Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Two volatile liquids A and B differ in their boiling points by 15K. The process which can be used to separate them is |
|
Answer» FRACTIONAL DISTILLATION |
|
| 2. |
Which of the following is not cyclic compound? |
|
Answer» Benzene |
|
| 3. |
When 2-butyne is treated with dil. H_2SO_4//HgSO_4, the product formed is |
|
Answer» butanol-1 |
|
| 4. |
Which are the various steps of electrophilic substitution of benzene ? |
|
Answer» Solution :Electrophilic (E) Substitution (S), reactions of benzene are shortly KNOWN as aromatic `S_(E)`. Aromatic `S_(E)` REACTION occurs in three steps : (i) Steps involve in formation of electrophile (ii) Steps involve in formation of intermediate which is a slow STEP. (iii) Step involve in liberation of PROTON from intermediate carbocation (`sigma`-complex) and produce substituted product This step is fast and is not rate determining. |
|
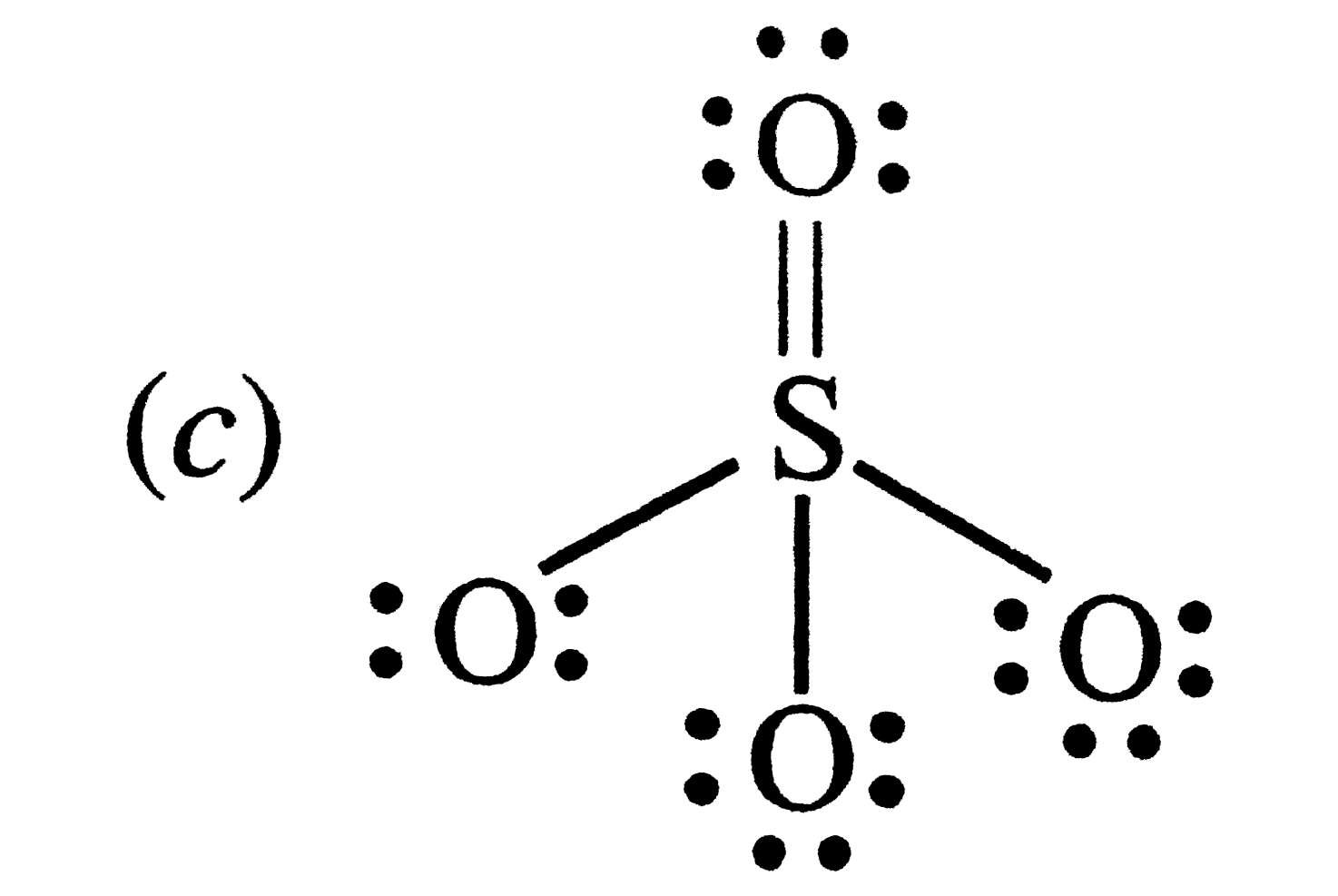
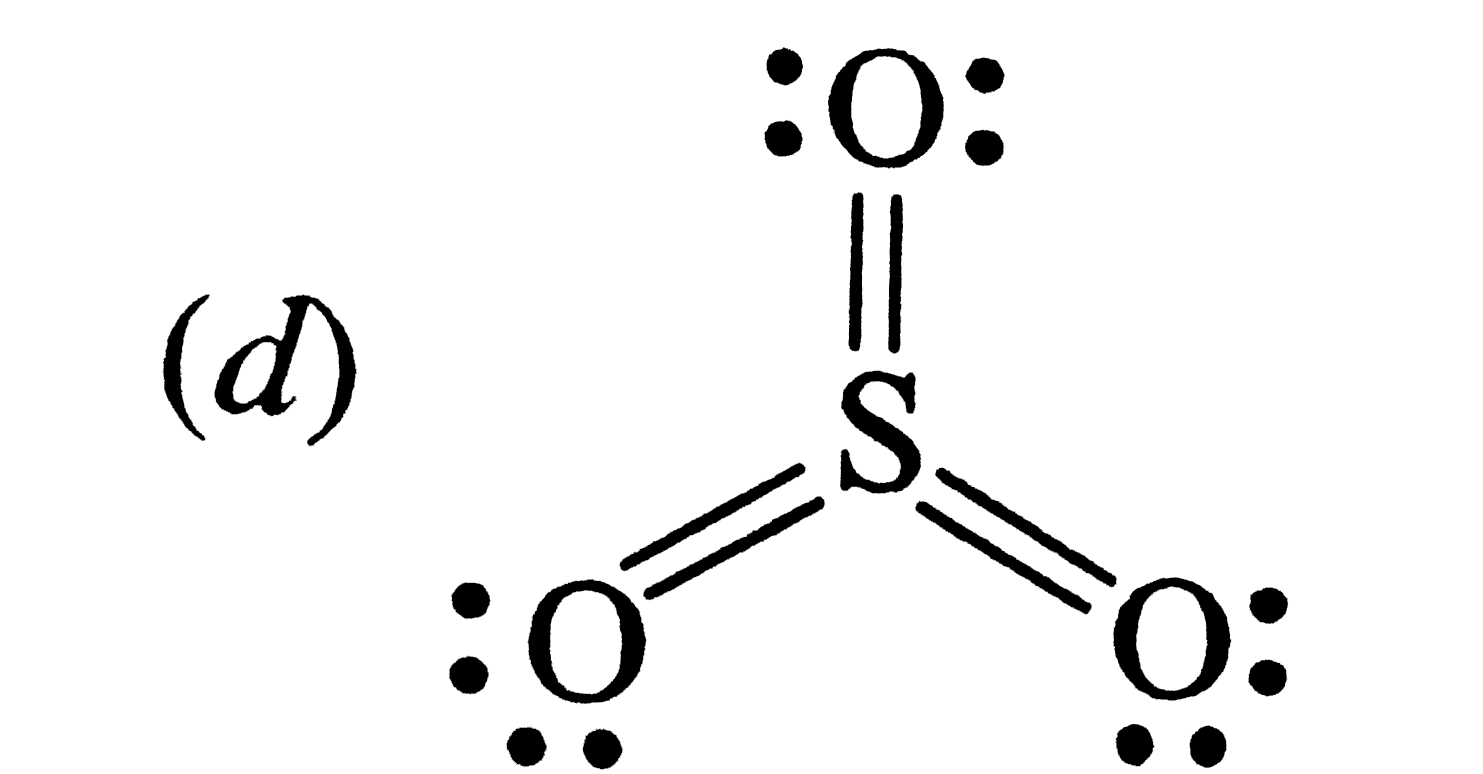
| 5. |
Which of the following structures is the most perferred and hence of lowest energy for SO_(3) ? |
|
Answer»
having the smallest FORMAL charges on ehgt ATOMS. Hence , option (d) is correct . Alternatively , (d) has maximum NUMBER of covalent bonds . |
|
| 6. |
Which of the following has the lowest boiling points? |
|
Answer» 2-Methylbutane |
|
| 7. |
Which coordinated numbered complexes are formed by EDTA with most metal ions in solution |
|
Answer» |
|
| 8. |
Which of the following changes is not realised in the laboratory? |
|
Answer» Absoption OS NO by alkaline sodium SULPHITE to from a compound |
|
| 9. |
Which type of covalent bond is form by hybrid orbitals ? |
| Answer» SOLUTION :SIGMA type COVALENT BOND is formed. | |
| 10. |
Which of the followings is not true about the ionic solids ? |
|
Answer» BIGGER ions form THECLOSE packed STRUCTURE. |
|
| 11. |
which of the following statements are true? |
|
Answer» flocculation value is inversely proportional to the coagulating power. |
|
| 12. |
