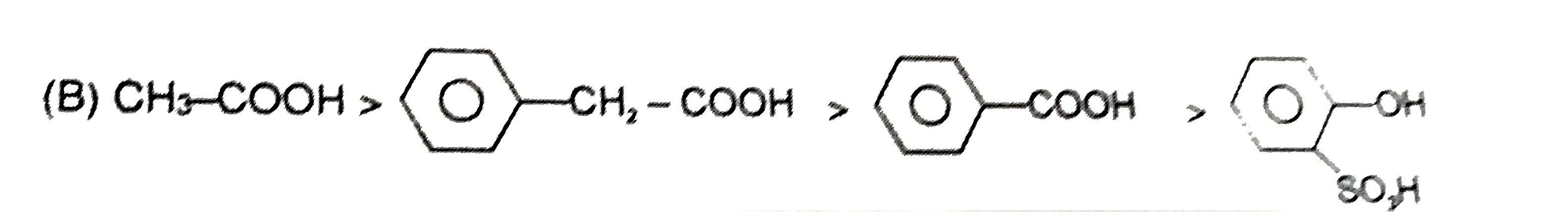Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Out of benzene, m–dinitrobenzene and toluene which will undergo nitration most easily and why? |
| Answer» SOLUTION :Toluene. This is because `-CH_3`GROUP is an electron releasing (electron-donating) group and ACTIVATES the ring. On the other HAND, `-NO_2` group is an electron-withdrawing group and DEACTIVATES the ring. This is why toluene undergoes nitration most easily. | |
| 2. |
Out of benzene, m-dinitrobenzene and toluene which will undergo nitration most easily and why ? |
|
Answer» Solution :The ease of nitration depends on the presence of electron density on the compound to form nitrates. Nitration reaction are examples of electrophilic substitution reactions where an electron-rich species is attacked by a nitronium ion `(NO_(2)^(-))`. Now, `CH_(3)^(-)` group is electron DONATING and `NO_(2)^(-)` is electron withdrawing. Therefore, toluene will have maximum electron density among the three COMPOUNDS followed by benzene. On the other hand, m-Dinitrobenzene will have the LEAST electron density. Hence, it will undergo nitration with difficulty. Hence, the INCREASING order of nitration is as FOLLOWS : 
|
|
| 3. |
Out of benzene , ethylene and acetylene, the carbon-carbon bond length is longest in _______ |
|
Answer» |
|
| 4. |
Out of benzen m - dinitrobenzene and toluene whichwill undergo nitration most easilyand why |
|
Answer» Solution :`CH_(3)` groupis an electrondonating groupwhile`NO_(2)` groupis electronwithdrawinggroupthereforemaximumdensitywill be therein toluenefollowedby benzeneand it islestin m- dimitrobenzenethereforecaseofnitrationdecreasesint the ORDER there `GT `benzene `gt m-` dinitrobenzne |
|
| 5. |
