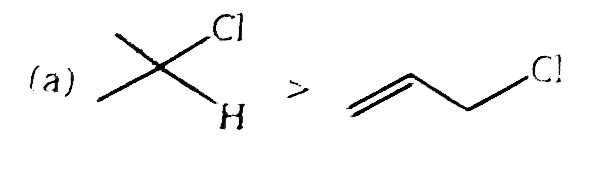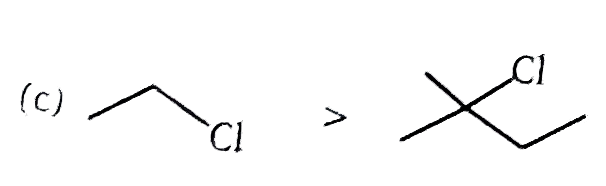Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The correct order of solubility is |
|
Answer» `CaCO_(3)ltKHCO_(3)ltNaHCO_(3)` So the order of SOLUBLITY `CaCO_(3) lt NaHCO_(3) lt KHCO_(3)` |
|
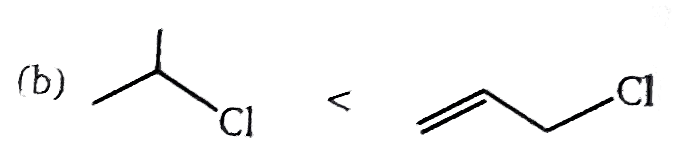
| 2. |
The correct order of S_N2//E2 ratio of the % yield of product of the following halide is : (P)CH_3-CH_2-undersetunderset(I)(|)oversetoverset(Ph)(|)C-CH_3 (Q)CH_3-undersetunderset(Ph)(|)CH-undersetunderset(I)(|)CH-CH_3 (R)CH_3-CH_2-I (S)CH_3-CH-undersetunderset(I)(|)CH-CH_3 |
|
Answer» R gt S gt Q gt P |
|
| 3. |
The decreasing order of second ionisation potential of K, Ca, Ba is |
|
Answer» `K gt CA gt Ba` |
|
| 4. |
The correct order of reducing character of alkali metals is |
|
Answer» `Rb LT K lt Na lt Li` |
|
| 5. |
The correct order of reactivity towards the electophilic substitution of the compounds aniline (I), benzene (II) and nitrobenzene (III)is |
|
Answer» `III GT II gt I` |
|
| 6. |
The correct order of reactivity towards electrophilic substitution is |
|
Answer» benzenegtphenolgtbenzoic acidgtchlorobenzen 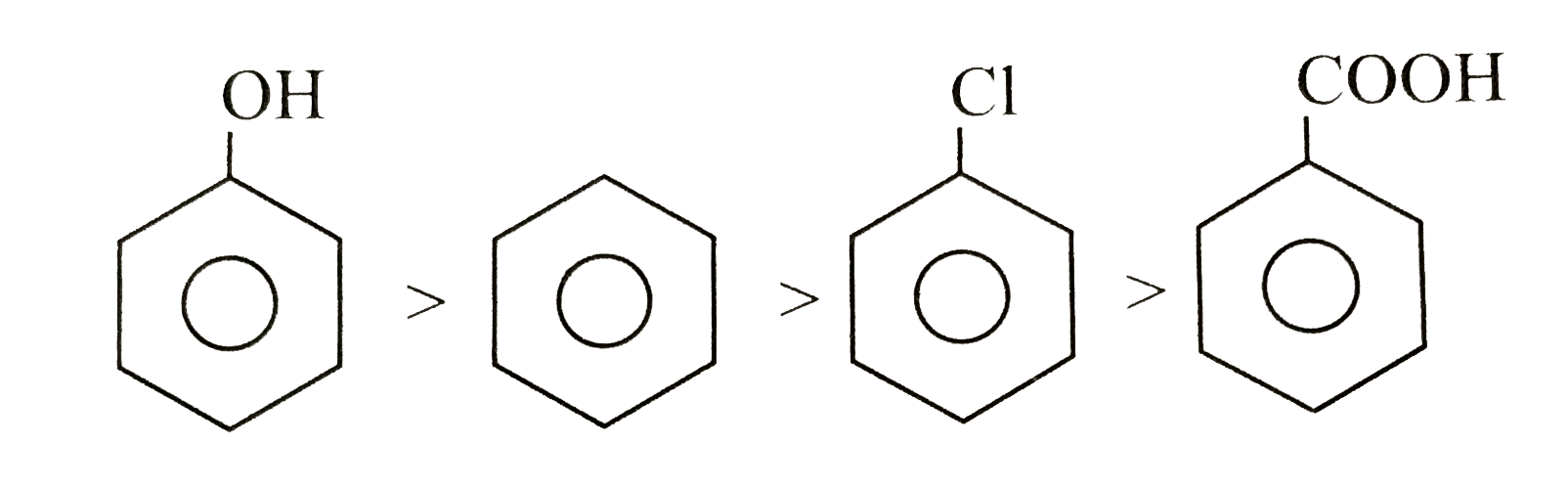
|
|
| 7. |
The correct order of reactivity of functional group is……. |
|
Answer» `= CO gt - OH gt - C -= N gt - OR` |
|
| 8. |
The correct order of reactivity of alkene towards an electrophile is mentioned in : |
|
Answer» `CH_2=CH-Cl GT CH_2 = CH -OCH_3` |
|
| 10. |
The correct order of radius of given ions is : |
|
Answer» `O^(2) gt F^(-) , S^(2-) , CL^(-)`  ` :. ` Ionic radii of : `(S^(2-) " and " Cl^(-)) gt (O^(2-) , F^-))`…..(i) As negative CHARGE is more , the ion has LARGER size. ` :. ` Ionic radius : `S^(2-) gt Cl^(-) " and " O^(2-) gt F^(-)` ….(ii) From (i) and (ii) results, it is easy to write the order of ionic radius as : `S^(2-) gt Cl^(-) gt O^(2-) gt F^(-)` |
|
| 11. |
The correct order of radii is..... |
|
Answer» `N lt Be lt B ` |
|
| 12. |
The correct order of radii is |
|
Answer» `Al^(3+) gt Mg^(2+) gt NA^(+) gt F^(-) gt O^(2-)` |
|
| 13. |
The correct order of priority of the functional groups of organic compounds in the IUPAC system of nomenclature is…… |
|
Answer» `-COOH gt -CHO gt -CN gt -OH gt -OR` |
