Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Whitethe resonatingstructuresfor SO_3 ,NO_2 andNO_(3)^(-) ? |
Answer» SOLUTION :
|
|
| 2. |
What is the structure of ammonia & water molecule ? |
| Answer» SOLUTION :PYRAMIDAL and BENT STRUCTURE respectiyely. | |
| 3. |
Which of the following indicates Charles' law mathematically (when n, P are constant)? a) VT = constant "" b) V_t = V_0 (1 + t/(273)) c)V_0 = V_t (1 + t/273)""d) V/T = constant (when n, P are constant) |
|
Answer» a,c |
|
| 5. |
Which of the following is called non benzonoid molecule / ion |
|
Answer»
|
|
| 6. |
What volume of O_(2) is liberated at STP by warning 3.4 g of hydrogen peroxide ? |
|
Answer» Solution :`2H_(2)O_(2)to2H_(2)O+O_(2)` `2xx34g` of `H_(2)O_(2)` can LIBERATE `22.4L` of `O_(2)` at STP. `3.4g` of `H_(2)O_(2)=(3.4xx22.4)/(2xx34)=1.12L` of `O_(2)` at STP. |
|
| 7. |
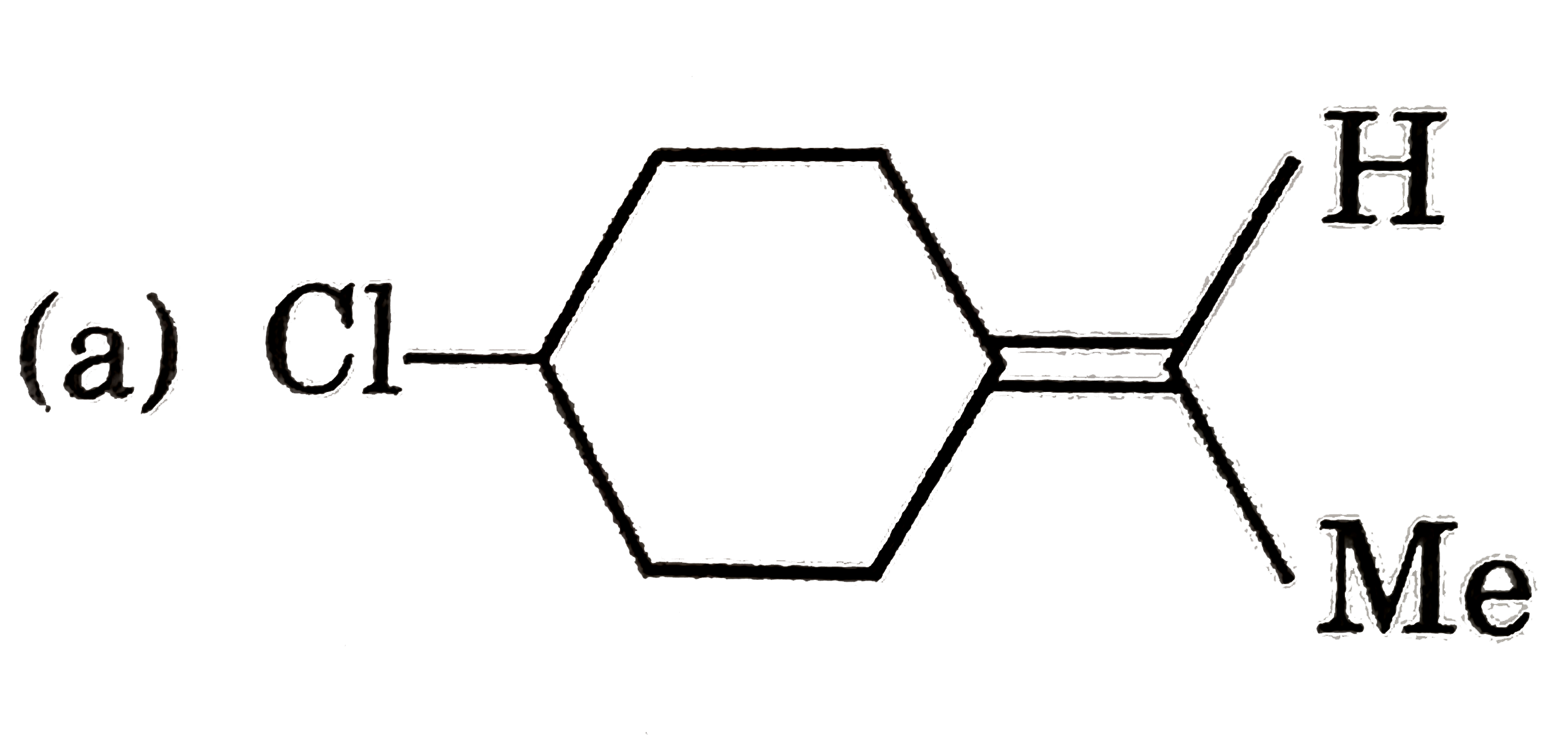
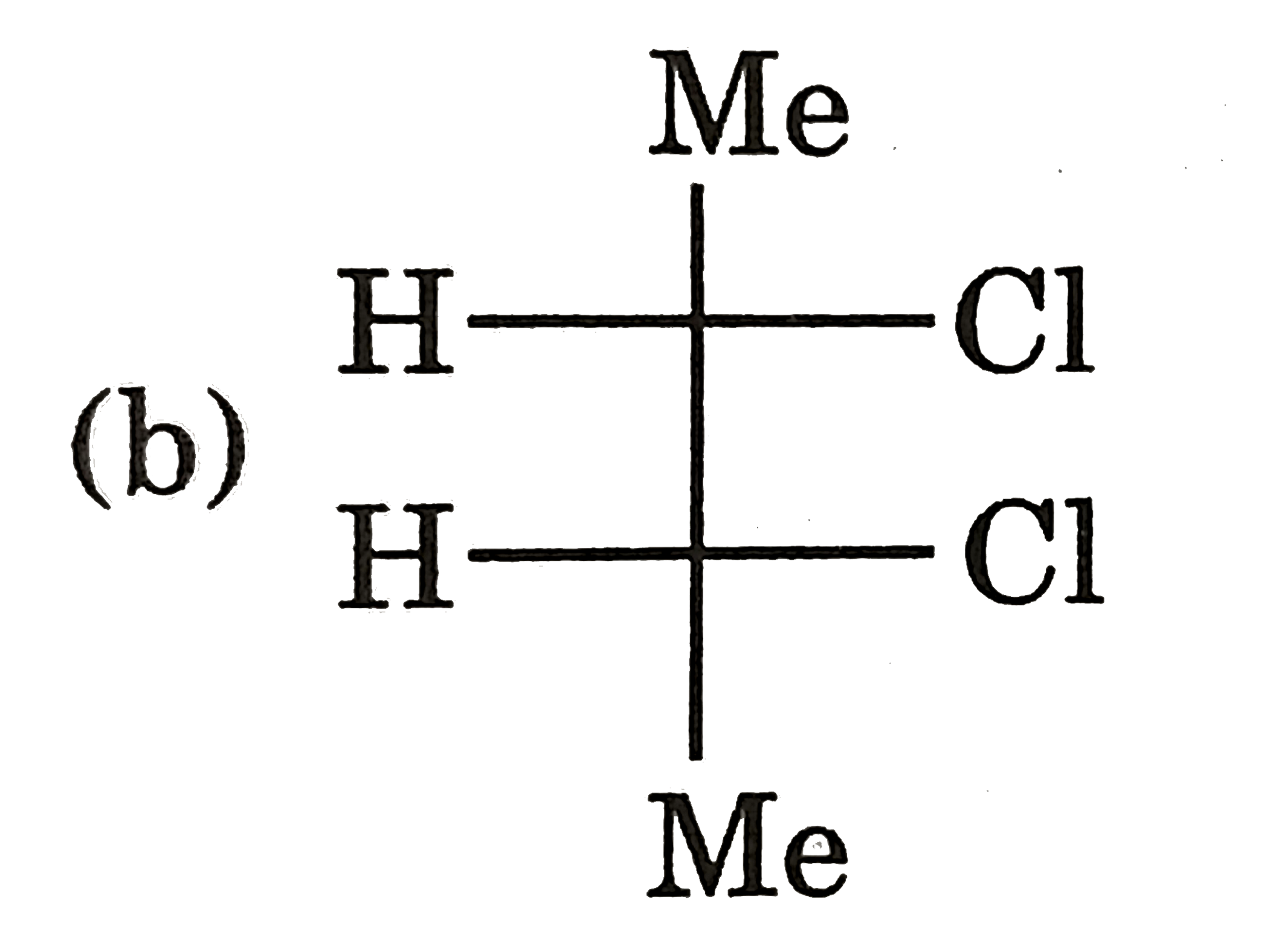
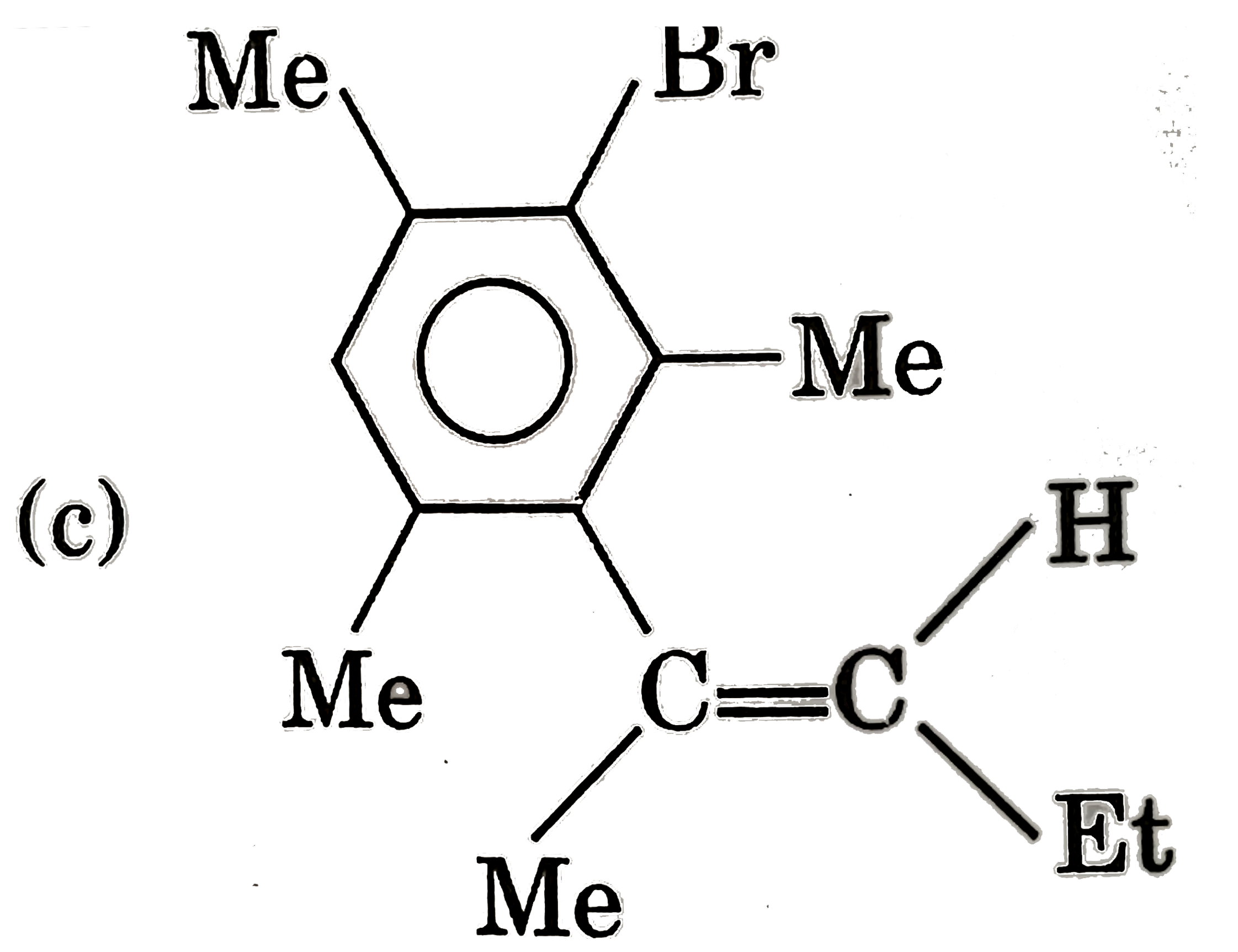
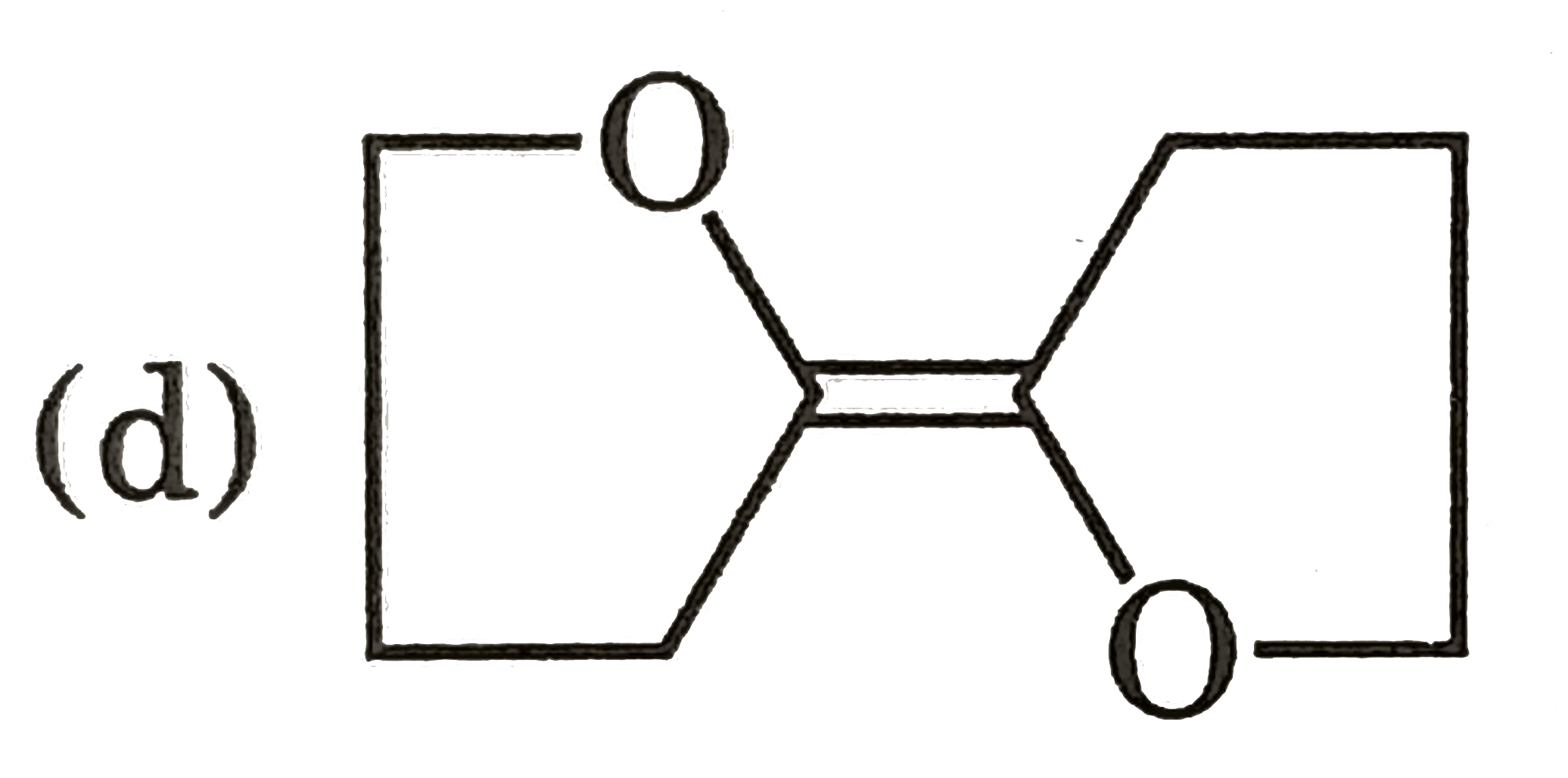
Which of the following shows geometrical isomerism |
|
Answer» `BrCH=C=C=CHBr` |
|
| 8. |
What of the following can not show optical isomerism? |