Out of a straight chain alkane and a straight chain alkene having the same number of carbon atoms, which has a higher octane number? |
| Answer» SOLUTION :STRAIGHT CHAIN ALKENE | |
| 6. |
Our environment consists of ......... |
|
Answer» LITHOSPHERE |
|
| 7. |
Ostwald's dilution law is not obeyed by |
|
Answer» `CH_(3)COOH` |
|
| 8. |
Ostwald dilution law is applicable to |
|
Answer» Strong ELECTROLYTES |
|
| 9. |
Osmotic pressure (pi)of a solution is given by the relation |
|
Answer» ` PI = NRT` |
|
| 10. |
What is meant by osmotic regulation? |
|
Answer» `pi = NRT` `pi = (n)/(V) RT` `pi V = nRT` |
|
| 12. |
Osmotic pressure of the solution can be increased by |
|
Answer» INCREASING the TEMPERATURE of the solution |
|
| 13. |
Orthohydrogen and parahydrogen have |
|
Answer» Same electronic spin, PHYSICAL and CHEMICAL properites |
|
| 14. |
Orthofluorophenol is more volatile than its meta and para isomers. Why? |
|
Answer» Solution :Intramolecular HYDROGEN bonding is PRESENT in orthofluorophenol, which does not lead to ASSOCIATION of molecules. Hence it is VOLATILE. Intermolecular hydrogen bonding is present in metal and para isomers, which LEADS to molecular association. |
|
| 16. |
Orthoboric acid (H_(3)BO_(3)) and metaboric acid (HBO_(2)) differ in respect of |
|
Answer» BASICITY Basicity, Structure and Melting point |
|
| 17. |
Orthoboric acid acts as a Lewis acid . Why ? |
|
Answer» Solution :It ACTS as a weak monobasic lewis acid by accepting a hydroxyl ion. `underset ("BORIC acid ( lewis acid)")(B(OH)_(3))+H^(+) OH^(-) RARR [B(OH)_(4)]^(-)+H^(+)` |
|
| 18. |
Ortho-Nitrophenol is less soluble in water than p- and m- Nitrophenols because |
|
Answer» <P>MELTING point of o-Nitrophenolis lower than m-nitrphenols because o-nitrphenol shows intramolecular HYDROGEN bonding . |