|
| 14. |
The correct order of polarising ability of cations of Alkaline earth metals is |
|
Answer» `CL^(-) gt BR^(-) gt I^(-) gt F^(-)` |
|
| 15. |
The correct order of O-O bond length in hyrdrogen peroxide, ozone and oxygen is |
|
Answer» `H_(2)O_(2)gt O_(3)gt O_(2)` |
|
| 16. |
The correct order of N compounds in its decresing order of oxidaton states is |
|
Answer» `HNO_(3),NO,NH_(4)CI,N_(2)` |
|
| 17. |
The correct order of N -O bond length in NO, NO_(2)^(-), NO_(3)^(-) and N_2O_4will be |
|
Answer» `NO GT N_2 O_4 gt NO_2gt NO_(3)^(-)` |
|
| 18. |
The correct order of N-compounds in its decreasing order of oxidation states is |
|
Answer» `HNO_(3),NO,N_(2),NH_(4)Cl` |
|
| 19. |
The correct order of mobility of the alkali metal ions in aqueous solution is |
|
Answer» `Rb^(+)GTK^(+)gtNa^(+)gtLi^(+)` |
|
| 20. |
The correct order of melting points of IIIA group elements is |
|
Answer» ` B GT AL gt Tl gt In gt Ga` |
|
| 21. |
The correct order of melting points is |
|
Answer» ETHANE `LT` PROPANE `lt` BUTANE |
|
| 22. |
The correct order of mathching of the following compounds is: |
|
Answer» A-iii,B-I,C -II,D-v,E-iv |
|
| 23. |
The correct order of magnitude of bond angles among the compounds CH_(4),NH_(3) and H_(2)O is |
|
Answer» `H_2 O LT NH_3 ltCH_4` |
|
| 24. |
The correct order of liquefaction of the gases NH_(3),CO_(2),SO_(2) and HCl is |
|
Answer» `NH_(3) GT CO_(2) gt HCl gt SO_(2)` `{:(,SO_(2),gt,NH_(3),gt,HCl,gt,CO_(2)),("Critical TEMP".(K),430,,405,,325,,304):}` |
|
| 25. |
Thecorrectorder of IstionizationenthalpyamongfollowingelementsBe, B,C, N, O is |
|
Answer» `B lt Be lt C lt O N` |
|
| 26. |
The correct order of IP_2 , values among F, Na, Mg and Al is |
|
Answer» `MG gt Al gt F gt NA` |
|
| 27. |
The correct order of ionisation energy of C, N, O and F is |
|
Answer» `F LT N lt C lt O` |
|
| 28. |
The correct order of increasing strength of the compounds (a) CH_(3)CO_(2)H (b) MeOCH_(2)CO_(2)H (c) CF_(3)CO_(2)H (d) |
|
Answer» `b lt d lt a lt c` |
|
| 29. |
The correct order of increasing thermal stability of K_(2)CO_(3) , MgCO_(3) , CaCO_(3)and BeCO_(3) is |
|
Answer» `K_(2)CO_(3) lt MgCO_(3) lt CaCO_(3) lt BeCO_(3)` |
|
| 30. |
