Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Ozonolysis of products of 2-methyl-2-butene are |
|
Answer» 2 MOLES of HCHO |
|
| 2. |
Ozonolysis of hydrocarbon containing two double bond, ethanal, propanone and glyocal is formed. Give the structure of hdyroacarbon ? |
|
Answer» Solution :Hydrocarbon contains 2 double bond, it is called diene. Product (a) Ethanal `CH_(3)CHO 2C` are there (B) Propnone `CH_(3)COCH_(3) 3C` are there and (c) Glyocal `CHO-CHO 2C` are there `THEREFORE` Diene contains total of carbon. ALDEHYDE group are at near and at the end. At the end their is ketone, position, of OXYGEN O are ADDED, structure of hydrocarbon is as follows :  IUPAC Name : 2-Methyl-hexa-2, 4-diene |
|
| 3. |
Ozonolysis of products obtained from alkene, 2-methyl-2-butene are |
|
Answer» `HCHO+HCHO` |
|
| 4. |
Ozonolysis of one mole of benzene gives |
|
Answer» One mole GLYOXAL and two moles methyl glyoxal |
|
| 5. |
Ozonolysis of an organic compound gives formaldehyde as one of the products. This confirms the presence of |
|
Answer» a VINYL group 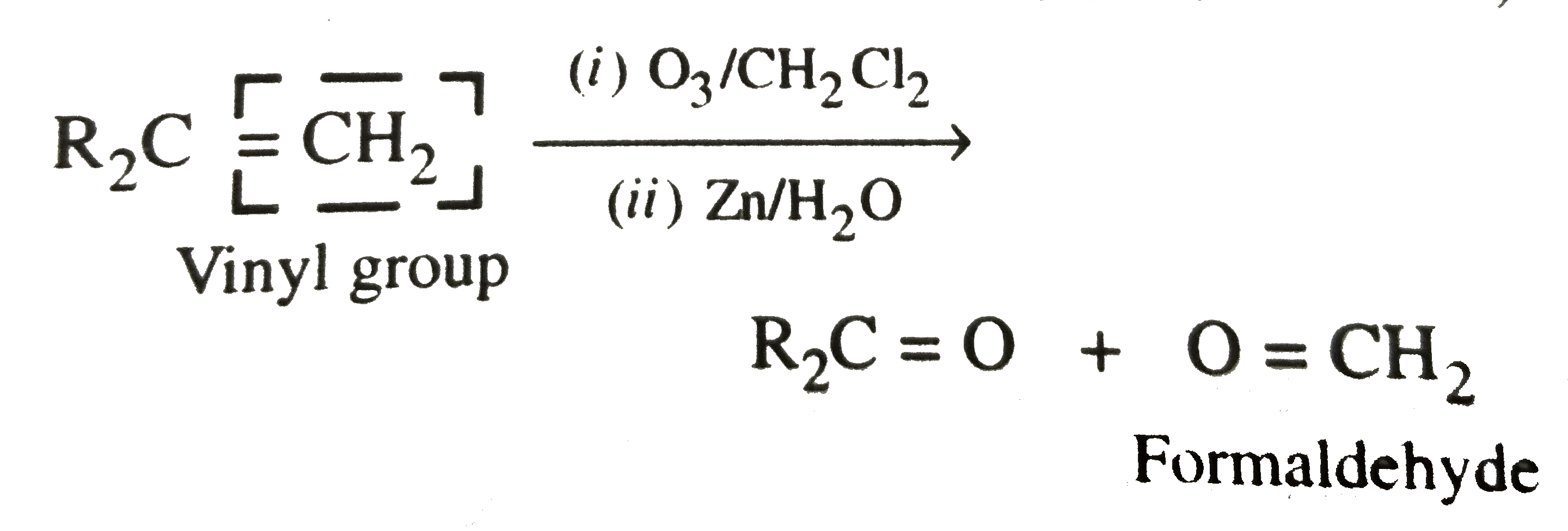
|
|
| 6. |
Ozonolysis of an organic compound gives formaldehyde as one of products. This confirms the presence of |
|
Answer» TWO ethylenic double bonds |
|
| 7. |
Ozonolysis of an organic compound A produces acetone and propionaldehyde in equimolar mixture. Identify A from the following compounds. |
|
Answer» 2-methyl pent-l-ene |
|
| 8. |
Ozonolysis of an alkene produces only one dicarbonyl compound. The structure of the alkene is |
|
Answer» `CH_3-CH=CH-CH_3` 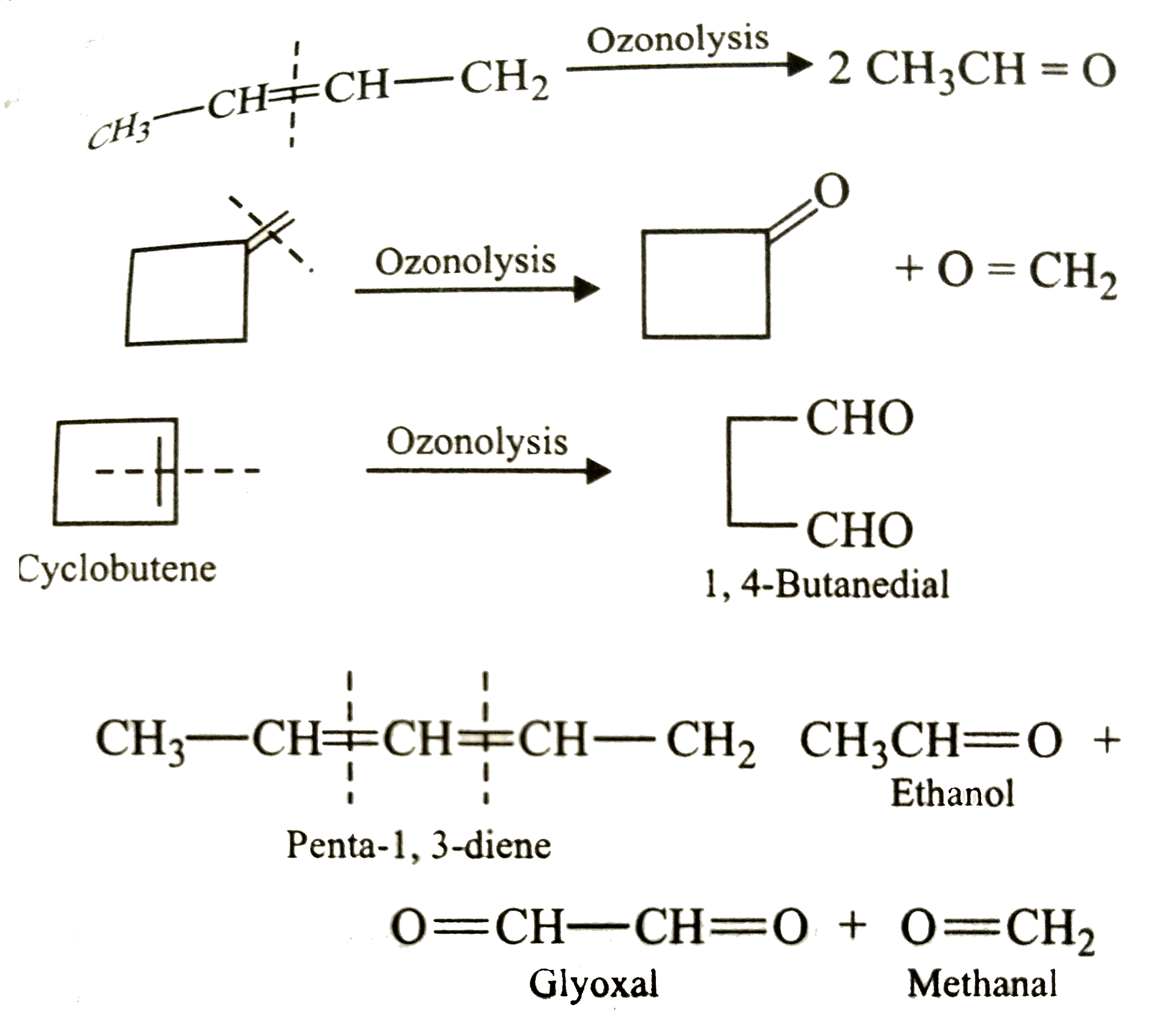 Thus, only CYCLOBUTENE PRODUCES only one dicarbonyl COMPOUND , i.e. , OPTION (b) is correct. |
|
| 9. |
Ozonolysis of an alkene gave two molecules of ethanal (acetaldehyde). Give the structure of the alkene. |
|
