Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The correct order of electron gain enthalpy with negative sign of F.Cl.Br and I, having atomic 9,17,35, and 53 respectively, is : |
|
Answer» `IgtBrgtClgtF` |
|
| 2. |
The correctorder of electronegativitiesof N, O , Fand P is |
|
Answer» `F gt N gt P gt O` |
|
| 3. |
The correct order of electron gain enthalpy with negative sign of F, CI, Br and I having atomic number 9, 17, 35 and 53, respectively is ............. |
|
Answer» `I GT BR gt CL gt F` |
|
| 4. |
The correctorder ofelectron gain enthalpywithnegativesign of F, C1 , Br andI , havingatomicnumber 9, 17, 35 and 53 respectivelyis |
|
Answer» `I GT BR gt C1 gt F` Thuscorrectorder ofelectrongain enthalpy(in kJ `mol^(-1))`with negativesigh followsthe ORDER: `C1 (-349 ) gt F (-328) gt Br (-325) gt (- 295)` i.e.,option ( c ) ISCORRECT. |
|
| 5. |
The correct order of electron gain enthalphy with negative sigh of F, Cl , Br and I having atomic number 9,17 ,35 and 53 respectively is |
|
Answer» `IgtBrgtCIF` |
|
| 6. |
The correct order of electron affinity of the elements of oxygen family in the periodic table is |
|
Answer» O GT S gt SE |
|
| 7. |
The correct order of dielectric constant values |
|
Answer» `H_2O GT D_2O gt H_2O_2` |
|
| 8. |
The correct order of decreasing second ionisation energy of Li, Be, Ne,C,B |
|
Answer» `Ne GT B gt LI gt C gt Be` |
|
| 9. |
The correct order of decreasing second ionization enthalpy of Ti(22), V(23), Cr (24) and Mn(25) is |
|
Answer» `Cr lt MN GT VGT Ti ` |
|
| 10. |
The correctorderof decreasingionicradiiamongthe followingisoelectronicspeciesis |
|
Answer» `K^(+) LTCA^(2+) gt C1^(-) gt S^(2-)` |
|
| 11. |
The correct order of decreasing ionic radii among the following isoelectronic species is |
|
Answer» `CA^(+2) GT K^(+) gt S^(-2) gtCl^(-)` |
|
| 12. |
The correct order of decreasing H-C-H angle in the following molecule is: |
|
Answer» `I GT II gt III` |
|
| 13. |
The correct order of decreasing first ionisation energy is..... |
|
Answer» `C GT B gt Be gt LI` |
|
| 14. |
The correct order of decreasing electronegativity values among the elements X,Y,Z and A with atomic numbers 4,8,7 and 12 respectively |
|
Answer» |
|
| 15. |
The correct order of decreasing acidic nature of H_(2)O, ROH, CH -= CH and NH_(3)is |
|
Answer» `CH-=Ch gt H_(2) O gt ROH gt NH_(3)` Smaller the `pK_(a)` value, stonger is the ACID. Hence, decreasing order of acidic NATURE is `H_(2)O gt ROH gt CH-=CH gt NH_(3)`. |
|
| 16. |
The correct order of C - 0 bond length among CO , CO_(3)^(2-)and CO_(2) is |
|
Answer» `CO lt CO_(3)^(2-) lt CO_(2)` All these structures exhibits RESONANCE and can be represented by the FOLLOWING resonating structures More is the single bond character. More will be the bond length. HENCE, the correct order is : CO`lt CO_(2) lt CO_(3)^(2-)` 