The correct order of increasing solubility of AgCl in (a) water (b) 0.1M NaCl (c) 0.1 BaCl_(2) (d) 0.1M NH_(3) is |
|
Answer» `d GT a gt B gt C` |
|
| 31. |
The correct order of increasing rate of hydrolysis of the following compund is |
|
Answer» `I LT II lt III` |
|
| 32. |
The correct order of increasing polarising power of the cations in the following AlCl_(3) , MgCl_(3) , NaCl is |
|
Answer» `AlCl_(3) lt MgCl_(3) lt NaCl` `overset(+1)(Na)Cl lt overset(+2)(Mg) Cl_(2) lt overset(3+)(Al) Cl_(3)` Thus , option (c) is correct . |
|
| 33. |
The correct order of increasing polarising power of the cations in AlCl_(3), MgCl_(2) and NaCl is |
|
Answer» `AlCl_(3) lt MgCl_(2) lt NACL` increasing charge `{:(""(+1),(+2),(+3) ),(NaCl lt ,MgCl_(2), ltAlCl_(3)):}` |
|
| 34. |
The correct order of increasing ionic character is..... |
|
Answer» `BeCl_(2) LT MgCl_(2) lt CaCl_(2) lt BaCl_(2)` |
|
| 35. |
According to Fajans'rules, the percentage of covalent character in an ionic compound increase if the cation is highly charged or small in size and the anion is large or cation has pseudoinert gas configuration. As a result of the increasedcovalent character, solubility in less polar solvent increases and the melting point decreases. The correct order of increasing ionic character is |
|
Answer» `Be Cl _(2) lt Mg Cl_(2) lt CaCl_(2) lt BaCl_(2)` |
|
| 36. |
The correct order of increasing [H_(3)O^(+)] in the following aqueous solutions is |
|
Answer» `0.01 M H_(2) S lt 0.01 M H_(2) SO_(4) lt 0.01 M NACL lt 0.01 M NaNO_(2)` |
|
| 37. |
The correct order of increasing [H_(3) O^(+)] in the aqueous solution is |
|
Answer» `0.01 M H_(2) S LT 0.01 M H_(2) SO_(4) lt 0.01 M NaCl lt 0.01 M NaNO_(2)` |
|
| 38. |
The correct order of increasing energy of atomic orbitals is |
|
Answer» 5P lt 4f lt 6s lt 5d 1s lt 2s lt 2p lt 3s lt 3p lt 4s lt 3D lt 3p lt 5s lt 4d lt 5p lt 6slt 4f lt 5d lt 6p lt 7s. |
|
| 39. |
The correct order of increasing C-O bond length of CO, CO_(3)^(2-) , CO_(2) is |
|
Answer» `CO_(3)^(2-) lt CO_(2)lt CO`  Thus, `CO_(2)` CONTAINS bond , `CO_(2)` contains DOUBLE bond while `CO_(3)^(2-)` contains between singleltbegt and double bond . Hence , C-Obond LENGTHS will be in the order : `CO lt CO_(2) lt CO_(3)^(2-)` |
|
| 40. |