|
Answer»
|
|
| 9. |
Which of the following pairs of compounds are geometrical isomers |
|
Answer» Crotonic ACID and cinnamic acid  Fumaric ACIDS are GEOMETRICAL ISOMERS |
|
| 10. |
Which of the following reactions requires an oxidant - |
|
Answer» `Cu^(2-)TOCU` |
|
| 11. |
Which of the following is Lindlar's catalyst ? |
|
Answer» Pd+Pt |
|
| 12. |
Two liquids X and Y on mixing gives a warm solution. The solution is |
|
Answer» 1) ideal |
|
| 13. |
What is resonance energy? |
| Answer» Solution :Resonance ENERGY is ALSO known as STABILISATION energy. ..The difference in energy between the actual STRUCTURE and the LOWEST energy resonance structure is called the resonance energy.. | |
| 14. |
This reaction is called |
|
Answer» Dakin-REACTION |
|
| 15. |
Which of the following is the most stable conformer of 1, 2, 4 -trimethyl cyclohexane |
|
Answer»
|
|
| 16. |
What mass of magnesium hydroxide is required to neutralise 125 mL of 0.136 M HCl solution ? ("Substance")/(Mg(OH)_(2)) "" ("Molar mass")/(58.33 " g mol"^(-1)) |
|
Answer» 0.248 g |
|
| 17. |
The recation H_(3)PO_(2)+D_(2)OtoH_(2)DPO_(2)+HDO indicates that hypo-phosphrous acid is…. |