|
| 19. |
ortho-Nitrophenol is less soluble in water than p and m-nitrophenols because |
|
Answer» o-nitrophenol is more VOLATILE in steam than those of m-and p-isomers 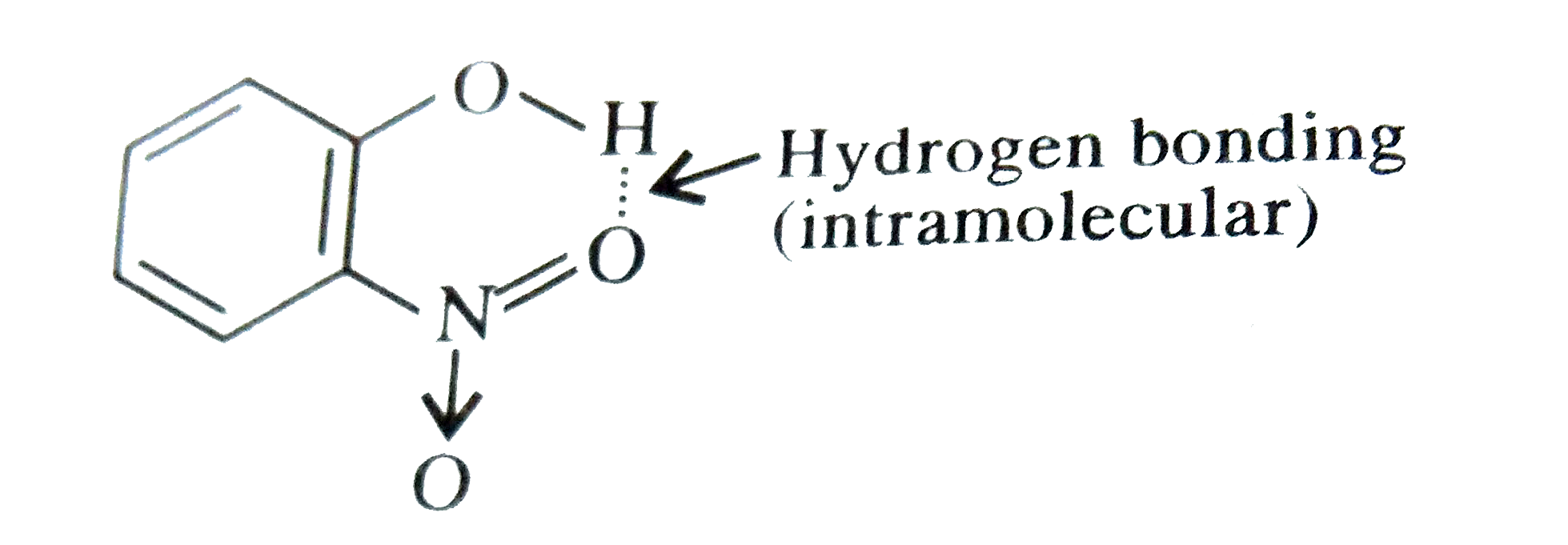
|
|
| 20. |
Ortho nitro phenol and paranitro phenol are |
|
Answer» Cis-trans ISOMERISM |
|
| 21. |
Ortho hydrogen has magnetic moment___________ that of a proton. |
|
Answer» twice |
|
| 22. |
Ortho, meta and para dichlorobenzenes are |
|
Answer» CHAIN ISOMERS  They are POSITION Isomers. |
|
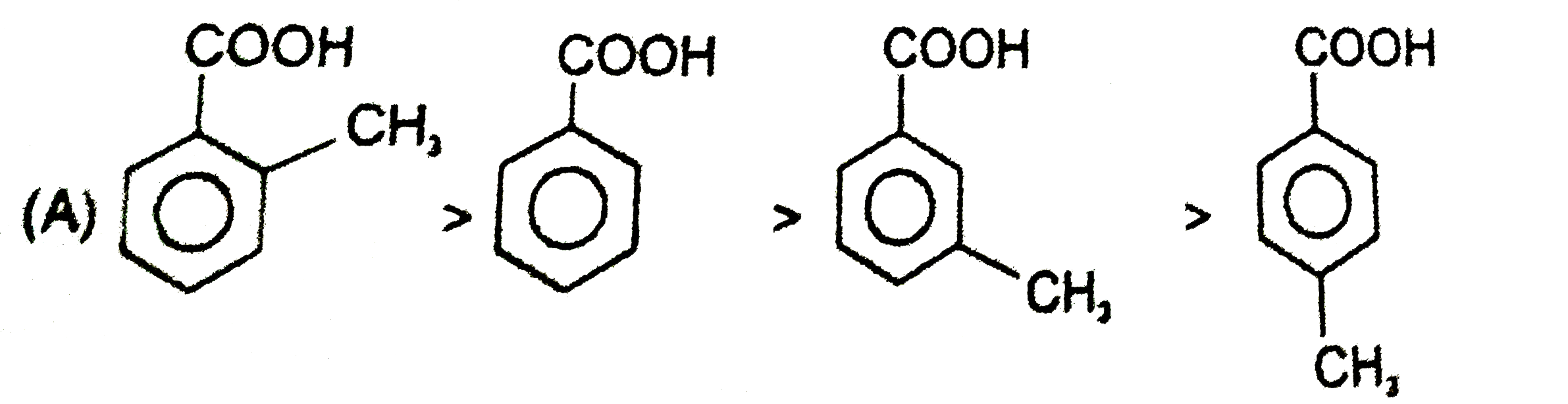
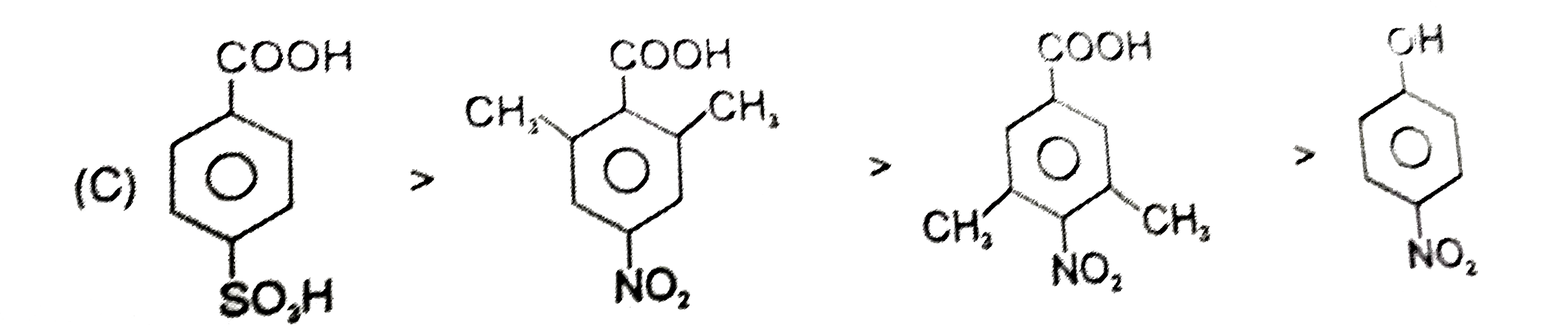
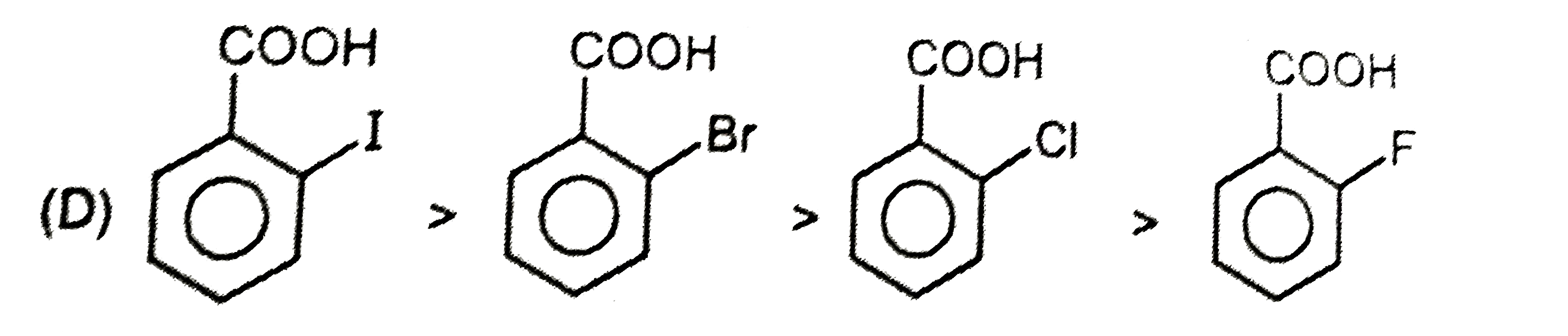
| 23. |
Ortho effect is special type of effect that is shown by o-subsituents .This ortho-effect operates at the benzoic acids irrespective of the polar type. Nearly all o-substituted benzoic acid are stronger than benzoic acid. Benzoic acid is a resonance stabilised and so the carboxyl group is coplaner with the ring. An o-subsituent tends to prevent this coplanarity. |
|
Answer»
|
|
| 24. |
Ortho effect is special type of effect that is shown by o-subsituents .This ortho-effect operates at the benzoic acids irrespective of the polar type. Nearly all o-substituted benzoic acid are stronger than benzoic acid. Benzoic acid is a resonance stabilised and so the carboxyl group is coplaner with the ring. An o-subsituent tends to prevent this coplanarity. What is the order of K_(a) of following compounds ? |
|
Answer» `I gt II gt III gt IV gt V` |
|
| 25. |
Ortho boric acid and metaboric acid differ in respect of |
|
Answer» Basicity |
|
| 26. |
Ortho and parahydrogen differ in… |
|
Answer» ATOMIC number |
|
| 27. |
Ortho and para hydrogen differ in.... |
|
Answer» PROTON SPIN |