Answer» Solution :[Write the structures of the PRODUCTS with carbobtl groups facing each other. Then remove oxygen atoms from each of the CARBONYL compound and put double BONDS between the two carbon atoms to get the ALKENE.] Products of OZONOLYSIS:`CH_3-CHO`(ethanol)`OHC-CH_3` (ethanal) Hence the structure of alkene is `CH_3-CH=CH-CH_3`(but-2-ene) |
|
| 10. |
Ozonolysis of an alkence gave propanone (acetone) and ethanal. Give the structure and IUPAC name of the alkene. |
Answer» SOLUTION :
|
|
| 11. |
Ozonolysis of a organic compound gives formaldehyde as one of the products. This confirms the presence of : |
|
Answer» two ethylenic DOUBLE bonds |
|
| 12. |
Ozonolysis of 2,3-dimethylbut-1-ene followed by reduction with zinc and water gives |
|
Answer» METHANAL and hexanoic ACID 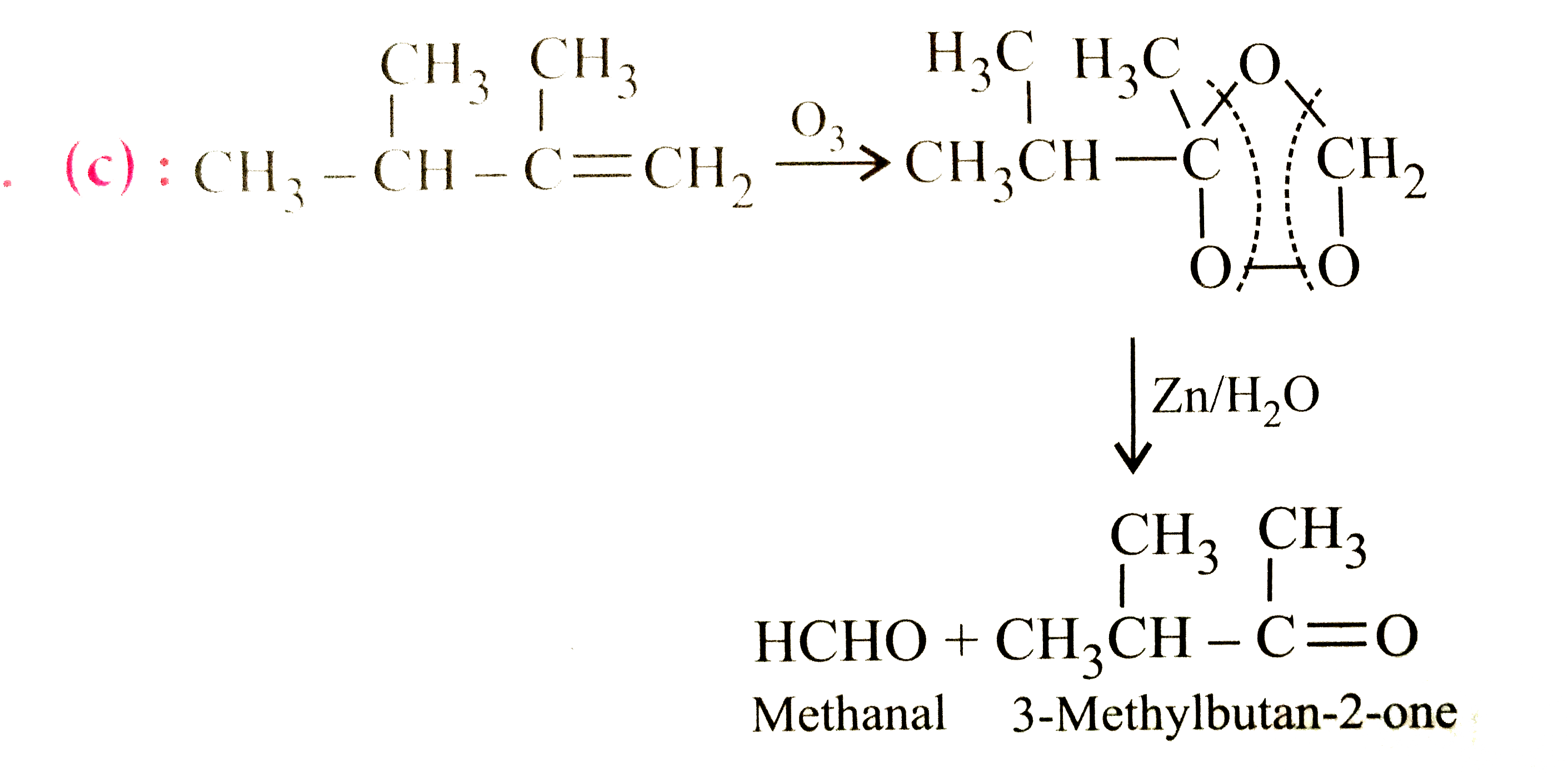
|
|
| 13. |
Ozonization of propene gives …….. (i) ethanol and methanal (ii) ethanol and methanol. (iii) hybridization of carbon is change. (iv) propene gives monotriozonied. |
|
Answer» |
|
| 15. |
Ozone layer of stratosphere requires protection from indiscriminate use of |
|
Answer» Pesticides |
|
| 16. |
Ozone layer is present in |
|
Answer» TROPOSPHERE |
|
| 17. |
Ozone layer is depleted by the reactive________ |
|
Answer» HYDROGEN atom |
|
| 19. |
Ozone layer acts as one of the most important life support system. The major cause of ozone layer destruction is believed to be the release of chlorofluorocarbon compounds. UV radiation causes the chlorofluouocarbons to dissociate : CF_(2) Cl overset(hv)(rarr) Cl* +CF_(2) CI A highly reactive chlorine atomis responsible for the decomposition of ozone. Cl* + O_(3) (g) rarr ClO* + O_(2) (g) ClO* + O(g) rarr Cl* + O_(2) (g) Free radicals of chlorine decompose ozone molecules in a chain reaction. The depletion of ozone layer leading to ozone hole