Which one of the following pairs gives effervescence with aq. NaHCO_(3)? I. CH_(3)COCl II. CH_(3)COCH_(3) III. CH_(3)COOCH_(3) IV. CH_(3)COOCOCH_(3) |
|
Answer» I and II |
|
| 13. |
Which one of the following has only primary hydrogen atom? |
|
Answer» isobutane |
|
| 14. |
The stability off K_(2)O,K_(2)O_(2) and KO_(2) is in order K_(2)O lt K_(2)O_(2) lt KO_(2). This increasing stability as the size of metal ion increases is due to stabilisation of |
|
Answer» larger CATION by smaller anions through lattice ENERGY effects |
|
| 15. |
Which equation gives the quantitative temperature dependence of equilibrium constant ? |
|
Answer» HESS LAW |
|
| 16. |
To get an n-type semiconductor from silicon, it should be doped with a substances with valence _____ |
|
Answer» 2 |
|
| 17. |
Which one is main cause for deforestation? |
|
Answer» URBANIZATION |
|
| 18. |
Which statement(s) is correct, |
|
Answer» <P>`((DELTA H)/(delta T))_(P) - ((delta U)/(delta T))_(V) = R` |
|
| 19. |
What is meant by intramolecular hydrogen bond? Give one example |
|
Answer» Solution :INTRA molecular hydrogen bonds are those which occur within ONE single molecule. This OCCURS between two functional groups within a molecule. Eg :ortho - NITROPHENOL and salicyladehyde. |
|
| 20. |
Which of the following is the weakest base ? |
|
Answer» `Ca(OH)_(2)` |
|
| 22. |
Which effect is observed in Isothermal graph with temperature ? |
|
Answer» SOLUTION :Value of K changes with CHARGE with temperature. SLOPE increases if temperature increases. Slope : `T_(3)GT T_(2)gt T_(1)` where, Temperature : `T_(3)gt T_(2)gt T_(1)` |
|
| 23. |
Which of the following reagents give white precipitate with MgSO_(4) solution, which is readily soluble on addition of ammonium salts? |
|
Answer» `NaOH` |
|
| 24. |
Which of the following cannot be used for detecting formic acid ? |
|
Answer» `NaHCO_(3)` |
|
| 25. |
Which of the following molecules has the electronic configuration ? (sigma 1s)^(2) , (sigma^(**) 1s)^(2) , (sigma 2s)^(2) , (sigma^(**) 2s)^(2) (pi 2p_(x))^(2) = (pi 2p_(y))^(2) , (sigma 2p_(z))^(2) |
