Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which of the following is the most correct electron displacement for a nucleophilic reaction to take place ? |
|
Answer»
|
|
| 2. |
What do youunderstand byinterphase ? |
|
Answer» Solution :Linear combination of ATOMIC orbitals (LCAO0): (i) The wave functions for the molecular orbitals can be obtained by solving Schrod1nger Wave equation for the molecule. Since solving Schrodinger wave equation is too complex, a most common method linear combination of atomic orbitals (LCAO) IS Used to OBTAIN wave function for molecular orbitals. (ii) Atomic orbitals are represented by wave functions `Psi.` Consider two atomic orbitals represented by the wave functions `Psi_(A) and Psi_(B)` with COMPARABLE energy that combines to FORM two molecular orbitals. (iii) One is bonding molecular orbital `(Psi " bonding")` and the other is anti-bonding molecular orbital `Psi " anti-bonding").` (IV) The wave function for molecular orbitals, `Psi_(A) and Psi_(B)` can be obtained by the LCAO as hown below: `Psi_("bonding")=PsiA+Psi_(B)` `Psi_("anti-bonding")=Psi_(A)-Psi_(B)` (v) The formation of bonding molecular orbital can be considered as the result of constructive interference of the atomic orbitals and the formation of anti-bonding molecular orbital can be the result of the destructive interference of the atomic orbitals. (vi) The formation of two molecular orbitals from two Is orbitals is show below. Constructive interaction: The two ls orbitals are in phase and have the same signs. 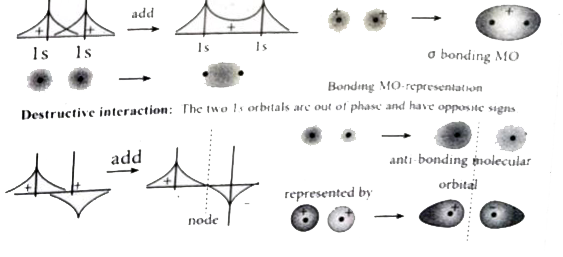 Bonding MO-representation Destructive interaction: The two Is orbitals are out of phase and have opposite signs. 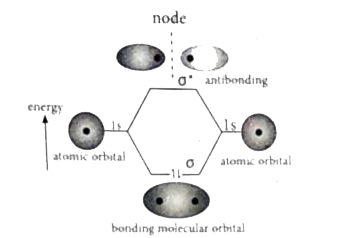
|
|
| 3. |
x mol N_(2) O_(4) is taken at P_1 atm in a closed vessel & heated . When 75% N_2 O_4 dissociated at equilibrium , total pressure is found to be P_2 atm . The relation between P_1 and P_2 is |
|
Answer» `P_1 : P_2 = 7 :4` |
|
| 4. |
Whichof thefollowingalkalineearthmetaldoesNOTreactwithwater? |
|
Answer» Be |
|
| 5. |
Zinc powder +PH-OH rarr X. In the above reaction the product X will be |
|
Answer» Benzaldehyde |
|
| 6. |
Which of the following species is electron defficient |
|
Answer» CARBONIUM ion |
|
| 7. |
Which is not a pollutant from exhaust of motor vehicle? |
|
Answer» Hydrocarbns |
|
| 8. |
Which one of the following alkaline earth metal sulphates has its hydration enthalpy greater than lattice enthalpy - |
|
Answer» `BaSO_4` |
|
| 9. |
Which of the following is ionic |
|
Answer» `AlF_3` |
|
| 10. |
Which of the following does not react with diborane |
|
Answer» `Cl_(2)` |
|
| 11. |
When DeltaH gt 0 andDeltaS lt 0, reaction is never spontaneous. Explain . |
| Answer» Solution :When `DeltaH GT 0`, energy FACTOR opposes the reaction. When `Delta S lt 0 `, i.e.,RANDOMNESS decreases, this factor also opposes the process. Asboth factors OPPOSE the process, reaction is never spontaneous. | |
| 12. |
Which one of the following statements about water is FALSE ? |
|
Answer» Ice FORMED by heavy water sinks in normal water. |
|
| 13. |
The vapour pressure of is higher than due to |
|
Answer» Dipole moment |
|
| 14. |
What are nucleophiles and electrophiles. Explain with examples. |
| Answer» SOLUTION :For ANSWER, CONSULT SECTION 12.30 | |
| 15. |
The statement(s) not correct about lithium is (are) |
|
Answer» Lithium is least reactive of all ALKALI METALS. |
|
| 16. |
Which of the following is a p-type semiconductor ? |
|
Answer» Ge doped with In |
|
| 17. |
When a mixture of methyl iodide, ethyl iodide are heated with sodium in dry ether solvent, the alkane(s) obtained |
|
Answer» Ethane |
|
| 18. |
Which of the following reaction(s) is/are involved in thermit process ? |