has, however, been mainly observed in the stratosphere over Antarctica. Special type of cloud called polar stratospheric colouds are formed over Antarctica.These clouds (contain solid HNO_(3).3H_(2)O) play an important role in ozone depletion. ClO* + NO_(2) rarr ClONO_(2) ClONO_(2) + H_(2) O rarr HOCl + HNO_(3) ClONO_(2) + HCl rarr Cl_(2) + HNO_(3) HOCl overset(hv)(rarr) HO* + Cl* The reactive chlorine atoms thus formed are rendered to deplete ozone layer. Peeling of ozone umbrella, which protects us from UV rays is caused by: |
|
Answer» ACID rain |
|
| 20. |
Ozone layer acts as one of the most important life support system. The major cause of ozone layer destruction is believed to be the release of chlorofluorocarbon compounds. UV radiation causes the chlorofluouocarbons to dissociate : CF_(2) Cl overset(hv)(rarr) Cl* +CF_(2) CI A highly reactive chlorine atomis responsible for the decomposition of ozone. Cl* + O_(3) (g) rarr ClO* + O_(2) (g) ClO* + O(g) rarr Cl* + O_(2) (g) Free radicals of chlorine decompose ozone molecules in a chain reaction. The depletion of ozone layer leading to ozone hole has, however, been mainly observed in the stratosphere over Antarctica. Special type of cloud called polar stratospheric colouds are formed over Antarctica.These clouds (contain solid HNO_(3).3H_(2)O) play an important role in ozone depletion. ClO* + NO_(2) rarr ClONO_(2) ClONO_(2) + H_(2) O rarr HOCl + HNO_(3) ClONO_(2) + HCl rarr Cl_(2) + HNO_(3) HOCl overset(hv)(rarr) HO* + Cl* The reactive chlorine atoms thus formed are rendered to deplete ozone layer. The ozone layer is present in : |
|
Answer» TROPOSPHERE |
|
| 21. |
Ozone is useful in ______ and harmful in ___ |
|
Answer» TROPOSHERE & Mesosphere |
|
| 22. |
Ozone is used for purifying water because.... |
|