|
Answer» CO |
|
| 26. |
Which of the following is correct name according to the IUPAC rules? |
|
Answer» 2-Methylcyclohexane |
|
| 27. |
Which of the following is not an oxo salt of alkali metals? |
|
Answer» `Na_(2)CO_(3)` |
|
| 28. |
The treatment of ethyl formate with excess of RMgX gives …………………….. . |
|
Answer» `R - UNDERSET(O)underset(||)C-R` |
|
| 29. |
Which of the following option is correct for the given equation ? 4NH_(3(g)) + 5O_(2(g)) to 4NO_((g)) + 6H_(2) O_((g)) |
|
Answer» `Delta H = Delta U` |
|
| 30. |
Which statement is incorrect about diastereomers |
|
Answer» They are non-mirror images |
|
| 31. |
What is the rate of reaction with Cl_(2), Br_(2) and I_(2) with CH_(2) -= CH_(2). |
| Answer» Solution :Reactivity order `Cl_(2) gt Br_(2) gtI_(2)`. Rate of REACTION of ALKYNE with iodine `(I_(2))` is so SLOW hence special condition is required to ARRANGE EQUILIBIRUM. | |
| 32. |
What is homologous series ? Give example |
|
Answer» Solution :A group or a series of ORGANIC compounds each forms a HOMOLOGOUS series and the members of the series are called homologous. (i) The members of a homologous series can be represented by general molecular formula. (ii) The successive members differ from each other in molecular formula by a `-CH_(2)` UNIT. (iii) All compounds of homologous series are CONTAIN same functional group. (iv) The compounds of homologous series are MADE by general method and containing game geneal properties. (v) The physical properties homologous series are successively change. 
|
|
| 33. |
Value of R in SI units is |
|
Answer» `8.315 xx 10^7erg K^(-1) " mole"^(-1)` |
|
| 34. |
Using your knowledge of electronegativity, tell whether each of the following bonds will be ionic. (a) H-H , b) O-CI c) Na-F , d) C-N , c) Cs - F , f) Zn CI |
| Answer» Solution :(a) covalent (B) covalent (C) Ionic (d) Covalent(E) ionic(F) covalent | |
| 35. |
The type of hybrid orbitals used by chlorine atom in ClO_(3)^(-) is |
|
