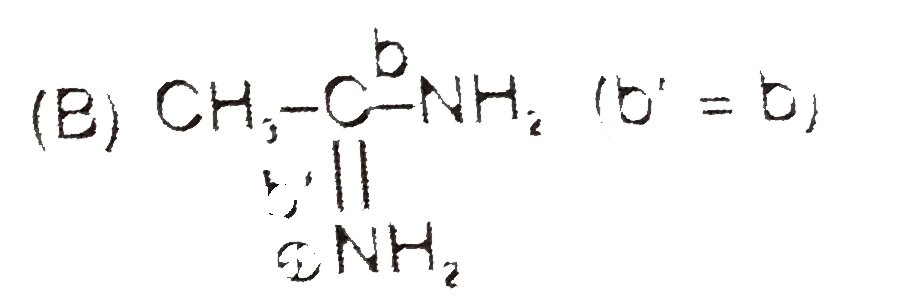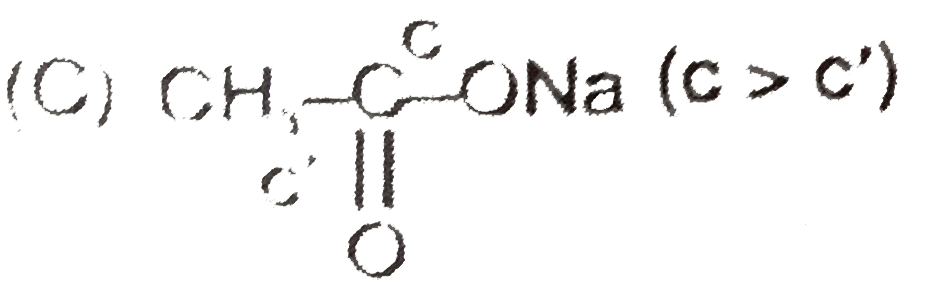Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The correct stability orderof the following resonance structure is (I)H_(2)bar(C)-overset(+)N-=N(II) H_(2)C = overset(+)N =bar(N)(III) H_(2)overset(+)C-N =bar(N)(IV) H_(2)bar(C) -N=overset(+)N |
|
Answer» `(I) GT (II) gt (IV) gt (III)` |
|
| 2. |
The correct stability order of the following resonance structure is : I) H_(2)C=overset(oplus)(N)=overset(ө)(N)"II) "H_(2)overset(oplus)(C)-N=overset(ө)(N)"III) "H_(2)overset(oplus)(C)-N=overset(ө)(N)"IV) "H_(2)overset(ө)(C)-N=overset(oplus)(N) |
|
Answer» `(I) gt (II) gt (IV) gt (III)` |
|
| 3. |
The correct stability order of the following resonanating structures is : underset("(I)")(CH_(2)=C=O)""underset("(II)")(C_(3)overset(-)C-overset(+)C=O)""underset("(III)")(H_(2)C=overset(+)C-overset(-)O)""underset("(IV)")(H_(2)overset(-)C-C-=overset(+)O) |
|
Answer» `(IV)GT(I)gt(III)gt(II)` |
|
| 4. |
The correct stability order of the following resonance structures is underset((I))(H_2C = overset(+)N = overset(-)(N)) underset((II))(H_2overset(+)C-N= overset(-)(N)) underset((III))(H_2overset(-)(C)-overset(+)N -=N) underset((IV))(H_2C overset(-)C-N = overset(+)(N)) |
|
Answer» `I GT II gt IV gt III` |
|
| 5. |
The correct stability order for the following species is |
|
Answer» `II GT IV gt I gt III` |
|
| 6. |
The correct stability order for boron halides is |
|
Answer» `BF_(3)gtBCl_(3)gtBBr_(3)gtBl_(3)` |
|
| 7. |
The correct signs of Delta S for the following four processes respectively are (i) Devitrification of glass (ii) Desalination of sea water (iii) N_(2) (g, 10 atm) rarr N_(2)(g, 2 atm) (iv) C (s, graphite) rarr C (s, daimond) |
|
Answer» `-,-,+,-` |
|
| 8. |
the correctset of the four quantumnumber forthe valenceelectronor Rubidumatom (z=37) is |
|
Answer» `5,0,0, +(1)/(2)` |
|
| 9. |
The correct set of quantum numbers for the unpaired electron of chlorine atom is |
|
Answer» n=2, l=1, m=0 |
|
| 10. |
The correct set of quantum numbers for a 4d electron is (Kerala Engineering) |
|
Answer» `4, 3, 2, +1//2` |
|
| 11. |