|
Answer» `3Mn_(3)O_(4) +8Al RARR +9Mn+4Al_(2)O_(3)` |
|
| 19. |
answer any one question : (ii) find the equation of the plane passing through the intersection of the planes 2x+y+2z=9 and 4x-5y-4z=1and through the point (3,2,-1). |
|
Answer» d |
|
| 20. |
Which of the following process has negative value of DeltaS? |
|
Answer» Dissolution of SUGAR in water |
|
| 21. |
The ZnS zinc blende structure is cubic. The unit cell may be described as a face-centered sulfide ion sublattice with zinc ions in the centers of alternating minicubes made by partitioning the main cube into 8 equal parts (as shown in fig) (a) How many nearest neighbors does each Zn^(2+) have ? (b) How many nearest neighbors does each S^(2-) have? (c) what angle is made by the line connecting any Zn^(2+) to any two of its nearest neighbors ? (d) What minimum r_(+)//r_(-) ratio is needed to avoid anion -anion contact, if closest cation-anion pairs are assumed to touch ? |
|
Answer» C.N. of `Zn^(2+) " & " S^(2-) = 4 " & " 4` (b) Similarly `S^(2-)` ion have coordination number = 4 (c) As `Zn^(2+)` ion is present in tetrahedral void that's why line's connecting any two NEAREST NEIGHBOUR and `Zn^(2+)` have angle `= 109^(@)28` (d) For tetrahedral VOIDS radius ratio is `(r_(Zn^(2+)))/(r_(S^(2-))) = 0.225` |
|
| 23. |
What is the oxidation number of sulphur in the following molecules/ ions H_2S |
|
Answer» SOLUTION :The OXIDATION NUMBER of Hydrogen is +1, and s=x then, Let oxidation number = in `H_2S`, i.e., `+(1 times2) +x=0 implies 2+ x=0 therefore x=-2` Oxidation number of S in `H_2S` is -2. |
|
| 24. |
Which of the following is not Decomposition reactions |
|
Answer» `2HgO rarr 2Hg+O_2` |
|
| 25. |
Which is not an example of basic refractory material ? |
|
Answer» CALCINED limestone |
|
| 26. |
Which one of the following reactions represents the oxidizing property of H_2O_2 ? |
|
Answer» `2KMnO_4 + 2H_2SO_4 + 5H_2O_2 to K_2SO_4 + 2MnSO_4 + 8H_2O + 5O_2` |
|
| 27. |
Which of the following does not indicate high level of pollutants or the toxic substances I) High DO value II) High COD value Ill) High BOO value IV) High TLV The correct combination is |
|
Answer» All are CORRECT |
|
| 28. |
Write the formulae of the first 5 members of homologous series starting with the underlined H-COOH |
|
Answer» |
|
| 29. |
The volume strength of 1N solution of H_(2)O_(2) |
|
Answer» 11.2 V |
|
| 30. |
Why alkaline earth metals have higher tendency to form complexes than alkali metals? |
|
Answer» SMALLER size |
|
| 31. |
What are theatomicnumbersof elementswhoseoutermostelectronare representedby (a )3s^(1) (b ) 2p^(3)and (c ) 3p^(5) ? |
|
Answer» Solution :(a ) Valanceorbit`3s^(1)` so`VEC( e)`outermostconfigurationis `1S^(2) 2S^(2)2p^(6)3s^(1)` (b )Valanceorbitis `2p^(3) ` so `vec(e )` configurationis `1s^(2) 2s^(2) 3p^(3)` (c )Valanceorbitis `3p^(5)` so `vec( e)`configurationis`1s^(2)2s^(2)2p^(6)3s^(2) 3p^(5)` Totalelectron=2 + 2 +6+ 2 + 5=17 Atomicnumber =17 |
|
| 32. |
Which sequence of reaction show chemical relation between sodium and its compound |
|
Answer» `NA + O_(2)rarr Na_(2)O overset(HCI_(aq))rarr NaCl overset(CO_(2))rarr Na__(2)CO_(3)overset(TRIANGLE)rarrNa` |
|
| 33. |
What is the effect of the following processes on the bond order in N_(2) and O_(2) ? (A) N_(2) rarr N_(2)^(+) + e^(-) "(B) " O_(2) rarr O_(2)^(+) + e^(-) |
|
