant figures in 78.000 g, 0.0206 g and 3.002 g respectively are |
|
Answer» 3,4 and 5 |
|
| 5. |
Number of significant figures in 5 are? |
|
Answer» 1 |
|
| 6. |
Number of significant figures in 5.00? |
|
Answer» 2 |
|
| 7. |
Number of sigma sp^2 - sp^(2) bonds present in a molecule of X in the process C_6H_6 overset(H_2 //"Ni") rarrX is |
|
Answer» 6 |
|
| 8. |
Number of sigma bonds in ethane formed by the overlap of sp^(3) and s orbital is |
|
Answer» 5 |
|
| 9. |
Number of short periods in the long form of periodic table |
|
Answer» 3 |
|
| 12. |
The number of radial nodes for a 3p orbital is |
|
Answer» 0 |
|
| 13. |
Number of protons in the nucleus of carbon atom is |
|
Answer» 7 |
|
| 14. |
Number of primary, secondary, tertiary and neo carbons in the following compound respectively are CH_(3) - CH_(2) - overset(overset(CH_(3))(|))(CH) - underset(underset(CH_(3))(|))overset(overset(CH_(3))(|))(C ) - CH_(3) |
|
Answer» 4,2,1,1 |
|
| 15. |
Number of primary hydrogens in isobutane is |
|
Answer» |
|
| 16. |
Number of possible spectral lines which may be emitted in Brackett series in H atom, if electrons present in 9^(th) excited level returns to ground level, are |
|
Answer» 21 `sum 10-1 = 9 +8+7 + 6 + 5 + 4 +3+2 + 1` No of lines in BRACKETT series = 6 |
|
| 17. |
Number of possible resonance structures of |
Answer» 
|
|
| 18. |
Number of possible position isomers for dichlorobenzene is |
|
Answer» 2 |
|
| 19. |
Number of possible optically active isomers |
|
Answer» |
|
| 20. |
Number of possible geometrical isomers for 1, 3-pentadiene is |
|
Answer» 8 |
|
| 21. |
Number of possible geometrical isomers for 2, 4 - hexadjenie is |
|
Answer» 8 |
|
| 22. |
Number of possible alkynes with formula C_5H_8 is |
|
Answer» 3 (i)`CH_3CH_2CH_2C-=CH` , (II)`CH_3CH_2C-=C CH_3`, (III)`(CH_3)_2CH-C-=CH` |
|
| 23. |
Number of positional isomers of the compound 2-Bromo Bicyclo [2,2,1] Heptane(including the given one |
|
Answer» 2 
|
|
| 24. |
Number of positional isomers possible for methyloctane |
|
Answer» `C-C-underset( C )underset(|)C-C-C-C-C-C` `C-C-C-underset( C )underset(|)C-C-C-C-C` |
|
| 26. |
Number of pi bonds and sigma bonds in the following structure is |
|
Answer» 6,19 In the given compound will have the correctstructure as There 5 `pi`-bonds and 8 C - H + 11C - C `SIGMA`-bonds. So, 19 `sigma`-bonds are present in the above MOLECULE. 
|
|
| 27. |
Number of per oxide bonds in per xenate ion[XeO_(6)]^(4-) is : |
|
Answer» |
|
| 28. |
Number of periods present in the long form of periodic table |
| Answer» | |