The correct set of quantum number for the unpaired electron of chlorine atom is |
|
Answer» `2, 1, -1, +(1)/(2)` |
|
| 12. |
The correct set of four quantum numbers for the valence electron of rubidium (Z = 37) is |
|
Answer» 5, 1, 1, `+1//2` HENCE, for the valence electron `(5s^(1)), N = 5, l = 0, m = 0, s = 1//2` |
|
| 13. |
The correct set of oxidation number of nitrogen atom in cyanide ion, ammonium ion, nitrite and nitrate ion, respectively is : |
|
Answer» `-3,+3,-3,-5` 
|
|
| 14. |
The correctset of quantum numberfor 4delectron |
|
Answer» `4,3-1+(1)/(2)` `m=+2 `to `-2,2 =+ (1)/(2)` |
|
| 15. |
The correct set of four quantum numbers for the valence electrons of Rubidium atom (Z = 37) |
|
Answer» `5,1,+1/2` |
|
| 16. |
The correct set of four quantum numbers for the valence electron of rubidium atom (Z = 37) is |
|
Answer» 5,0,0,+1/2 |
|
| 17. |
The correct set of four quantum number for the unpaired electron of the element Z = 21 is |
|
Answer» `N=3 , l =2 m =1 s=+1/2` (i) n = 3 = principal quantum number (ii) d orbital has l=2 (iii) as l=2 values of m =+2,1 ,0,-1 ,-2 `therefore` m=+1 is possiblebut only(A)has l=2 (iv) firstelectronin 3d has spin quantum no =+`1/2` thus n=3, l=2,m=1 and s=`+1/3` |
|
| 18. |
The correct sequence which shows decreasing order of the ionic radii of the elements is..... |
|
Answer» `Na^(+) gt F^(-) gt Mg^(+2) gt O^(-) gt AL^(+3)`  "All the ions are isoelectronic having different atomic numbers and the isoelectronic ion of smaller nuclear CHARGE has larger ionic SIZE." |
|
| 19. |
Identify the correct order of innic radii of the following ions |
|
Answer» `Na^(+)gtMg^(2+)gtAt^(3+)gtO^(2-)GTF^(-)` |
|
| 20. |
The correct sequence of the oxidation state of underlined elements is Na_(2)[ul(Fe)(CN)_(5)NO],K_(2)ul(Ta)F_(7),Mg_(2)ul(P)_(2)O_(7),Na_(2)ul(S)_(4)O_(6),ul(N)_(3)H |
|
Answer» <P>`+3, +5, +5, +2.5, -(1)/(3)` `K_(2)ul(Ta)F_(7):+2+x+(-7)=0rArr x=+5` `Mg_(2) ul(P)_(2)O_(7):+4+2x+(-14)=0 RARR x=+5` `Na_(2)ul(S)_(4)O_(6):+2+4x+(-12)=0` or `4x=10 rArr x=+2.5` `ul(N)_(3)H:3x+1=0 rArr x=-(1)/(3)` |
|
| 21. |
The correctsequence whichshowsdecreasingorder of the ionicradii ofthe elementis |
|