Answer» it dissociates and release oxygen. It inactivates and oxidizes organic matter, contaminates, pesticides, viruses and bacteria faster than chlorine. `O_3` do not form TMH which have unpleasent odour and are also carcinogenic. `O_3` is very good biocide. Ozone also ABSORB UV radiation. |
|
| 23. |
Ozone is present in ________(region) of the atmosphere |
|
Answer» |
|
| 24. |
Ozone is present in ........ layer of the atmosphere |
| Answer» SOLUTION :STRATOSPHERE | |
| 25. |
Ozone is harmful in the environment sigment .r. but it is useful in the environment sigment .y.. What are r and y? |
| Answer» Solution : .X. is TROPOSPHERE. Ozone is a POLLUTANT. .y. is stratosphere. Ozone ACTS as a PROTECTIVE umbrella. | |
| 26. |
Ozone is a toxic gas and is a strong oxidising agent even then its presence in the stratosphere is very important. Explain what would happen if ozone from this region is completely removed ? |
|
Answer» Solution :The ozone layer in the stratosphere is a natural feature of the EARTH.s environment. The ozone layer exists between 20 to 35 km above the earth surface layer. This layer protects the earth from the harmful effects of the ultraviolet RADIATION of the sun. A depletion of ozone layer is considered as a serious THREAT to all forms of life on the earth. A 5% DECREASE in ozone concentration could increases the including cataract formation. It can cause genetic mutations and destroy crops and other forms of vegetation. Aquatic animals and aquatic plants are generally affected by UV-radiation. |
|
| 27. |
Ozone is a gas heavier than air. Why does ozone layer not settle down near the earth? |
| Answer» Solution :Ozone LAYER is formed in the stratosphere at an ALTITUDE of about 25-30 KM from earth's SURFACE. A this altitude, the force of gravitation is NEGLIGIBLE | |
| 28. |
Ozone in the stratosphere is depleted by |
|