|
|
| 17. |
The correct orderof bond order values among the following : A . NO^(-) B.NO^(+) C.NOD.NO^(2+) E. NO^(2-) is |
|
Answer» `A lt D lt C lt B lt E` B.O. `= (1)/(2) (8-3) = (5)/(2) = 2.5 ` `NO^(-)=KK sigma_(2)^(2) sigma_(2s)^(**2) sigma_(2p_(z))^(2) pi_(2p_(x))^(2)pi_(2p_(y))^(2)pi_(2p_(x))^(**1)pi_(2p_(y))^(**1)` B.O. = `(1)/(2) (8-4)= 2` `NO^(2-)=KK sigma_(2)^(2) sigma_(2s)^(**2) sigma_(2p_(z))^(2) pi_(2p_(x))^(2)pi_(2p_(y))^(2)pi_(2p_(x))^(**2)pi_(2p_(y))^(**1)` B.O. = `(1)/(2) (8-5) = (3)/(2) = 1.5 ` `NO^(+)=KK sigma_(2)^(2) sigma_(2s)^(**2) sigma_(2p_(z))^(2) pi_(2p_(x))^(2)pi_(2p_(x))^(2)pi_(2p_(y))^(2)` B.O. `= (1)/(2) (8-2) = 3 ` `NO^(2+)=KK sigma_(2)^(2) sigma_(2s)^(**2) sigma_(2p_(z))^(2) pi_(2p_(x))^(2)pi_(2p_(y))^(1)` B.O. ` = (1)/(2) (7-2) = (5)/(2) = 2.5 ` THUS `NO^(2-) lt NO^(-) NO = NO^(2-) lt NO^(+)`, i.e. `E lt A lt C = D lt B ` |
|
| 18. |
The correct order of bond lengths is |
|
Answer» `H-CI gtH-BR gtH-I` |
|
| 19. |
The correct order of Boiling points is |
|
Answer» `H_2Ogt H_2O_2 gt D_2O` `H_2O_2 gt D_2O gt H_2O` |
|
| 20. |
The correct order of boiling point of 2,2-dimethyl-propane , 2-methylbutane and n-pentane is |
|
Answer» n-pentane GT 2,2-dimethylpropane gt 2-methyl-butane |
|
| 21. |
The correct order of boiling point order for corresponding hydrocarbons is : |
|
Answer» `"alkyne"gt"ALKANE" gt"ALKENE"` |
|
| 22. |
The correct order of boiling point for primary (1^(@)), secondary (2^(@)) and tertiary (3^(@)) alcohols is |
|
Answer» `1^(@) gt 2^(@) gt 3^(@)` |
|
| 23. |
The correct order of basicity of the following species is |
|
Answer» `III lt IV lt II lt I` |
|
| 24. |
The correct order of atomic radii is.... |
|
Answer» `NA lt MG lt Al lt SI` |
|
| 25. |
The correct order of atomic radius of Li, Be and B is |
|
Answer» B GT Be gt LI |
|
| 26. |
The correct order of atomic radii is: |
|
Answer» EugtCegtHogtN |
|
| 27. |
The correct order of atomicradiiin group 13elements is |
|
Answer» `B LT Al lt INLT GA lt TL` |
|
| 28. |
The correct order of atomic radii in group 13 elements is |
|
Answer» B LT GA lt Al lt In lt TI 
|
|
| 29. |
The correct order of atom orbitals in terms of energy between 8s and 8 p ordital are . |
|
Answer» 7D,6f `8P RARR 8+1=9` ` 5g rarr 5+4=9` `6f rarr 6+3=9` ` 7d rarr 7+2=9` CORRECT order `rarr 8s 5g 6f 7d 8p` |
|
| 30. |
The correct order of acidity of the C-H proton is- |
|
Answer» ACETYLENE `GT` ETHYLENE `gt` ETHANE |
|
| 31. |
The correct order of acidity for the following is |
|
Answer» `HCN gt ClCH_(2)CO OH gt HCO OH gt CH_(3)CO OH` |
|
| 32. |
The correct order of acid strength of the following substituted phenols in water at 28^(@)C is |
|