|
| 28. |
Ortho and para-nitro phenol can be separated by |
|
Answer» AZEOTROPIC distillation |
|
| 29. |
Ortho- and par - hydrogen differ in |
|
Answer» Atomic number para- hydrogen ` = + 1/2 - 1/2 = 0` |
|
| 30. |
Ortho and para hydrogen differ in |
| Answer» Answer :A | |
| 31. |
Original solution of salt or mixture should not be prepared in concentrated HNO_(3) because it: |
|
Answer» is highly corrosive |
|
| 32. |
Orientation of orbitals is given by |
|
Answer» PRINCIPAL QUANTUM NUMBER |
|
| 33. |
Organic reactions are given in column-I and reaction names are given in column-II. Match column-I with column-II and chose the correct answer |
|
Answer» `(i) rarr s, (ii) rarr q, (iii) rarr p, (iv) rarr r` `CH_(3)CH_(2)I + KOH rarr CH_(3)CH_(2)OH + KI` (ii) ELIMINATION reaction: HBr is eliminated `CH_(3)CH_(2)Br + KOH overset("alcohol")rarr CH_(2)=CH_(2) + KBr + H_(2)O` (iii) Addition reaction: `Cl_(2)` is added to `pi`-BOND `CH_(2)= CH_(2) + Cl_(2) rarr CH_(2)CL - CH_(2)Cl` (iv) Rearrangement reaction: `CH_(3)CH_(2)- CH= CH_(2) underset("catalyst")overset("ACID")rarr CH_(3)-underset(underset(CH_(3))(|))(C )HCH_(3)` |
|
| 34. |
Organic liquid vapourises at a temperature below its boiling point in steam distillation because |
|
Answer» Mixture boils when sum of vapour pressure of water and ORGANIC becomes EQUAL to atomspheric pressure. |
|
| 35. |
Organic particulates include PAH which is an abbreviation for _____ |
|
Answer» |
|
| 36. |
Organic halogen compounds used in industry as solvents are chlorides rather than bromides and iodides. Explain. |
| Answer» Solution :Organic ALKYL chlorides are used in industry as solvent because chlorides are more VOLATILE than bromides and IODIDES. | |
| 37. |