Answer» `CF_2Cl_2` |
|
| 29. |
Ozone hole refers to |
|
Answer» Hole in ozone LAYER |
|
| 30. |
Ozone have a angular shape and it has - |
|
Answer» `2 sigma` and `2pi` BOND |
|
| 31. |
Ozone depletion will cause........ |
|
Answer» FOREST fires |
|
| 32. |
Ozone an estimated in air by passing certaing volume of air through an acidified or neutral or basic KI solution when O_(2) is evolved and I^(ɵ) is oxidised to I_(2). Free I_(2) evolved is titrated with standard Na_(2)S_(2)O_(3) solution. In an experiment 10 L of air at 1 atom and 27^@C was passed through an alkaline KI solution and I_(2) liberated requried 2 " mL of " (M)/(800)Na_(2)S_(2)O_(3) solution. Calculate the volume percentage of O_(3) in sample. |
|
Answer» Solution :Write the balanced REDOX REACTIONS in basic medium. `2I^(ɵ)+H_(2)O+O_(3)toO_(2)+I_(2)+2overset(ɵ)(O)H` (n factor `O_(3)=2`) `I_(2)+2e^(-)to2I^(ɵ)(n=2)` `S_(2)O_(3)+10overset(ɵ)(O)Hto2SO_(4)^(2-)+5H_(2)O+8e^(-)(n=8)` `thereforem" Eq of "I_(2)=m" Eq of "Na_(2)S_(2)O_(3)` `=2xx(1)/(800)xx8` (n factor) `=0.02` mmol es of `I_(2)=(0.02)/(2)=mmol es of O_(3)` `[becausen=2` for both or molar ratio of `O_(3):I_(2)::1:1`] `P_(O_(3))=(nRT)/(V)` `=(0.01xx10^(-3)xx0.0821xx300)/(10)` `=246.3xx10^(-7)atm` Volume `% of O_(3)=246.3xx10^(-7)xx100` `=2.463xx10^(-3)%` |
|
| 33. |
Ozonc layer is present in the region |
|
Answer» TROPOSPHERE |
|
| 34. |
Oxygen when passed through an ethereal solution of phenyl magnesium iodide followed by acid hydrolysis acid hydrolysis gives |
|
Answer» Benzene 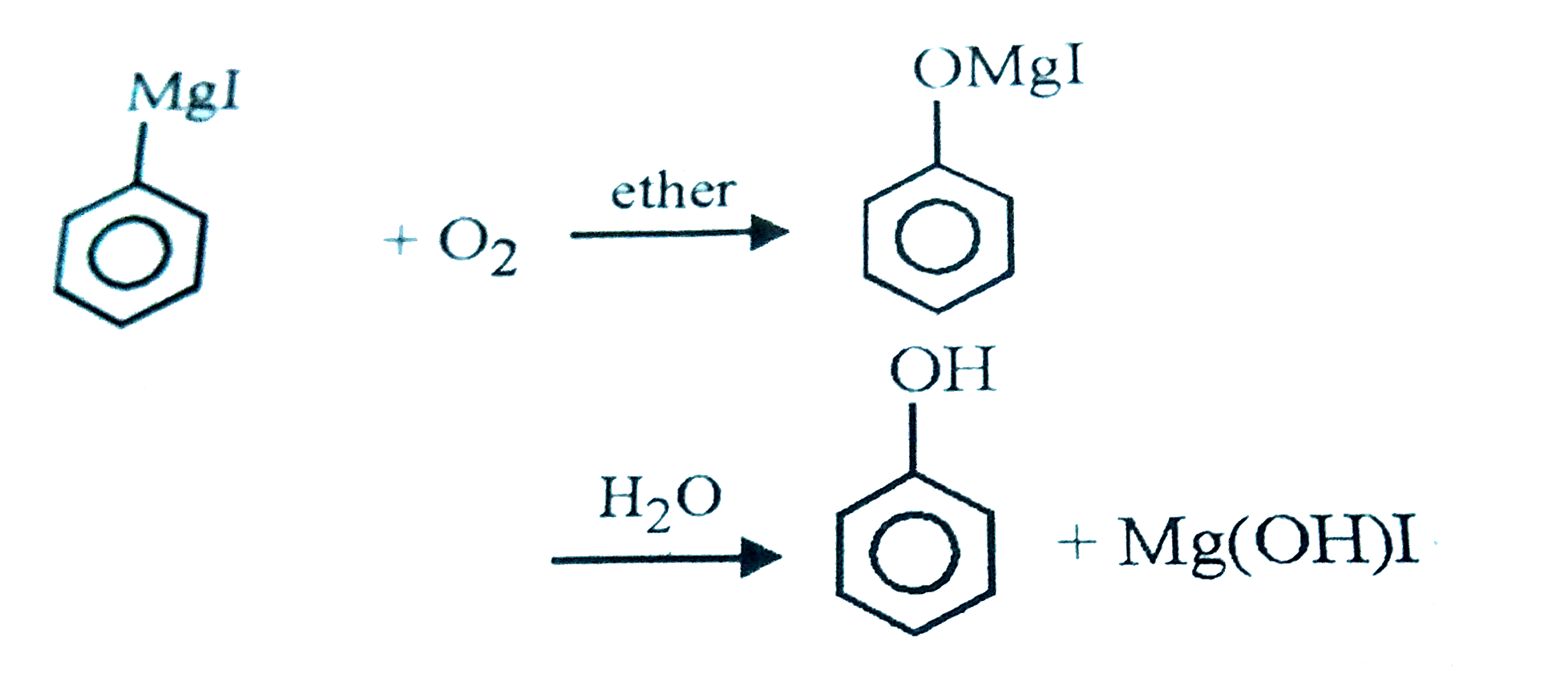
|
|
| 36. |
Oxygen, which is 16 times as dense as hydrogen, diffuses: |
|
Answer» `(1)/(16)` TIMES as fast |
|
| 37. |
Oxygen is present in a 1 litre vessel at a pressure of 10^(-7) Nm^(-2). Calculate the number of oxygen molecule at 0^(@)C |
|
