Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The temperature at which all the three phases of a substance exist together is called ………………….. |
|
Answer» |
|
| 2. |
The substance which can not be used for preparing H_(2)O_(2) solution is |
|
Answer» `Na_2O_2` |
|
| 3. |
What is the molar solubility of Fe(OH)_2 (K_(sp) =8.0 xx 10^(-16) )at pH 13.0 ? |
|
Answer» `8.0 xx 10^(-18) ` ` "" S "" (2S +0.1 ) ` ` 8 xx 10 ^(-16)=(S) ( CANCEL (2S) +0.1 ) ^(2) ` ` S= 8 xx 10 ^(-14)M` |
|
| 4. |
Which of the following redox reaction is oxidation and which is reduction? Fe toFe^(2+)+2e^(-) |
| Answer» SOLUTION :OXIDATION | |
| 5. |
Which are the type of molecular orbitals ? Give information in short. |
|
Answer» Solution :Molecular orbitals of DIATOMIC molecules are designated or `SIGMA `(sigma), `pi` (Pi) `delta` (Delta) etc. `sigma `(sigma) molecular orbitals: The sigma `(sigma)` molecular orbitals are symmetrical around the bond axis. e.g. `sigma_(1s) and sigma_(1s)` : The linear combination of 1s orbitals centered on two nuclei produces two molecular orbitals which are symmetrical around the bond-axis. Such molecular orbitals are of the o type and are DESIGNED as ols and `sigma` 1 s and `sigma^(**)` eg. `sigma 2p_(z)` and `sigma^(**)2p_(z)`:If internuclear axis is taken to be in the Z-direction, it can be seen that a linear combination of `2p_(z)` orbitals of two atoms also produces two sigma molecular orbitals designated or `sigma (2p_(z)), sigma^(**) (2p_(z)) sigma (1s), sigma^(**) (1s), sigma(2s), sigma^(**) , sigma(2p_(z)) , sigma^(**)(2p_(z))` are `sigma` types ad