organic compound sometimes adjust their electronic as well as steric structures to attain stability. Among the following, the compound having highest dipole moment is: |
|
Answer»
|
|
| 39. |
Organic compound is………… soluble in organic solvent then continuous extraction technique is used |
|
Answer» |
|
| 40. |
Organic catalysts differ from inorganic catalysts |
|
Answer» By acting at very high temperature |
|
| 41. |
Organic acid without a carboxylicacidgroup is |
|
Answer» ASCORBIC ACID |
|
| 42. |
Ordinary water contain one part of heavy water per 6000 parts by weight. The number of heavy water molecules present in drop of water of volume 0.01 ml is (Density of water is 1gm//ml) |
|
Answer» `2.5 XX 10^(16)` No. of molecules `(W)/(Mw) xx N` |
|
| 43. |
Oridinary water contain one part of heavy water per 6000 parts of water by weight. The number of heavy water molecules present in a drop of water of volume 0.01 mL is (density of water 1 g/mL) |
|
Answer» `2.5XX10^(16)` |
|
| 44. |
Order of the bond strength of C – H bonds involving sp, sp^(2) and sp^(3) hybridized carbon atom is : |
|
Answer» `SP gt sp^(2) gt sp^(3) ` |
|
| 45. |
Order of rate of electrophilic addition reaction with HBr will be : |
|
Answer» IV gt I gt III gt II |
|
| 46. |
Order of oxidation number of S in SO_(3)^(-2),S_(2)O_(4)^(-2)andS_(2)O_(6)^(-2). |
|
Answer» `S_(2)O_(4)^(-2)ltSO_(3)^(-2)ltS_(2)O_(6)^(-2)` |
|
| 47. |
Order of radioactive disintegration reaction is |
|
Answer» Zero |
|
| 48. |
Order of K_(a) which can be predicted by following reaction is: |
|
Answer» `NH_(3) gt Ph_(3)CH gt C_(2)H_(2) gt H_(2)O` |
|
| 49. |
Order of ionization enthlphy in second period of the following is true. |
|
Answer» `Li LT B lt Be lt N lt C ` `Li lt B lt Be lt C lt O lt N lt F lt NE` |
|