Answer» Solution :`PV=nRT,P=10^(-7) Nm^(-2), v=10^(-3) m^(3), T=273K` `n=(PV)/(RT)=(10^(-7)xx10^(-3))/(8*314xx273)=2*653xx10^(10)` |
|
| 38. |
Oxygen is present in a flask of 1.12L capacity at a pressure of 7.6 xx 10^(-10) mm of Hg at 0^@C. The number of oxygen molecules in the flask is |
|
Answer» `1.5 XX 10^10` |
|
| 39. |
Oxygen is present in a 1-litre flask at a pressure of 7.6 xx 10^(-10) mmHg at 0°C. Calculate the number of oxygen molecules in the flask. |
|
Answer» Solution :PRESSURE `=7.6 xx 10^(-10)` mm `=0.76 xx 10^(-10) cm` `=(0.76 xx 10^(-10))/76` ATM (1 atm = 76 cm) `=10^(-12)` atm Volume = 1 litre, R = 0.0821 lit. Atm/K/mole. Temperature = 273 K We know that pV = nRT or `n=(pV)/(RT)` `n=(10^(-12) xx 1)/(0.0821 xx 273) = 0.44 xx 10^(-13)` No. of molecules = no. of MOLES `xx` AV. const. `=0.44 xx 10^(-13) xx 6.022 xx 10^(23) = 2.65 xx 10^(10)` |
|
| 40. |
Oxygen is present in a 1 litre flask at a pressure of 7.6 xx 10^(-10) mm of Hg. Calculate the number of oxygen molecules in the flask at 0^@C |
|
Answer» Solution :According to the ideal gas equation, PV = nRT In the present case, `P= 7.6 XX 10^(-10)mm = (7.6xx10(-10))/760 = 1.0xx10^(-12)` atm ( `:. 1 " atm " = 760 mm Hg)` `V = 1 L, and T = 0^@C = 273 K, " and "R=0.0821 " litre atm " K_(-1) mol^(-1)` `:.n=(PV)/(RT)= (1.0 xx 10^(-12) xx1)/(0.0821 xx 273) = 4.5 xx 10^(-14)` moles `:. " 1 mole " = 6.022 xx 10^(23)` molecules `:.4.5 xx 10(-14) " moles "= 6.022 xx 10^(23) xx 4.5 xx 10^(-14) = 2.7 xx 10^(10)`molecules Hence, the number of molecules of OXYGEN present in the flask at `0^@C " are " 2.7 xx 10^(10)` . |
|
| 41. |
Oxygen occurs in nature as a mixture of isotopes ""^16O, ""^17O " and " ""^18O having massesof 15.995 u, 16.999 u and 17.999 u and relative abundance of 99.763%, 0.037% and 0.200% respectively. What is the average atomic mass of oxygen? |
|
Answer» |