symmetrical around the bond axis. `pi and pi^(**)` types molecular orbitals : The `pi` molecular orbitals are not symmetrical around the bond axis because in these above and below the bond axes (+) and (- ) symbol are not symmetrical. Molecular orbitals obtained form`2p_(X) and 2p_(y)` orbitals are not symmetrical around the bond axis because of the presence of positive lobes above and negative lobes below the molecular plan. Such molecular orbitals are labelled as `pi and pi^(**)` . A `pi` bonding MO has larger electron density above and below the inter NUCLEAR axis. The `pi^(**)` antibonding MO has a node between the nuclei. |
|
| 6. |
Which of the following is not true in Rutherford's nuclear model of atom |
|
Answer» protons and neutrons are present INSIDE nucleus |
|
| 7. |
Which of the following sets of quantum num bers represents the least energy of an atom ? |
|
Answer» N = 3,1 = 0, m = 0, s = +1/2 |
|
| 8. |
What are d-block elements? Why are they called so? |
|
Answer» Solution :• The elements WHOSE last electron enters into the d-orbital of the penultimate shell (n-1) are having SIMILAR properties and called as d-block elements. • The groups of 3 to 12 in the CENTRE of the periodic table belongs to d-block. |
|
| 9. |
XeF_(2) is isostructural with : |
|
Answer» `TeF_(2)` are isostructural . |
|
| 10. |
Which of the following statements is correct for a reversible process in a state of equilibrium ? |
|
Answer» `DeltaG = -2.30 "RT log K"` because at EQUILIBRIUM `Delta G = 0` |
|
| 11. |
Whatare ortho para directors expalin why OHgroupis an ortho para directorand activator |
