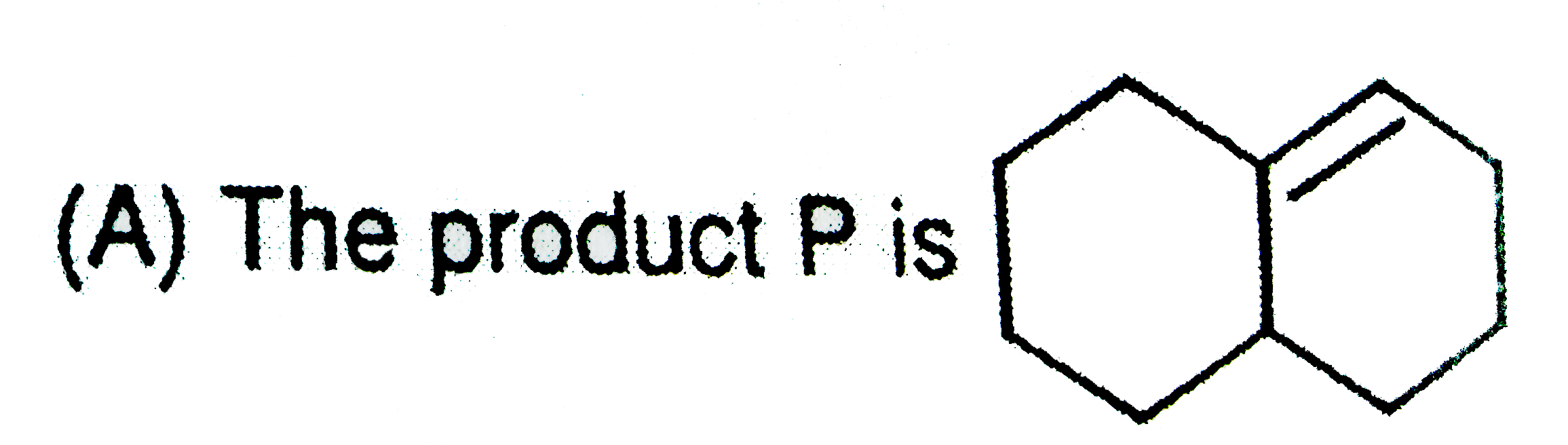Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The correct statements regarding green chemistry |
|
Answer» It is a cost effective approach that involves minimum chemical USAGE, minimum WASTE GENERATION |
|
| 2. |
The correct statement(s) regarding defects in solids is (are) |
|
Answer» Frenkel defect in usually favoured by a very small difference in the sizes of cation and anion. The ELECTRONS trapped in anion vacancies are referred to as F - centers. Schottky defects asrise when some atoms ot IONS are missing from their normal lattice points. Due to the presence of large number of vacancies in crystals, its density (i.e., physical property) is lowered. |
|
| 3. |
The correct statements is / are : (i) BeCl_2 is a covalent compound (ii) BeCl_2 Can form dimer (iii) BeCl_2 is an electron deficient molecule (iv) The hybridisation of Be in BeCl_2 is sp^2 |
|
Answer» `(i) and (III) |
|
| 4. |
The correct statements is are : |
|
Answer» `BeCI_(2)` is a covalent COMPOUND C) `BeCI_(2)` is electron deficient |
|
| 5. |
The correct statements for the following addition reactions is (are) |
|
Answer» <P>(M and O) and (N and P) are two pairs of enantiomers |
|
| 6. |
The correct statement(s) for orthoboric acid is/are |
|
Answer» it behaves as a weak acid in water due to self ionization  On adding ethylene glycol, it acidity increases  `H_(3)BO_(3)`, however, does notundergo self ionization to releasea proton.  Further due to H-bonding , `H_(3)PO_(3)` is a planarmolecule .Thus only OPTION (b,d) correct. |
|
| 7. |
The CORRECT statement(s) for cubic close packed (ccp) three- dimensional structure is (are) |
|
Answer» The number of neighbours of an atom PRESENT in the topmost layer is 12. (b) is a known FACT. is correct because in ccp (fcc), number of atoms per unit cell is 4. HENCE, octahedral voids =4 and tetrahedral voids =8. therefore , number of octahdral voids per atom =1 and number of tetrahdral voids per atom =2 (d)For ccp (fcc), ` r= a/(2sqrt2) or a = 2sqrt2 r ` |
|
| 8. |
The correct statements are |
|
Answer» The isotopes of chlorine with mass NUMBER 35 and 37 EXIST in ratio `3:21` if its average atomic mass is 35.5 d) Molarity = `("No. of molecules of solute")/(6.023xx10^(23))xx(1000)/("volume of solution in ml")` Molarity = `(6.023xx10^(21))/(6.023xx10^(23))xx(1000)/(10)=0.1M` |
|
| 9. |
The correct statement(s) among the following is/are: |
|
Answer» All d-orbitals except `d_(Z^(2))` have two ANGULAR NODES. |
|
| 10. |
The correct statement(s) among the following is (are) |
|
Answer» first IONIZATION POTENTIAL of Al is less than the first ionization potential of MG |
|
| 11. |
The correct statements among I to III regarding group 13 element for oxides are: (I) Boron trioxide is acidic (II) Oxides of aluminium and gallium are amphoteric. (III) Oxides of indium and thallium are basic. |
|
Answer» (I) and (II) only . |
|
| 12. |
The correct statements about 'X' gas is: |
|
Answer» It is recognized by its PUNGENT ORDER and production of white fumes, on blowing across the mouth of the test tube. |
|
| 13. |
The correct statements about the following reaction are [Hint: Al_2O_3 gives satyzeff's product without any rearrangement] |
|
Answer»

|
|
| 14. |
The correct statement(s) about the flow diagram is/are: |
|
Answer» `CH_(3)COONA` may be the salt solution 'X' |
|
| 15. |
The correct statement(s) about above flow diagram is/are: |
|
Answer» ADDING an excess of hyposolution to the brown solution, 'D' is reduced to colourless solution and WHITE precipitate BECOMES visible. |
|
| 16. |
The correct statement with regard to H_(2)^(+) and H_(2)^(-)is |
|
Answer» both`H_(2) ^(+) and H_(2)^(-)` do not EXIST |
|
| 17. |