Answer» `O^(2-)GT F^(-)gt Na^(+) gt Mg^(2+) gt A1^(3+)` |
|
| 22. |
The correct sequence which shows decreasing order of the ionic radii of the elements is |
|
Answer» `NA^(+) gt MG^(2+) gt AL^(3+) gt O^(2-) gt F^(-)` |
|
| 23. |
The correct sequence of reagents from those listed below for the following conversion is I. NaNH_(2) II. Br_(2) III. H_(2)//Pd-C,quinoloneIV. H_(3)O^(+) |
|
Answer» IV-I-III |
|
| 24. |
The correct sequence of reagents for the following conversion will be : |
|
Answer» `CH_(3)MGBR, H^(+)//CH_(3)OH, [AG(NH_(3))_(2)]^(+)OH^(-)` |
|
| 25. |
The correct sequence of reactions to convert p-nitrophenol into quinol involves............ |
|
Answer» REDUCTION, DIAZOTIZATION and hydrolysis |
|
| 26. |
The correct sequence of decrease in the bond angles of the following hybrides is |
|
Answer» `NH_(3) gt PH_(3) gt AsH_(3) gt SbH_(3)` the central atom decreasesand the size INCREASES, the bond angle decreases. |
|
| 27. |
The correct sequence of deceasing mass of the following is : (i) 6.022 xx 10^(23) atoms of oxygen (ii) 1.0 xx 10^(23) molecules of H_(2)S (iii) 6.022 xx 10^(23) molecules of oxygen |
|
Answer» (i) `GT` (ii) `gt` (iii) (ii) `6.022xx 10^(23)` molecules of `H_(2)S` have mass = 34.0 g `1.0 xx 10^(23)` molecules of `H_(2)S` have mass `= (1xx10^(23))/(6.022xx10^(23))xx(34.0g)=5.645g` (iii) Mass of `6.022xx10^(23)` molecules of oxygen `(O_(2)) = 32 g`. |
|
| 28. |
The correct Schordinger.s wave equation for a electron with total energy E and potential energy V is not correctly given by |
|
Answer» `(DELTA^2 Psi)/(deltax^2) +(delta^2 Psi)/(delta z^2) + (delta^2 Psi)/(delta z^2) + (8 pi^2)/(mh^2) (E-V) Psi = 0` |
|
| 29. |
The correct representation of 4-hydroxy-2-methylpent-2-en-1-al is |
|
Answer» `CH_(3)-underset(OH)underset(|)(C)H-CH=underset(CH_(3))underset(|)(C)-CHO` |
|
| 30. |
The correct representation for solubility product of SnS_(2) , is |
|
Answer» `[SN^(4+)] [S^(2-) ]^(2) ` |
|
| 31. |
The correct relationship between free energy change in a reaction and the coresponding equilibrium constant K is : |
|
Answer» `-DELTAG^(CIRC)`=RTInK |
|
| 32. |
The correct relationship between enthalpy (in cal mol^(-1)) of vaporisation of liquid and its boiling point is |