|
Answer» Solution :Thegroupwhichincreasethe electrondensityat orthoand parapositionare calledasortho -paradirection Example`-OH , - NH_(2)- NHR -OCH_(3)-CH_(3) - C_(2)H_(5)` etc Letus considerthe directioninfluence of phenolicgroup PHENOL istheresonancehybrid ofthe followingstructure  intheseresonancestructuresthe negativeresidue in thepresentat orthoand parawhichis attachedto theisinvolvedin resonanceand it makesthe ringmoreelectronrichmeta positionthereforephenolicgroupactivatesthe benzeneringforelectrophilicattackatorthoand parapositionHenceOHgroupis an orthoparadirectionand ACTIVATOR. |
|
| 12. |
What is the type of hybridization exhibited by Boron in Boron trifluoride? |
| Answer» SOLUTION :`SP^(2)` | |
| 13. |
When 100gm Ethene polymerises entirely to poly ethene, the weight of poly ethene formed as per the equation nCH_3 = CH_2 to (CH_2- CH_2)_n is |
|
Answer» `(n)/(2) g` = Moles of POLY Ethene ` xx n xx 2 ` `(100)/(28) xx 2 = ("weight of poly ethene ")/(28 xx n) xx n xx 2` Weight of poly ethene = 100g |
|
| 14. |
What will be the number of sigma and pi bonds in Biphenyl respectively ? |
|
Answer» `12 SIGMA` and `6 pi` |
|
| 15. |
What are the oxidation number of the underlined elements in each of the following and how do you rationalise the results ? |
| Answer» SOLUTION :C of COOH has o0XIdation NUMBER +2, C of `CH_3` has o0XIdation number -2. | |
| 16. |
Which of the following arrangements represent increasing oxidation number of the central atom ? |
|
Answer» `CrO_(2)^(-),CIO_(3)^(-),CrO_(4)^(-2),MnO_(4)^(-)` |
|
| 17. |