The correct statement (s) regarding defects in solids is (are): |
|
Answer» Frenkel defect is usually favoured by a very small difference in the sizes of cation and anion |
|
| 18. |
The correctstatement (s) about O_(3)is (are) : |
|
Answer» O-O bond length are EQUAL  (`sp^(2)` hybridization) ltbgt O-O bond lengths are equal due to REASONANCE . It (`sp^(2)` hybridization) ltbgt O-O bond lengths are equal due to REASONANCE . It is DIAMAGNETIC andhas a bent structure. However, thermal decomposition of `O_(3)` is not endothermic but it is exothermic `2O_(3)to3O_(2) +`Heat |
|
| 19. |
The correct statement regarding various types of molecular speeds are |
|
Answer» increasing TEMPERATURE increases the fraction of molecules having `U_(mps)` |
|
| 20. |
The correct statement regarding the comparison of staggered and eclipsed conformations of ethane, is : |
|
Answer» The eclipsed CONFORMATION of ethane is more STABLE than STAGGERED conformation, because eclipsed conformation has no TORSIONAL strain |
|
| 21. |
The correct statement regarding the comparison to staggered and eclipsed conformations of ethane is : |
|
Answer» The eclipsed conformation of ETHANE is more stable than STAGGERED conformation, because eclipsed conformation has no torsional strain. |
|
| 22. |
The correct statement regarding the comparison of stagged and eclipsed conformations of ethane is ………….. . |
|
Answer» the eclipsed CONFORMATION of ETHANE is more stable than staggered conformation EVEN though staggered conformation has torsional strain. |
|
| 23. |
The correct statement regarding the compariso of staggered and eclipsed conformations of ethane is …………………… |
|
Answer» the eclipsed conformation of ETHANEIS more stable than staggered conformation EVEN though the eclipsed conformation has torsional strain |
|
| 24. |
The correct statement regarding structure of ice. (A) Ice has a highly ordered three dimensional hydrogen bonded structure. (B) Each oxygen atom in ice is surrounded tetrahedrally by four other oxygen atoms at a distance of 276 pm. (C) Hydrogen bonding gives ice a rather open structure with wide holed. These holes can hold some other molecules of approapriate size interstitially. |
|
Answer» A and B |
|
| 25. |
The correct statement regarding structure of ice. |
|
Answer» ICE has a HIGHLY ordered three dimensional hydrogen bonded structure. |
|
| 26. |
The correct statement regarding Graphite |
|
Answer» Graphite is not a CONDUCTOR because, it does not contain free ELECTRONS |
|
| 27. |
The correct statement regarding Graphite is |
|
Answer» GRAPHITE is not a conductor because, it does not contain free electrons |
|
| 28. |
The correct statement regarding ethane conformation is….. |
|
Answer» Rotation around carbon-carbon bond in ethane MOLECULE is not POSSIBLE, because ethane molecule contains a pi `(pi)` bond between the carbon and carbon and ethane has very low melting point |
|
| 29. |
The correct statement regarding entropy is |
|
Answer» at absolute zero temperature, entropy of a PERFECTLY CRYSTALLINE SOLID is zero |
|
| 30. |
The correct statement regarding electrophile is |
|
Answer» ELECTROPHILE is a NEGATIVELY charged SPECIES and canform a bond by accepting a pair of electrons from another electrophite |
|
| 31. |
The correct statement regarding defects in crystalline solids is |
|
Answer» Frenkel DEFECT is a dislocation defect |
|
| 32. |
The correct statement regarding defect in solids is (are) |
|
Answer» Frenkel defects are USUALLY favoured by a very small differences in the sizes of the cation and anion |
|
| 33. |
The correct statement regarding a carbonyl compound with a hydrogen atom on its alpha carbon is |
|