|
Answer» TRIBASIC acid |
|
| 18. |
What is meant by the term auto-protolysis of water ? |
|
Answer» Solution :Auto-protolysis of water means self ionization of water. In other words, TWO molecules of water react with each other through proton transfer, i.e., one acts as the ACID while the other acts as the base. The molecule which accepts a proton is CONVERTED into `H_(3)O^(+)` ion while that which loses a protonis converted into `OH^(-)` ion. Thus. `underset("Acid"_(1))(H_(2)O(l))+underset("Base"_(2))(H_(2)O(l)) HARR underset("Acid"_(2))(H_(3)O^(+)(aq))+ underset("Base"_(1))(OH^(-)(aq))` |
|
| 19. |
Which ofthe statements are false? |
|
Answer» Physical quantity REPRESENTED by volume is `dm^(3)` `implies 5` joules (not Joules) |
|
| 20. |
Which of the following is the most stable carbocation . |
|
Answer»
|
|
| 21. |
Write IUPAC names of the products obtained by the ozonolysis of the following compounds: (i) Pent-2-ene (ii) 3,4-Dimethyl-hept-3-ene (iii) 2-Ethylbut-1-ene (iv) 1-Phenylbut-1-ene |
Answer» SOLUTION :
|
|
| 22. |
The term Vander Waals forces refers to A) Dipole -- dipole interaction "" B) Dipole - induced dipole C) Dispersion forces |