The volume of the reaction vessel containing an equilibrium mixture is increased in the following reaction SO_(2) Cl_(2(g))hArrSO_(2(g)) + Cl_(2(g))when equilbrium is re-establised. |
|
Answer» the amount of `Cl_(2(g))` REMAINS unchanged |
|
| 19. |
X ml of H_2 gas effuses through a hole in a container in 5 sec. The time taken for the effusion of same volume of gas specified below under identical conditions is |
|
Answer» 10 SEC, He |
|
| 20. |
Three students, Manish, Ramesh and Rajni were determining the extra elements present in an organic compound given by their teacher. They prepared the Lassaigne's extract (L.E) independently by the fusion of the compound with sodium metal. Then they added solid FeSO_(4) and dilute sulphuric acid to a part of Lassaigne's extract. Manish and Rajni obtained prussian blue colour but Ramesh got red colour. Ramesh repeated the test with the same Lassaigne's extract, but again got red colour only They were surprised and went to their teacher and told him about their observation. Teacher asked them to think over the reason for this. Can you help them by giving the reason for this observation. Also, write the chemical equations to explain the formation of compounds of different colours |
|
Answer» Solution :If the organic compound contains both N and S, then during fusion it may form either SODIUM thiocynate (NaSCN) or a mixture of sodium cyanide (NaCN) and sodium SULPHIDE (Na2S) depending upon the amount of sodium metal used. If the sodium metal used is less, only NaSCN is produced. This then reacts with `Fe^(3+)` ions (Produced by oxidation of `Fe^(2+)` ions