|
| 43. |
Oxygen exhibits positive oxidation number in the molecule |
|
Answer» `K_2O` |
|
| 44. |
Oxygen deviates more from ideal nature at |
|
Answer» <P>`T = 25^(0) C , P= 1 ` atm |
|
| 45. |
Oxygen cannot exhibit tetravalency and hexavalency like sulphur. This is because |
|
Answer» Oxygen has two unpaired electrons |
|
| 46. |
Oxygen at 1 atm and 0^@C has a density of 1.4290 gL^(-1). Calculate the RMS velocity of oxygen molecules. |
| Answer» SOLUTION :`4.61 XX 10^4 " CM " s^(_1)` | |
| 48. |
Oxidizing power of chlorine in aqueous solution can be determined by the parameters indicatedbelow: (1)/(2)Cl_(2)overset((1)/(2) Delta_("disso")H^(c-))(rarr)Cl(g) overset(Delta_(cg) H^(c-))(rarr) Cl^(-)(g)overset(Delta_(hyd)H^(c-))(rarr) Cl^(-) (aq)The energy involved inthe conversion of (1)/(2)Cl_(2)(g) toCl^(-) (aq)( using the data,Delta_(disso) H_(Cl_(2))^(c-)= 240 kJmol^(-1),Delta_(eg)H = - 349 kJ mol^(-1) Delta_(hyd)H_(cl) ^(c-) = - 381kJ mol^(-1)) will be |
|
Answer» `- 850kJ MOL^(-1)` `DeltaH =(1)/(2)Delta_(diss)H_(Cl_(2))+ Delta_(eg) Cl_+Delta_(hyd) H_(Cl^(-))` `= ( 240)/(2) - 349-381kJmol^(-1)` `=120 -730=-610 kJ mol^(-1)` |
|
| 49. |
Oxidizing agents are spectes that |
|
Answer» OXIDIZE other substances |
|
| 50. |
Oxidising power of chlorine in aqueous solution can be determined by parameters indicated below: 1/2CI_(2)(g) overset(1/2Delta_("diss")H) to CI(g) overset(Delta_(hyd)H) to CI^(-aq) The energy involved in the conversion of 1/2CI_(2)(g) to CI^(-)(aq) using data, Delta_("diss")H_(CI2) = 240 kJ"mol"^(-1), Delta_("eg")HCI = -349 kJ"mol"^(-1), Delta_("hyd")H_(ci) = - 381 kJ "mol"^(-1) will be |
|
Answer» `+ 120 kJ"mol"^(-1)` `1/2CI_(2)(g) to CI^(-)(AQ)` `DeltaH = 1/2Delta_("diss")H(CI_(2)) + Delta_("eg") H(CI) + Delta_("HYD")H(CI^(-))` ` = (240)/(2) + (-349) + (-38) = -610 kJ " mol^(-1)` |
|