The correct order of increasing bond length of C-H, C-O, C-C and C= C is……… |
|
Answer» `C- H LT C-O lt C-C lt C= C` |
|
| 41. |
The correct order of increasing bond angles in the following triatomic species is : |
|
Answer» `NO_(2)^(+) LT NO_(2) lt NO_(2)^(-) ` |
|
| 42. |
The correct order of increasing bond angles in the following species are : |
|
Answer» `Cl_2O lt ClO_2 lt ClO_2^-`  In `ClO_2O^-` there are 2 lone pairs of ELECTRONS present on the central chlorine atom. Therefore the bond angle in `ClO_2^-` is less than `118^@` which is the bond angle in `ClO_2` which has less NUMBER of electrons on chlorine. |
|
| 43. |
The correct order of increasing boiling points of the following compounds is |
|
Answer» `I LT II lt III lt IV` `underset(underset(underset("boiling")("hydrogen"))("Absencce of"))(CH_(3) COCl) lt CH_(3) CO_(2)CH_(3) lt underset(underset(underset("and more surface area")("molecular mass"))("Higher"))((CH_(3)CO)_(2)O) lt underset(underset(underset("hydrogen boding")("INTERMOLECULAR"))("EXTENSIVE"))(CH_(3)CONH_(2))` |
|
| 44. |
The correct order of increasing basicity of the given conjugate bases (R=CH_(3)) is |
|
Answer» `CRCOO^(-) lt NH_(2)^(-) lt HC -= C^(-) lt R^(-)` `RCOO^(-)ltHC-=C^(-)ltNH_(2)^(-)LTR^(-)` (Remember that `sp^(3)` C is less electronegative than `sp^(3)` N. Hence, alkyl carbanion `(R^(-))` is more basic than `bar(N)H_(2)`. However, sp-hybridized carbon is more electronegative than `sp^(3)` N Hence `bar(N)H_(2)` is more basic `HC-=bar(C)`. |
|
| 45. |
The correct order of increasing basicity of the given conjugate bases (R=CH_3) is |
|
Answer» `RCOO^(-) LT HC -=C^(-) lt NH_2^(-)lt R^(-)` |
|
| 46. |
The correct order of increasing basicity of the given conjugate bases (R=CH_(3)) is : |
|
Answer» `RCObarO LT HC -=BARC lt barNH_(2)` |
|
| 47. |
The correct order of increasing acidic strength of the following acids is |
|
Answer» `I LT II lt III` |
|
| 48. |
The correct order of increasing acid strength of the compounds. (a) CH_(3)CO_(2)H""(b) MeOCH_(2)CO_(2)H""(c)CF_(3)CO_(2)H "" (d) |
|
Answer» `B LT d lt a lt C` |
|
| 49. |
The correct order of hydration enthalpies of alkali metal ions is: |
|
Answer» `LI^(+) GT NA^(+) gt K^(+) gt RB^(+) gt Cs^(+)` |
|