during `"prep"^(n)` of lassaigne.s extract) to give red colouration due to the from of ferric thiocyanate. `FO^(2+) overset("Aerial oxidation")rarr Fe^(3+), FO^(3+) + 3NaSCN rarr Fe(SCN)^(3) + Na^(+)` (Ferric thiocyanate (Red colour) NaCN they formed then reacts with `FeSO_(4)`, more NaCN and Fe3+ ions to give ULTIMATELY Prussian blue colour due to the FORMATION of ferric ferro cyanide or iron (III) hexacyanoferrate (II). `2NACN + FeSO_(4) rarr Na_(2)SO_(4) + Fe(CN)_(2)` `Fe(CN)_(2) + 4NaCN rarr Na_(4) [Fe(CN)_(6)]` Sodium hexacyanoferrate (II) `3Na_(4)[Fe(CN)_(6)]+ 4Fe^(3+) rarr Fe_(4)[Fe(CN)_(6)]_(3) + 12Na^(+)` Iron (III) hexacynoferrate (II) (Prussian blue) From the above discussion, it follows that Manish and Rajni used excess sodium and hence formed NaCN in the Lassaigne.s extrat which gave Prussian blue colour due to the formation of iron (III) hexacyanoferrate (II) while Ramesh used less sodium and hence formed NaSCN in the Lassaigne.s extract which gave red colouration due to the formation of ferric thiocyanate |
|
| 21. |
Which of the following is not involved in Born-Haber cycle? |
|
Answer» Sublimation ENERGY |
|
| 22. |
Which of the following species have + 6 state with sulphur? |
|
Answer» `H_(2)S_(2)O_(7)`oleum `overset(+1)H-overset(-2)O-overset(-2)overset(O)overset(||)underset(-2)underset(O)underset(||)underset(x"")(S)-overset(-1)O-overset(-1)O-underset(-2)underset(O)underset(||)overset(-2)overset(O)overset(||)underset(x"")(S)-overset(-2)O-overset(+1)H` `2x +2 -14 =0, ""x =+6` `overset(+1)H-overset(-2)O-overset(-2)overset(O)overset(||)underset(-2)underset(O)underset(||)(S^x)-underset(-1)O-underset(-1)O-underset(+1)H` `x +2 -8 =0 , x=+6` |