|
Answer» `DELTA H_("vap") ~~ 21 T_(b)` |
|
| 33. |
The correct relationship betw een quantum numbers n, I and m is : |
|
Answer» `n lt l lt m` 
|
|
| 34. |
The correct relation among the following is |
|
Answer» `x=y` |
|
| 35. |
The correct reactivity order of halogen with hydrogen is ? |
| Answer» SOLUTION :`F_2 GT Cl_2 gt Br_2 gt I_2` | |
| 36. |
The correct proportion of Ca_(3)SiO_(3) is portland cement is …………. . |
|
Answer» 0.26 |
|
| 37. |
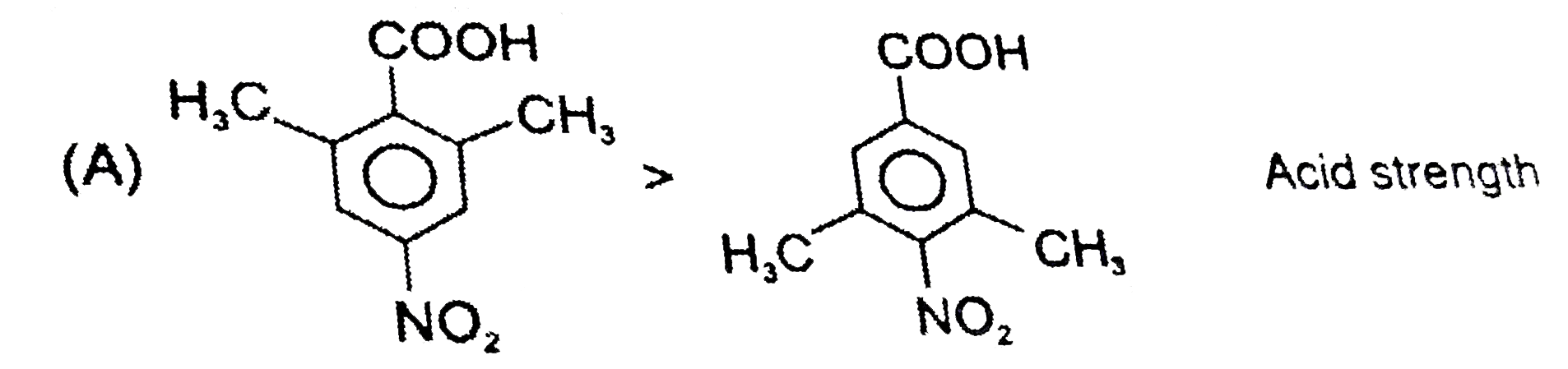
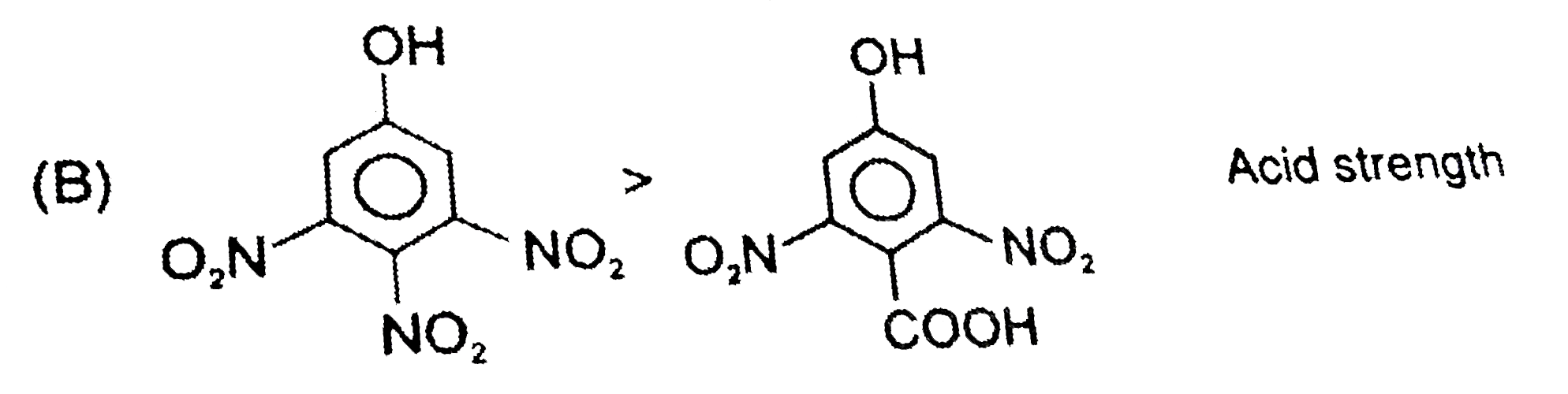
The correct pK_(a) order of the following acids is : |
|
Answer» `I gt II gt III` |
|
| 39. |
The correct orders are: |
|
Answer»
|
|
| 40. |
The correct order to the packing efficiency in different types of unit cells is _______ |
|
Answer» FCC LT bcc lt SIMPLE CUBIC |
|
| 41. |
The correct order to the packing efficiency in different types of unit cells is …. |
|
Answer» fcc LT bcc lt simple cubic |
|
| 42. |
The correctorder regarding the electrongetiveively of hybrid orbitals of carbon is |
|
Answer» ` SP LTSP^(2) GT sp^(3)` |
|
| 43. |
The correctorderregardingthe electronegativityof hybrid orbitalsof carbonis |
|
Answer» `sp LTSP^(2)GT sp^(3)` |
|
| 44. |
The correct order regarding the electronega tivity of hybrid obritals of carbon is |
|
Answer» `sp GT sp^(2) lt sp^(3)` |
|
| 45. |
The correct order of Vander Waals radius of F, Cl and Br is : |
| Answer» Answer :B | |
| 46. |
Arrange in the increasing order of oxidation state of nitrogen for following nitrogen oxides N_(2)O, NO_(2), NO, N_(2)O_(3) |
|
Answer» `NO_(2)ltNOltN_(2)O_(3)ltN_(2)O` |
|
| 47. |
The correct order of the energy of molecular orbitals in a molecules having four electrons |
|
Answer» `SIGMA^(*)_(2s) GT sigma_(2PZ) gt pi_(2px)` |
|
| 48. |
The correct order of the acidic nature of oxides is in the order |
|
Answer» `NOltN_(2)OltN_(2)O_(3)ltNO_(2)ltN_(2)O_(5)` `OVERSET(+1)(N_(2)O)ltoverset(+2)(NO)ltoverset(+3)(N_(2)O_(3))ltoverset(+4)(NO)ltoverset(+5)(N_(2)O_(5))` |
|
| 49. |
The correct order of stability of hydrides of alkali metals is |
|
Answer» `LiHgtNaHgtKHgtRbH` |
|