|
Answer» A,C |
|
| 23. |
Which of the following statements about photochemical smog is wrong? |
|
Answer» It has high concentration of oxidising agents |
|
| 24. |
The value of equilibrium constant depends on what ? |
| Answer» SOLUTION :The VALUE of equilibrium constant depends upon (i) NATURE of the REACTION (ii) TEMPERATURE. | |
| 25. |
Which dicarboxylic acid gives AcOH on stron heating ? |
|
Answer» Ethanedioic acid 
|
|
| 26. |
Which of the following alkenes on treatment with HBr in presence or absence of peroxide give the same product? |
|
Answer» Cyclohexene |
|
| 27. |
Which amog the followng statements is/are correct? |
|
Answer» `Phi^(2)` represents the ATOMIC orbitals |
|
| 28. |
why are reversible process non-static ? |
| Answer» Solution :In reversible processes, the RATE of two opposing reactions equals at a PARTICULAR stage. At this stage the conentration of reactants and products do not change with time. This condition is not STATIC and is dynamic, because both the FORWARD and reverse reactions are STILL occurring with the same rate . | |
| 29. |
Which one is correc for H_(2)O at 25^(0)C |
|
Answer» Ionic PRODUCT of water `K_(w) = 10^(-14)` `K_(C) = ([H^(+)][OH^(-)])/([H_(2)O]) = (10^(-7) xx 10^(-7))/(55.55) = 1.8 xx 10^(-16)` |
|
| 30. |
When sodium peroxide is treated with dilute H_(2)SO_(4), we get |
|
Answer» SODIUM sulphate and water |
|
| 31. |
Which solution has the lower freezing point ? 10 g of methanol ( CH_3 OH_)in 100 g of water ( or ) 20 gof ethanol(CH_2 H_5OH)in 200 g of water |
|
Answer» Solution :`Delta T _(f) = K _(f), m.i.e. Delta T_(f) prop m` `m_(CH_(3)-OH)= (((10)/(32)))/(0.1) = 3. 125m` `m _(C_(2)H_(5)-OH)= (((20)/(46)))/(0.2)= 2. 174 m` `therefore` Depression in freezing point is more in methanol solution and it will have lower freezing point. |
|
| 33. |
When ethyl iodide is allowed to reacts with sodium phenolate the product formed is |
|
Answer» Phenetole 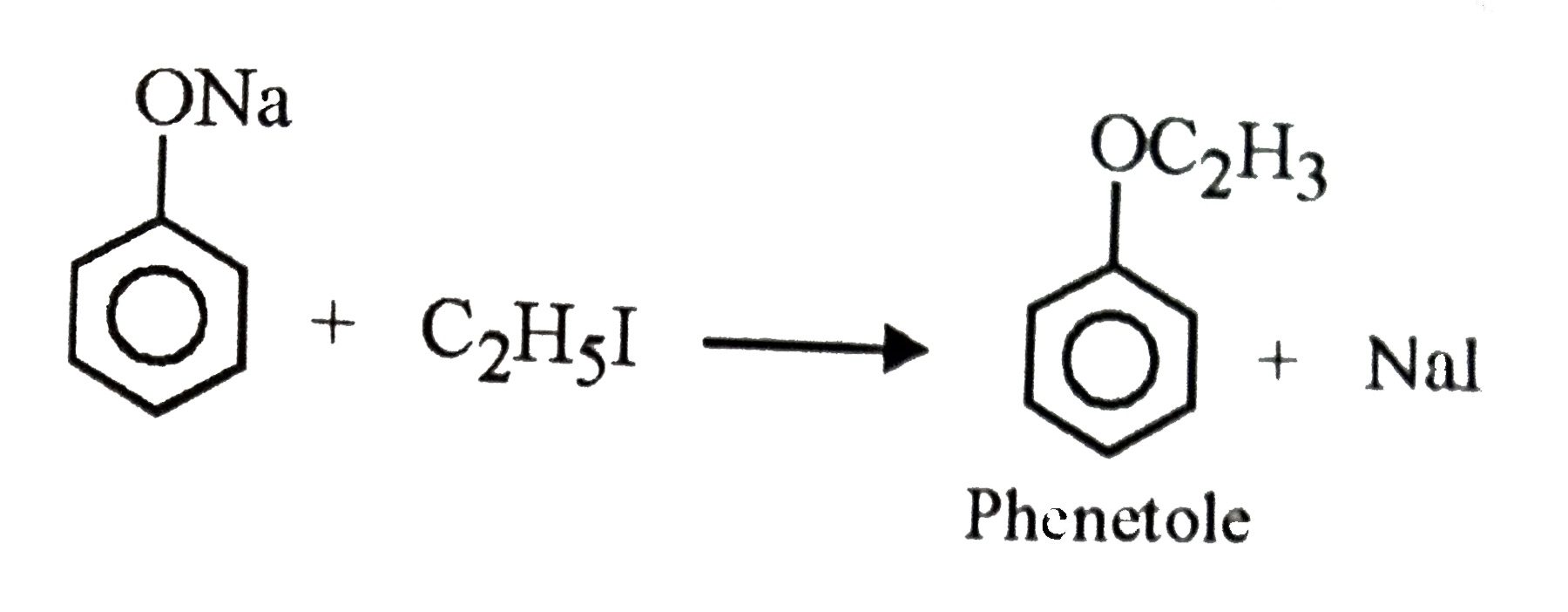
|
|
| 34. |
What are the limitations of the thermodynamics? |
|
Answer» Solution :(i)Thermodynamics suggests feasibility of REACTION but FAILS to suggest rate of reaction. It is concerned only with the initial and the final STATES of the system. It is not concerned with the path by which the change occurs (ii) It does not reveal the mechanism of a process |