Answer» `sp^(3)` `1S^(2) 2s^(2) 2p^(6) 3s^(2)underset(sp^(3) "hybridisation")ubrace(3s^(2)3P_(x)^(1)P_(y)^(1)P_(Z)^(1) )3d ^(1) 3d^(1)` STRUCTURE of `ClO_(3)^(-)` is 
|
|
| 36. |
What is the significance of the terms-'isolated gaseous atom' and 'ground state' while defining the ionization enthalpy and electron gain enthalpy? |
|
Answer» SOLUTION : Isolated gaseous atom means that the atom should be FEE from other atom in the gaseous state. No ENERGY should be required to SEPARATE it further from other atoms. GROUND state means the lowest energy state possible for that atom. These terms are used for comparison purposes. |
|
| 37. |
Which of the following species will have the largest and the smallest size Mg, Mg^(2+), Al, Al^(3+)?. |
|
Answer» Solution :Atomic radii decrease across a period. Cations are smaller than their parent atoms. AMONG isoelectronic ions, the one with the large positive nuclear charge will have a smaller radius. Hence the largest species is Mg, the smallest one is `Al^(3+)`The SIZE of an ANION greater while that of the cation is smaller than that of its parent ATOM, e.g. `F^(-) = (1.36 Å) gt F (= 0.72 Å) , CL^(-) (=1.81 Å) gt Cl (=0.99 Å) , Na^(+) =(0.95 Å) lt Na (=1.90 Å) , Ca^(2+) (=0.99 Å) lt Ca(=1.97Å)` |
|
| 38. |
What is green house effect? |
| Answer» SOLUTION :Atmosphere traps the sun.s HEAT near the earth.s surface and keeps it warm. This is CALLED natural green house effect. | |
| 39. |
What type of cleavage occurs in a compound containing non polar covalent bond formed between atoms of similar electronegativity ? |
|
Answer» Homolytic CLEVAGE |
|
| 40. |
Which is wrong according to kinetic theory |
|
Answer» <P>Average K.E. of molecules is proportional to the ABSOLUTE temperature |
|
| 41. |
Which of the following represents the absolute mass of .Na. atom? |
|
Answer» 23 gms |
|
| 42. |
Which of the following statements are not true about the following decomposition reaction? 2KCl_(3(s)) rarr 2KCl_((s)) +3O_(2(g))uarr |
|
Answer» Potassium is undergoing OXIDATION All the statements are incorrect because it is a REDOX reaction in which chlorine is under-going reduction while oxygen is undergoing oxidation. |