Answer» a carbonyl compound with a HYDROGEN atom on its alpha-carbon RAPIDLY equilibrates with its corresponding enol and this process is known as carbonylation |
|
| 34. |
The correct statement is / are |
|
Answer» `BeCI_(2)` is a covalent COMPOUND B) In `BeCI_(2)`Be has insuffcient octet configuration it is electron defficient C) `BeCI_(2)` can from a dimer at below 1200 K |
|
| 35. |
The correct statement is about following compound is- |
|
Answer» |
|
| 36. |
the correct statement is |
|
Answer» `BI_(3)` is the weakestLewis acid AMONG the BORON halides |
|
| 37. |
The correct statement is : |
|
Answer» Hydroxide of aluminimum is more acidic than the hydroxide of boron |
|
| 38. |
The correct statement for the molecule, CsI_(3) is |
|
Answer» It contains `CS^(3+)` and `I^(-)` ions. |
|
| 39. |
The correct statement foralpha -elimination is |
|
Answer» It forms CYCLIC compounds |
|
| 40. |
The correct statement amongst the following is: |
|
Answer» For every gas there exists a characteristic TEMPERATURE above which it cannot be liquefied no matter however high is the pressure applied called CRITICAL temperature of the gas. c) `CO_2` has highest value of aa d) Z gives measure of deviation. |
|
| 41. |
The CORRECT statement for cubic close packed (ccp) three-dimensional structure is (are) |
|
Answer» The number of neighbours of an atom present in the TOPMOST layer is 12 (b ) is a known FACT. (C ) is correct because in ccp (fcc), number of atoms per unit cell is 4. hence , octahedral voids =4 and tetrahedral voids=8. Therefore , number of octahedral voids per atom =1and number of tetrahedral voids per atom =2. (d)For ccp (fcc), `r=a/(2sqrt2)` or `a=2sqrt2r` |
|
| 42. |
The correct statement among the following (i) heat of reaction depends on the temperature at which the reaction is carried (ii) heat of neutralisation depends on the temperature at which the experiment is carried. (iii) experimentally heat of combustion is DeltaE. |
|
Answer» i only correct |
|
| 43. |
The correct statements among the following are. |
|
Answer» The density of `H_2O_2` is greater than `H_2O` A) The density of `H_2O_2` is greater than `H_2O` C) Boiling point of `H_2O_2` is more than `H_2O` because of more hydrogenbonding in `H_2O_2` D) On cooling water freezes earlier than `H_2O_2` |
|
| 44. |
The correct statement about the following is |
|
Answer» In `H_2` and `D_2`the RATIO of bond length is 1:2 |
|
| 45. |
The correct statement about the compounds (a), (b) and (c) is |
|
Answer» a and B are identical |
|
| 46. |
The correct statement about [C] is: |
|
Answer» Red precipitate is formed due to formation LEAD sulphochloride. |
|
| 47. |
The correct statement about adsorption are |
|
Answer» the chemisorption of `H_(2)` as `H` atoms on the surface of glass is endothermic |
|
| 48. |
The correct stability order of the following speicies is |
|
Answer» cltaltb |
|
| 49. |
The correct stability order of the following resonating structure is : underset("(I)")(H_(2)C=overset(+)N=overset(-)N)""underset("(II)")(H_(2)overset(+)C-N=overset(-)N)""underset("(III)")(H_(2)overset(-)C-overset(+)N-=N)""underset("(IV)")(H_(2)overset(-)C-N=overset(+)N) |
|
Answer» `(I)GT(II)gt(IV)gt(III)` |
|
| 50. |
The correct stability order of the following resonance structures is {:((a) H_(2)C = overset(+)N - N^(-) ,""H(2)overset(+)C-N=N^(-)),( (I), ""(II)),(H_(2) overset(-)C-overset(+)N-=N,H_(2)overset(-)C-N=overset(+)N),((III),""(IV)):} |
|
Answer» `(I) gt (II) gt (IV) gt (III)` (ii) A reaonating structure is more stable is the negative CHARGE is on ELECTRONEGATIVE atom and positive charge on electropositive atom. (iii) Like charge should not be CLOSE and unlike charges should not be WIDELY separated . Keeping above points in view , the stability order will be `(I) gt (III) gt (II) gt (IV)` . |
|