Answer» p-nitrophenols `LT` p-fluorophenol `lt` p-chlorophenol |
|
| 33. |
The correct order of acid strength of the following compounds: |
|
Answer» Phenol |
|
| 34. |
The correct order in which the O-O bond length increases in the respectively are |
|
Answer» `O_(3) lt H_(2)O_(2) lt O_(2)` `O_(2)(2) lt O_(3)(1.5) lt H_(2)O_(2) (1)` |
|
| 35. |
The correct order in which O-O bond length increases in the following is |
|
Answer» `O_(3) lt H_(2) O_(2) lt O_(2)` `O_(2)(2)` . GREATER the bond order, SHORTER is the bond length . |
|
| 36. |
The correct order for homolytic bond dissociation energis (Delta H in kcal/mol) for CH_(4)(A), C_(2)H_(6)(B) and CH_(3)Br(C ) is…. |
|
Answer» `C GT B gt A` |
|
| 37. |
The correct options (s) to distinguish nitrate salts of Mn^(2+) and Cu^(2+) taken separately is (are) |
|
Answer» `Mn^(2+)` show the CHARACTERISTIC GREEN colour in the flame test `E^(@) Cu^(2)//Cu (+0.34V)` is higher than `E^(@)Mn^(2+)//Mn (-1.18V)` in the electrochemical series. |
|
| 38. |
The correct of increasing bond angles in the following species is |
|
Answer» `CIO_(2)^(-) lt Cl_(2)O lt ClO_(2)` 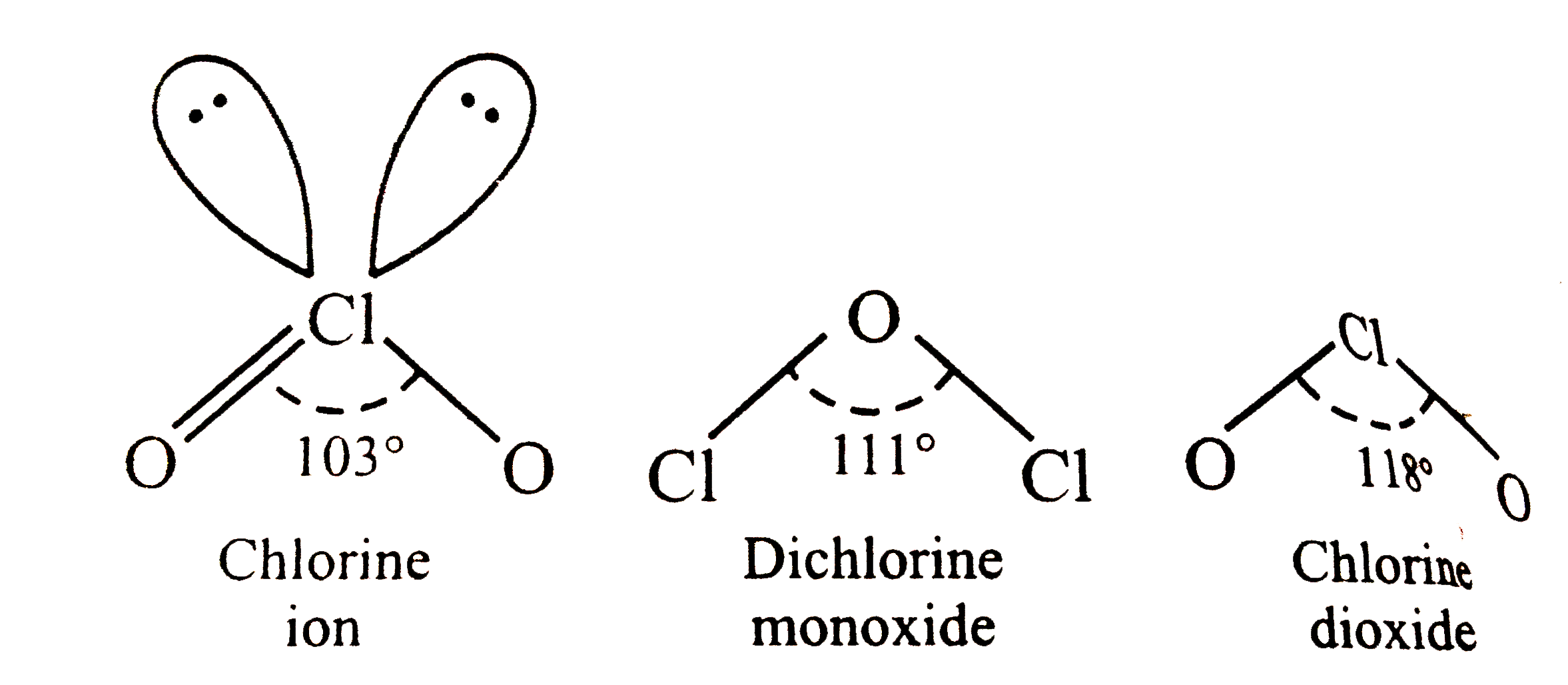 In `ClO_(2), Cl is sp^(2)` -hybridized and the bond ANGLE is `180^(@)` . In `Cl_(2)O,OIS sp^(3)` -hybridized . HOWEVER , dueto repulsions between two BIG sized Cl atoms, the bond angle increases from `109.5^(@)` to `111^(@)` . In `ClO_(2)^(-) ,Cl` ATOM is `sp^(3)`-hybridizedbut due to lp-bp repulsions, the angle decreases from `109.5^(@)` to `103^(@)` .Thus, the increasing order of bond angles is `ClO_(2)^(-) lt Cl_(2)O lt ClO_(2)`. |
|
| 39. |
The order of decrease in atomic radii for Be, Na and Mg is |
|
Answer» `Be gt Mg gt NA` |
|
| 40. |
The correct name of the product obtained is |
|
Answer» cis-1,2-dibromocyclohexane |
|
| 41. |
The correct name of CH_(3)CH_(2)-underset(O)underset(||)(C)-underset(CN)underset(|)(C)H-CHO is |
|
Answer» 2-cyano-3-oxopentanal |
|
| 42. |
The correct match of contents in column-I with those in column-II is ....... |
|
Answer» `1 -C, 2-a, 3-B, 4-d` |
|
| 43. |
The correct match is |
|
Answer» `{:(A,B,C,D),(2,5,4,1):}` |
|
| 44. |
The correct match is |
|
Answer» `{:(A,B,C,D),(2,1,3,4):}` |
|
| 45. |
The correct match is |
|
Answer» `{:(A,B,C,D),(1,5,3,2):}` |
|
| 46. |
The correct match is |
|
Answer» `{:(A,B,C,D),(4,2,3,1):}` |
|
| 47. |
The correct match is |
|
Answer» `{:(A,B,C,D),(4,5,1,2):}` |
|
| 48. |
The correct match is |
|
Answer» `{:(A,B,C),(2,1,3):}` `rArr Delta G LT 0` It is at EQUILIBRIUM with ICE at `0^(@)C rArr Delta G = 0` at `T gt 10^(@)C`, it doesnot freeze `rArr Delta G = 0` |
|
| 49. |
The correct match is |
|
Answer» `{:(A,B,C,D),(1,2,3,4):}` SA + SB = `-57.3` `CH_(3)COOH + NaOH` is WA + SB `CH_(3)COOH + NH_(4)OH` is WA `+ SB RARR` least |
|