|
| 35. |
Which of the following will not show defection from the path on passing thorugh an electric field ? Proton, cathode rays, electron, neutron |
| Answer» Solution :Neutron is a neutral PARTICLE. HENCE, it will not be deflected on PASSING through an ELECTRIC FIELD. | |
| 36. |
What are silicons ? |
Answer» Solution :SILICONSARE synthetic organosilicons compoundscontaining repeated UNITS held by `Si-O-Si`LINKAGES. They are hydrophobic (water-repellent) is nature. UNITS held by `Si-O-Si`LINKAGES. They are hydrophobic (water-repellent) is nature.
|
|
| 37. |
Which of the following statement is false |
|
Answer» Every solid SUBSTANCE can be brought into colloidal state |
|
| 38. |
Which of the following reagent will convert glycerol to acrolein ? |
|
Answer» <P>`P_(2)O_(5)` 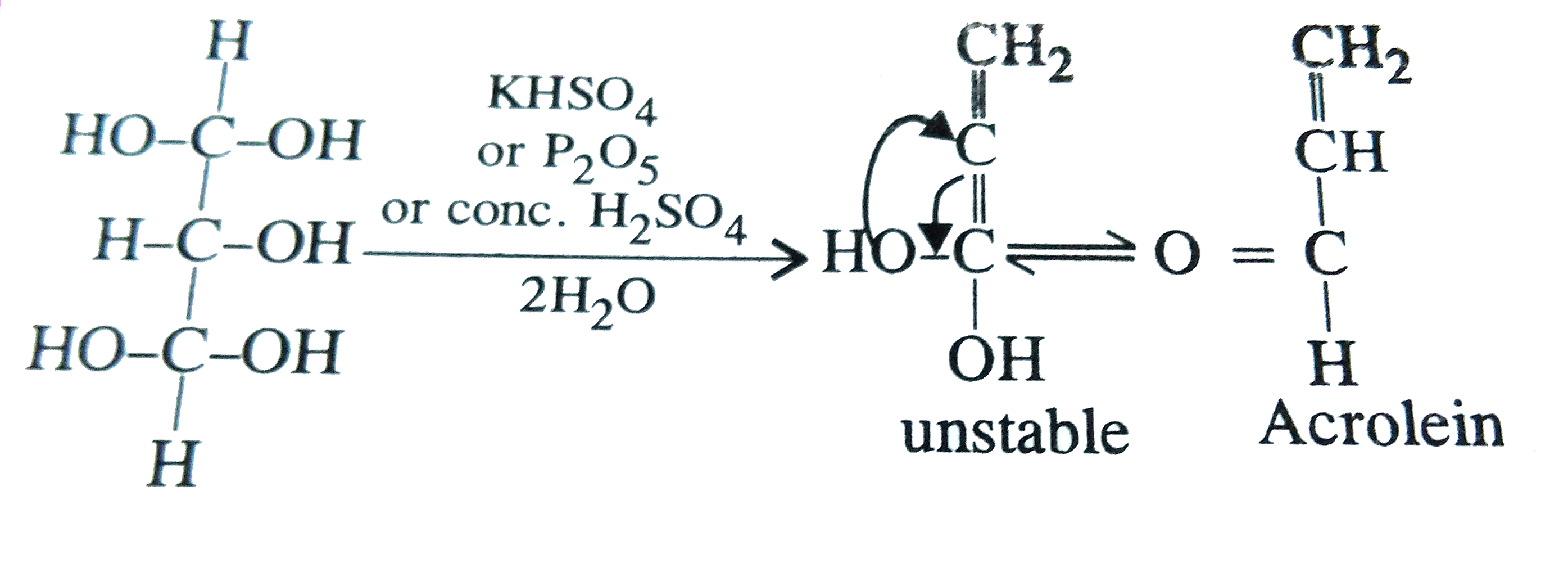
|
|
| 39. |
Which of the following boils at higher temperature ? |
| Answer» Answer :D | |
| 40. |
What type of forces do exist between the molecules of alkanes in liquid and solid states ? |
| Answer» SOLUTION :VAN DER WAALS. FORCES | |
| 41. |