|
| 23. |
Which of the following has least electron attraction capacity than hydrogen? |
|
Answer» `(CH_(3))_(3)C-` |
|
| 24. |
Which of the following forms vortex rings ? |
|
Answer» `P_(2)O_(5)` |
|
| 25. |
Write the resonance structure of benzene. |
Answer» SOLUTION : 
|
|
| 26. |
Which substance is used as a Fire Extenguisher ? |
|
Answer» SODIUM oxide |
|
| 27. |
Which of the following is the most stable |
|
Answer» `F_(3)C^(-)` |
|
| 28. |
Which among the following gives the expression for work done by ideal gas ? |
|
Answer» <P>`-P DELTAV` |
|
| 29. |
Which of the following statements are correct |
|
Answer» IUPAC name of neopentane is 2,2-di methyl propane |
|
| 30. |
Which color does alkali metals produces with ammonia ? |
|
Answer» Violet |
|
| 31. |
Whichoneof the followingis not a monocylic aromatic hydrocarbon |
|
Answer» BENZENE |
|
| 32. |
Which is more basic than hydroxide (OH^(-)) ion ? |
|
Answer» `CH_(3)-NH_(2)` |
|
| 33. |
What volume of 0.05 M Cr_(2)O_(7)^(2-) in acid medium is needed for complete oxidation of 200 mL of 0.6 M FeC_(2)O_(4) solution ? |
|
Answer» 0.6 L |
|
| 34. |
underset(hv)overset(Cl_(2))rarr number of theretically possible dichloro products are: |
Answer» 
|
|
| 35. |
Which of the following is not hyperconjugate structure of propene? |
|
Answer» `H- UNDERSET(underset(H)(|))OVERSET(overset(H)(|))(C )- underset(H^(+))overset(overset(H)(|))(C )- underset(underset(H)(|))overset(overset(H)(|))(C )` |
|
| 36. |