| 30. |
Number of oxidation states that the most electronegative element can exhibit is its compounds with other elements |
|
Answer» 9 |
|
| 31. |
Number of outer shells partially filled for representative elements |
| Answer» Answer :B | |
| 32. |
Number of orbitals of their other most orbits are more than 4. |
| Answer» SOLUTION :Because, NUMBER of ORBITALS of their OUTER most 9 orbits are more then 4. (1-3, 3-3p, 5-3d) | |
| 33. |
Number of organic products formed in following feaction. H-underset(O)underset(||)C-overset(O)overset(||)C-underset(O)underset(||)C-H underset("excess")overset(NH_(2)-NH_(2))rarr |
Answer» 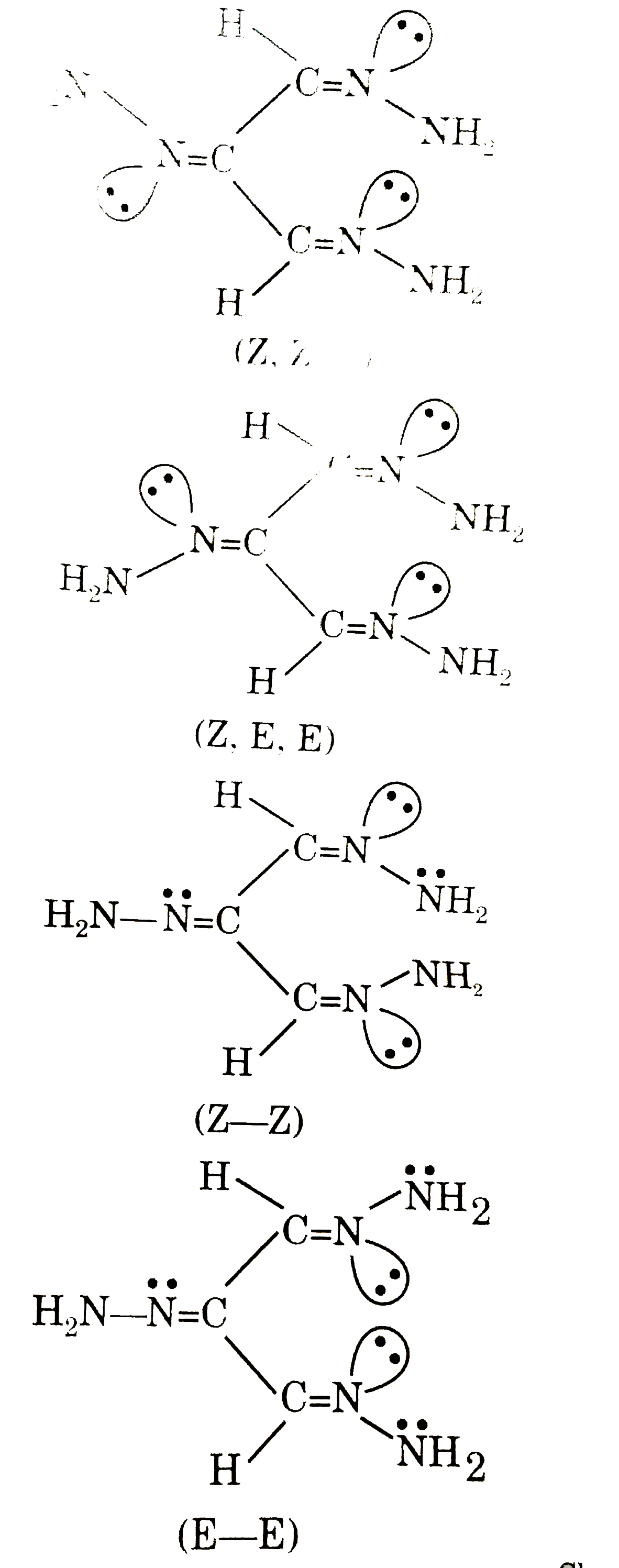
|
|
| 34. |
Number of orbitals in h sub-shell is |
|
Answer» Solution :`{:(s,p,d,F,g,h),(l=0,1,2,3,4,5):}` Number of orbitals = 5 X 2 +1 =11 |
|
| 35. |
Number of optically active isomers of |
|
Answer» |
|
| 36. |
Number of nodes in above plot is: |
|
Answer» |
|
| 37. |
Number of nodes in above plot is : |
|
Answer» 1 |
|
| 39. |
Number of nodal centres for 2s orbital |
|
Answer» 1 |
|
| 40. |
Number of nodal planes that a p-orbital has |
|
Answer» 0 |
|
| 41. |
Number of N_(2) molecules present in 1 litre of vessel at 1 atm, 273K when compressibility factor is 1.2. |
|
Answer» `2.23xx10^(24)` |
|
| 43. |
Number of monochloro benzenes is |
|
Answer» 1 |
|
| 44. |
Number of moles of water required for the hydroysis of 1 mole of Be_(2)C |
|
Answer» |
|
| 45. |
Number of moles of valency electrons present in 6.022xx10^(23)NH_(4)^(+) ions is__________. |
|
Answer» 1 ION `rarr8` valency `E^(-)` 1 mole of ions `rarr8` moles of valency `e^(-)` |
|
| 46. |
Number of moles of the solute dissolved per dm^(3) of solution is ……………… |
| Answer» SOLUTION :MOLARITY | |
| 47. |
Number of moles of ozone used for one mole of the given unsaturated hydrocarbons ? |
|
Answer» 1 |
|
| 48. |
Number of moles of oxygen required for thecombustion of hexyne and toluene respectively are |
|
Answer» 8 and 9 |
|
| 49. |
Number of moles of OH^(-) produced by dissolving 1 mole of B(OH)_(3) in water ? |
|
Answer» |
|
| 50. |
Number of moles of MnO_(4)^(-) required to oxidize one mole of ferrous oxalate completely in acidic medium will be |
|
Answer» 7.5 moles `FeC_(2)O_(4)rarrFe^(2+)+C_(2)O_(4)^(23-)` `Fe^(2+)rarrFe^(3+)+e^(-)` `C_(2)O_(4)^(2-)rarr2CO_(2)+2e^(-)` since one mole of `FeC_(2)O_(4)` loses 3 moles of electrons while one mole of `KMnO_(4)` accepts five moles of electrons THEREFORE NUMBER of moles of `KMnO_(4)` required to oxidised one mole of `FeC_(21)O_(4)=3//5 =0.6` mole |
|