The standard Gibbs energy change at 300 K for the reaction 2 A hArrB+ C " is"2494 *2 J At a given time, the composition of the reaction mixture is [A] =1/2 , [B] =2 and [C] =1/2 . The reaction proceeds in the : [R=8*314 J//K//mol, e=2*718] |
|
Answer» Forward direction because `Q gt K_(C)` ` 2494.2 = - 2*303 (8.314) (300) log K_(c)` orlog `K_(c) =-0*4342` or `K_(c)= "antilog" (bar 1*5658) = 0*3679` Now , when `[A] =1/2, [B] = 2 and [C] = 1/2` `Q_(c)= ([B][C])/([A]^(2)) = (2xx1/2)/((1/2)^(2))=4` As `Q_(c) gt K_(c),` reaction will proceed in the reverse direction. |
|
| 42. |
Tugsten has a density of19.35 " g cm"^(-3)and the length of the side of the unit cell of 316 pm. The unit cell in the most important crystalline from of tungsten is the body centred cubic unit cell. How many atoms of the element does 50g of the element contain ? |
|
Answer» |
|
| 43. |
What is the general valence shell electronic configuration of group 14 elements ? |
| Answer» SOLUTION :`ns^(2)np_(x)^(1)np_(y)^(1)np_(Z)^(0)` | |
| 44. |
Which type of 'defect' has the presence of cations in the interstitial sites ? |
|
Answer» SCHOTTKY defects |
|
| 45. |