When 1-Bromo-1-methylcyclohexane is heated in ethanol for an extended period of time, three products result : one either and two alkenes . Predict the products of this reaction, and propose mechanism for their formation.Also, mention the major elimination product. |
|
Answer» |
|
| 37. |
When a catalyst is added to a system the |
|
Answer» Value of equilibrium CONSTANT is decreased |
|
| 38. |
The treatment ofCH_(3) MgXwith CH_(3)-=C-H produces : |
|
Answer» `Ch_(3)-CH=CH_(2)` |
|
| 39. |
Which set of quantum numbers is not possible ? |
|
Answer» `{:(n,l,m,"s"),(3,2,0,+1//2):}` |
|
| 40. |
Which reaction is suitable for preparing alpha- Chloroacetic acid ? |
|
Answer» Hell -Volhard ZELINSKY REACTION |
|
| 41. |
Which pairs can not explain Dalton's law ? |
|
Answer» `H_2, O_2` `NH_3 + HCl to NH_4Cl` `NO + O_2 to NO_2`. |
|
| 42. |
Two closed bulb of euqal volume (V) containing an ideal gas initially at pressure p_(i) and TemperatureT_(1) are connectedthrough a narrow tube of negligible volume as shown in the figure below. The temperature of one of the bulbs is then released to T_(2). The final pressure p_(t) is : |
|
Answer» <P>`2p_(i)((T_(1))/(T_(1)+T_(2)))` `n_(1) = (P_(i)V)/(RT_(1))` and `n_(2) = (P_(i)V)/(RT_(2))` After the temperature of second bulb is raised to `T_(2)`, the number of moles of gas in both the bulbs are : `n_(1)^(') = (P_(i)V)/(RT_(1))` and `n_(2)^(') = (P_(1)V)/(RT_(2))` Number of moles of two gases in both the bulbs remain unchanged. `n_(1) + n_(2) = n_(1^(')) + n_(2^('))` `(2P_(i)V)/(RT_(1)) = (P_(f)V)/(RT_(1)) + (P_(f)V)/(RT_(2))` or `(2P_(i))/(T_(1)) = P_(f) [(T_(1) + T_(2))/(T_(1)T_(2))]` or `P_(f) = (2P_(i)T_(2))/(T_(1) + T_(2))` |