|
| 44. |
Write the structure of : (i) (E) penta-1,3-diene "" (ii) (2Z,4E)yhexa-2,4-diene (ii) (2E,4E)-3-ethylhexa-2,4-diene ""(iv) (R)-2-Bromopentane (v) (S)-3-bromo-3-chlorohexane "" (vi) (2S,3R)-2,3-dibromobutane |
|
Answer» |
|
| 45. |
Which of the following statements about ozone layer is true ? |
|
Answer» Ozone layer is beneficial to us because ozone CUTS out the ultraviolet radiation of the sun |
|
| 46. |
Which of the following is not correctly matched : |
|
Answer» LATICE acid `CH_3 - undersetoverset(|)(OH)(C )H - COOH` |
|
| 47. |
What feaction (n/N)of the lattice sites are vacent at 298 K for a crystal for which the energy required to make a defect is 1 eV ( 1 eV = 1.602 xx 10^(-19) J) ? |
|
Answer» Solution :If ` E_(v) , ` is the energy required to CREATE one vacant site, then by Boltzmann formula . ` n/N =e ^(-E _(v)//KT) `( k = Boltzmann constant =` R/N_(0)` ` or In n/N = - (E_(v))/(kT) ` ` or log n/N = - ( E_(v))/(2.303 kT) = - ( 1.602 xx10^(-19))/(2.303( 8.314//6.023 XX 10^(23)) xx 298)` ` =- 0.00169185 xx 10^(4) = 16.9185= bar(17) . 0815` `n/N = " Antilog" bar(17) .0815 = 1.206 xx 10^(-17)` |
|
| 48. |
When a substance reacts with water, it produces a combustible gas B and a solution of substance C in water. When another substance D reacts with this solution of C, it produces the same gas B on warming but D can produces gas B on reaction with dilute sulphuric acid at room temperature. A imparts a deep golden yellow color to a smokeless flame of Bunsen burner. A, B, C and D respectively are..... |
|
Answer» `Na,H_2,NAOH,Zn` |
|
| 49. |
Which of the following is true according to Charle.s Law ? |
|
Answer» Pressure - Volume |
|
| 50. |
What inferences do you observe by the values of Q and K_(C) ? |
|
Answer» Solution :If `Q=K_(C)`, the reaction is in EQUILIBRIUM STATE. If `QgtK_(C)`, the reaction will PROCEED in the reverse direction i.e., formation of reactants. If `QltK_(C)`, the reaction will proceed in the forward direction i.e., formation of PRODUCTS. |
|