When the molar concentrations of SO_(2), O_(2) and SO_(3)at equilbrium at certain temperature are 0.5 , 0.25& 0.25 M respectively, k_(c)for 2SO_(3) hArr 2SO_(2) + O_(2) is |
|
Answer» `0.5 lit MOL^(-1)` `K_(c)=([SO_(2)]^(2)[O_(2)])/([SO_(3)]^(2))` |
|
| 46. |
Whichof thefollowing isusedin qualiitiveas wellas quantitiveanalysis ? |
|
Answer» `NACL` |
|
| 47. |
Which of the following radial distribution graphs correspond to l=2 the H-atom- |
|
Answer»
|
|
| 48. |
Which of the following oxides behaves as conductor or insulator depending upon temperature ? |
| Answer» Answer :D | |
| 49. |
Those stereoisomers are called configurational isomers if they differ in spatial arrangement of atom or group and are not interconvertible without cleaving a bond. Geometrical isomer arises due to restricted rotation about a bond or centre. The number of G.I is 2^(n) where n= number of stereoisomers, in some cases the number of G.I is less than 2^(n) in case where either side nomenclature will be possible. The number of G.I. of the compound C_(2)H_(5)-CH=CH-CH=CH-CH=CH-CH_(3) is |
|
Answer»
|
|
| 50. |
Which of the following when reaction occur in present of catalyst ? |
|
Answer» ACTIVATION ENERGY decreases. |
|