|
| 43. |
What is energy gap in band theory ? Compare its size in conductors, semiconductors insulators |
| Answer» Solution :ENERGY GAP in BAND theory MEANS the gap of energy between valence band and conduction band. The gap is in the order : Conductor lt SEMICONDUCTOR lt Insulator | |
| 44. |
The triad not present in Group VIII of Mendeleeff's table |
| Answer» Answer :A | |
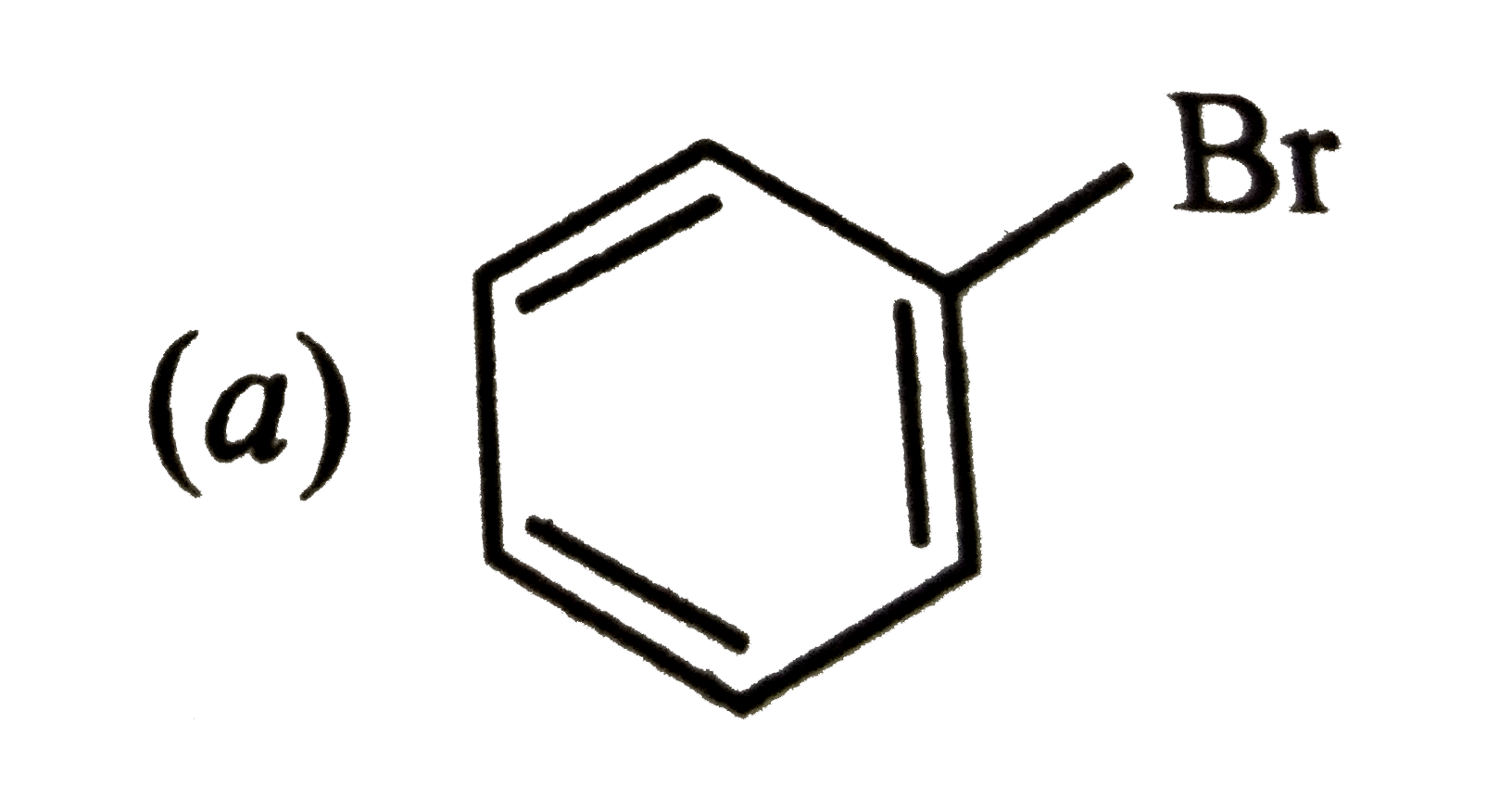
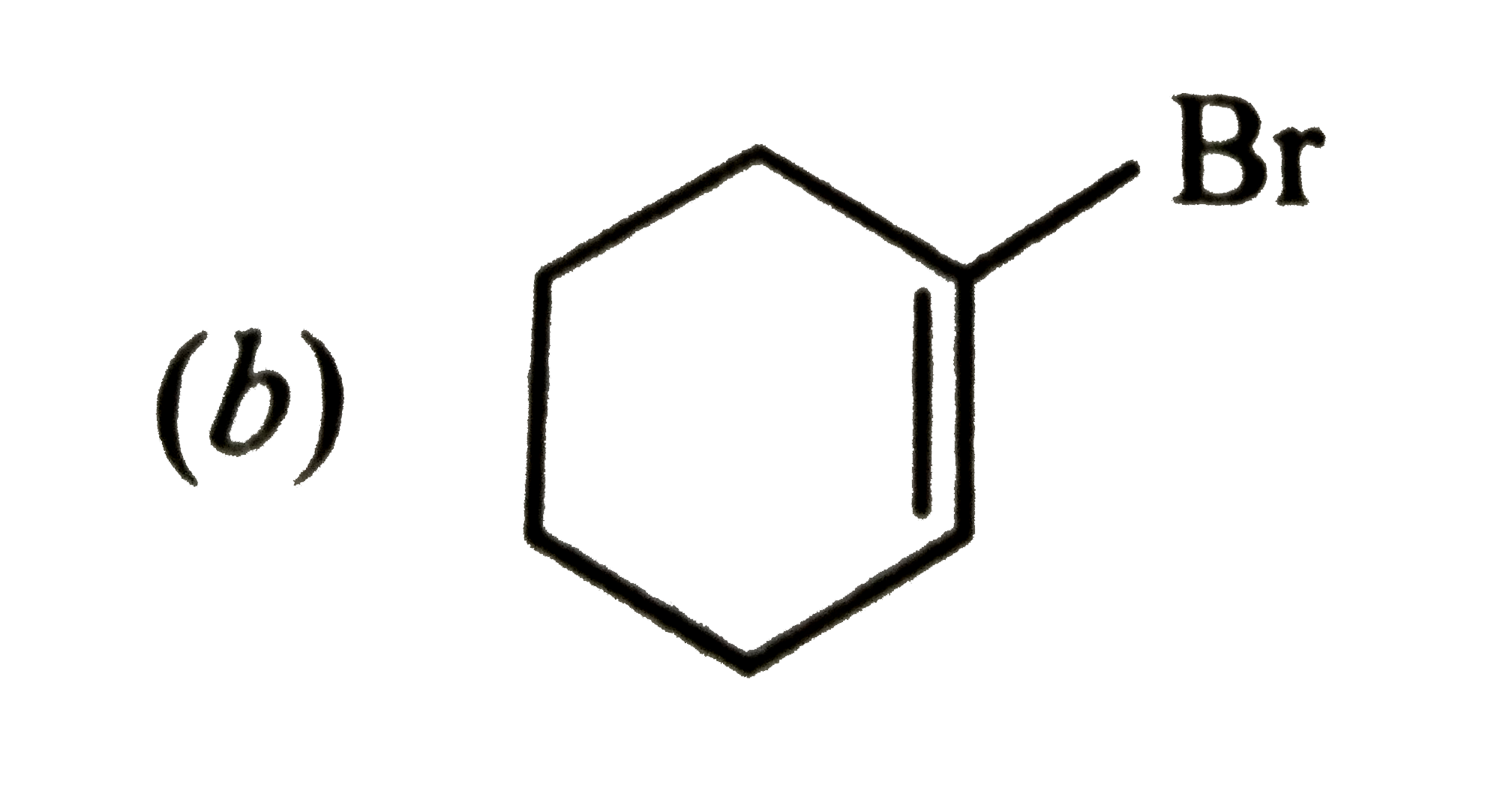
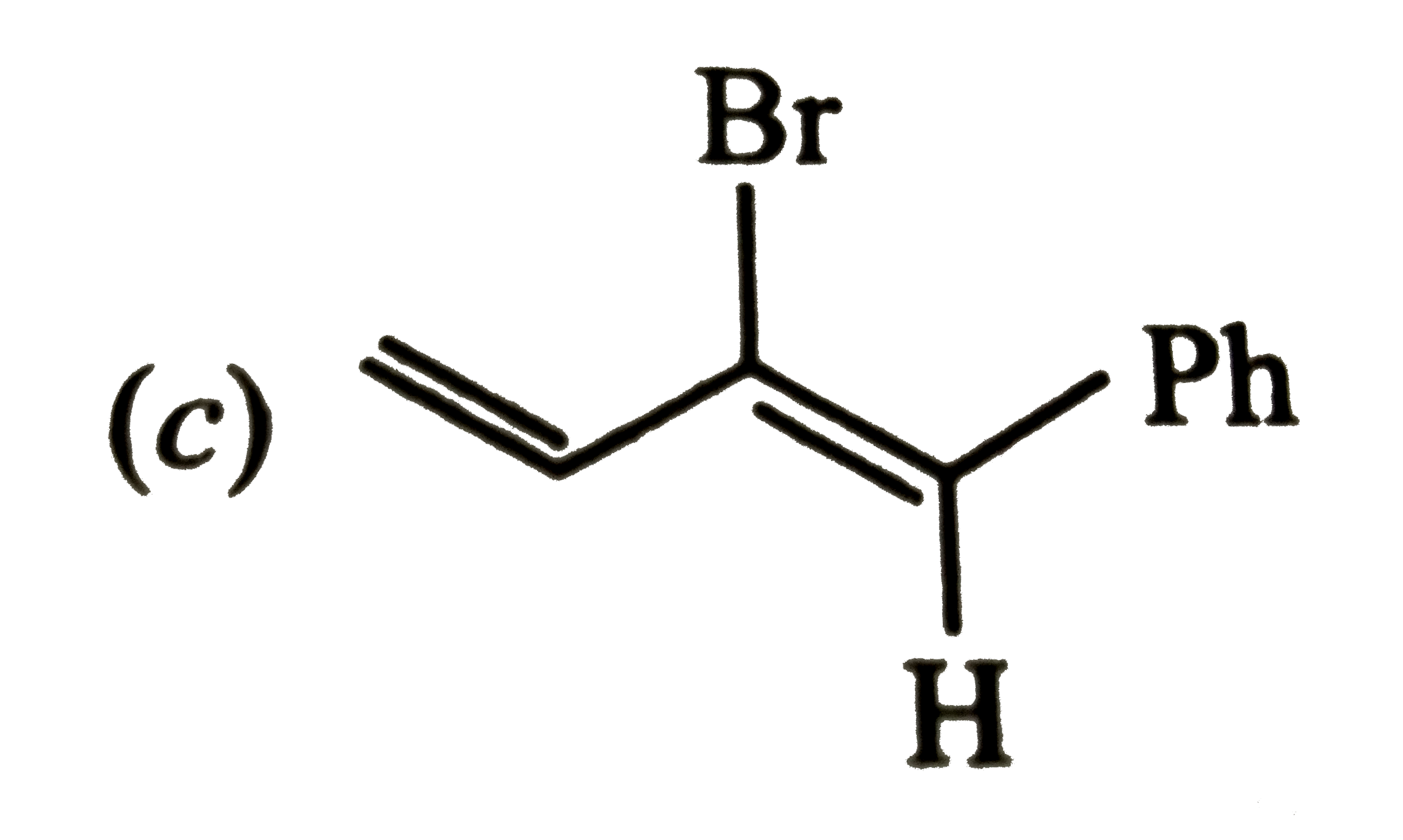
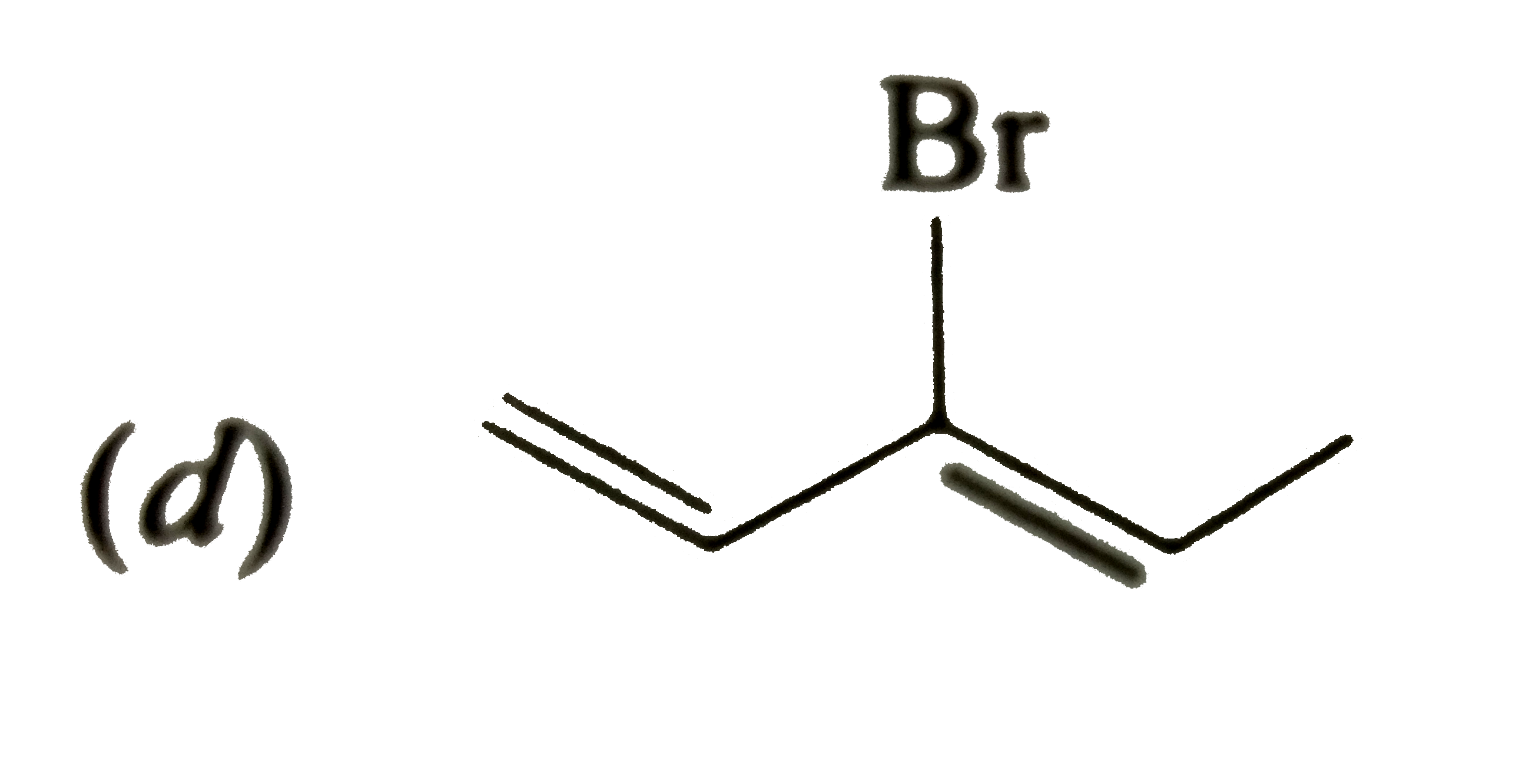
| 45. |
Which of the following will most readily give the dehydrohalogenation products ? |
|
Answer»
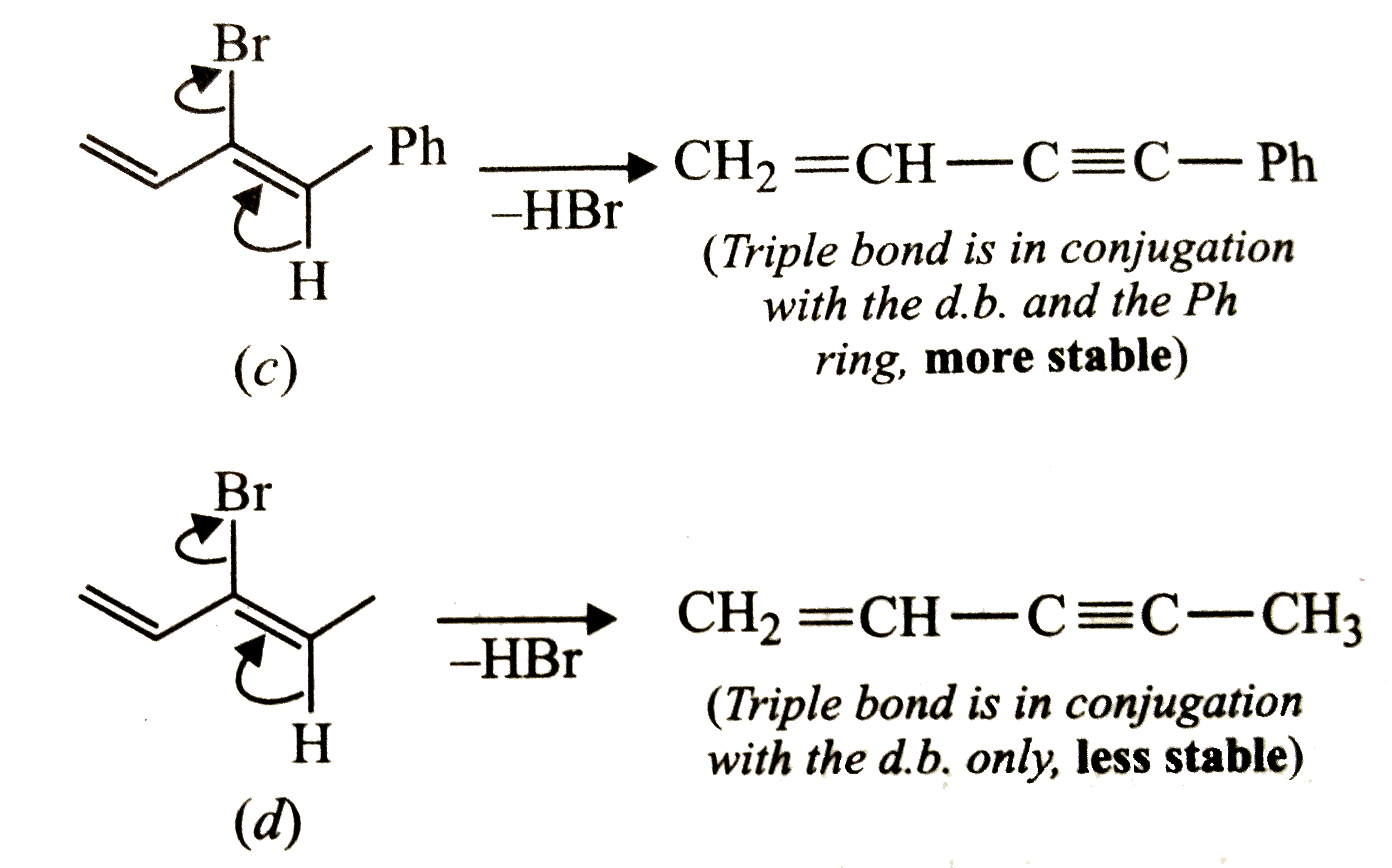 Thus, OPTION (c ) is correct. |
|
| 46. |
The region which is greatly affected by air pollution is |
|
Answer» TROPOSPHERE |
|
| 47. |
Which of the following statements is correct? A) CO_(2) is neither combustible nor supporter of combustion B) CO is a combustible gas C) CO burns with a blue flame |
|
Answer» A, B |
|
| 48. |
What is +E effect? |
|
Answer» Transfer of electron TAKES PLACE TOWARDS the attacking reagent |
|
| 49. |
Which of thefollowingare nod d- blockelements ? |
|
Answer» `[Xe] 5D^(1)6s^(2)` |
|
| 50. |
What are carbanions? |
Answer» SOLUTION :Let US consider the hetrolytic fission of the bond C-X present in an organic molecule.Ifthe carbon atom has greater ELECTRONEGATIVITY than the atom X, THEFORMER takes away thebonding electron pair and acquires a negative charge. The resulting carbon anion is known ascarbanion. 
|
|