Answer» Solution :According to molecular orbital theory, electronic configurations and bond order of `N_(2) , N_(2)^(+) , O_(2), and O_(2)^(+)` species as follows `N_(2) (14 e^(-)) = SIGMA 1s^(2), overset(**)(sigma) 1s^(2) , sigma 2s^(2) , overset(**)(sigma) 2s^(2), (pi 2p_(x)^(2) = pi 2p_(y)^(2)),sigma 2p_(z)^(2)` Bond order = `(1)/(2) [ N_(b) - N_(a) ]= (1)/(2) (10 - 4) = 3 ` `N_(2)^(+) ( 13 e^(-)) = sigma 1s^(2),overset(*)(sigma) 1s^(2) , sigma 2s^(2), overset(**)(sigma) 2s^(2), (pi 2p_(x)^(2) = pi 2p_(y)^(2)), sigma 2p_(z)^(1)` Bond order = `(1)/(2) [ N_(b) - N_(a)] = (1)/(2) (9 - 4) = 2.5 ` `O_(2) (16 e^(-))= sigma 1s^(2) , overset(**)(sigma) 1s^(2), sigma 2s^(2) , overset(**)(sigma) 2s^(2), sigma 2p_(z)^(2), (pi 2p_(x)^(2) = pi 2p_(y)^(2)), (overset(**)(pi) 2p_(x)^(1) = overset(**)(pi) 2p_(y)^(1))` Bond order = `(1)/(2) [ N_(b) - N_(a) ]= (1)/(2) (10 - 6) = 2 ` `O_(2)^(+) (15 e^(-) ) = sigma 1s^(2) , overset(**)(sigma), sigma 2s^(2) , overset(**)(sigma) 2s^(2), sigma 2p_(z)^(2) , (pi 2p_(x)^(2), = pi 2p_(y)^(2)), (overset(**)(pi) 2p_(x)^(1) = overset(**)(pi ) 2p_(y))` Bond order = `(1)/(2) [N_(b) - N_(a)] = (1)/(2) (10 - 5) = 2.5 ` (a) `N_(2) rarr N_(2)^(+) + e^(-)` B.O = 3 B.O. = 2.5 Thus, bond order decreases. (b)` O_(2) rarr "" O_(2)^(+) + e^(-)` B.O. = 3B.O.= 2.5 Thus, bond order increases. |
|
| 34. |
Tin reacts with |
|
Answer» Hot con. HCl `Sn+4HNO_(3)toSnO_(2)+4NO_(2)+2H_(2)O` `Sn+2HgCl_(2)toSnCl_(2)+HgCl_(2)` |
|
| 35. |
You are given the electronic configuration of five neutral atoms - AB, B, C, D and EA - 1s^(2) 2s^(2) 2p^(6) 3s^(2), B - 1s^(2), 2s^(2) 2p^(6) 3s^(1), C - 1s^(2) 2s^(2) 2p^(1) , D- 1s^(2) 2s^(2) 2p^(5), E - 1s^(2) 2s^(2) 2p^(6) Write the empirical formula for the substance containing (i) A and D (ii) B and D (iii) only D (iv) onlyE ? |
|
Answer» Solution : (i) Empirical formula of the compound formed by A and D `AD_(2)`as A has two valence electronsand D has seven. ATOM A transfers its two electrons to two D atoms to complete their octets . (II) Empirical formula of the compound formed by B and D is BD as B transfers its one electron to D. (iii) `D_(2)` as both the atoms of D share one electron each to form a covelent bond (iv) Since ITIS noble gas , nocompound is formed . |
|
| 36. |
Two electrons occupying the same orbital are distinguished by |
|
Answer» SPIN QUANTUM number |
|
| 38. |
The reason of hung no. of organic compound is…… |
|
Answer» VALENCY of carbon |
|
| 39. |
Zinc sulphate crystals contals contain 43.9% water and 22.6% zinc. What is the weight of water that 13.7g of zinc sulphate crystals contain? |
|
Answer» |
|
| 40. |
Vapour pressure of a pure liquid does not depend upon |
|
Answer» SURFACE area |
|
| 41. |
The weight of 1 mole of a gas of density 0.1784 g/L at NTP is |
|
Answer» 0.1748 G |
|
| 42. |
Which of the following is correct with respect to .p. orbitals ? |
|
Answer» <P>Spherical |
|
| 43. |
There is N and S both are present in Lassaigne's extraction then what is the reason to form red colour? |
|
Answer» FERRIC thiocynate |
|
| 44. |
Write the complete chemical equation for the following . Bromination of alkene |
| Answer» Solution :`{:(underset (" ETHYLENE")(H_(2)C )= CH_(2) + Br _(2) to H_(2),underset (Br) underset (|)(C)- underset (Br) underset (|)(C)H_(2)),(,"1,2- Dibromoethene"):}` | |
| 45. |
Which can show aromatic character ? |
|
Answer» FURAN |
|
| 46. |
The structure and hybridisation of Si(CH_(3))_(4)is , |
|
Answer» BENT, sp |
|
| 47. |
Which pair will show common ion effect ? |
|
Answer» `BaCl_2+Ba( NO_3)_2` |
|
| 48. |
Which of the following indicates RMS velocity of a gas? a) sqrt((2RT)/(M)) "" b) sqrt((3P)/(d)) "" c) sqrt((8RT)/(pi M)) "" d) sqrt((3RT)/(M)) |
|
Answer» C, d |
|
| 49. |
Write the name of the following compounds : |
|
Answer» Solution :(i) 3-Methylhexane (ii) Cis-but-2-ene (iii) PROPANOIC enhydride (iv) 5,6-Diethyl-3-methyldec-4-ene (v) 4-(1-methylethyl)-oct-2-ene (VI) 2,3-Dimethylbuta-1,3-diene (vii) 3-Methylhexa-1,3,5-triene (viii) 2-Ethylbut-1-ene (ix) 4-Hydroxy-4-methylpentan-2-one (x) 3,3,5-Trimethylhex-1-en-2-ol (xi) 3-Methylcyclopentan-1-ol (xii) 3-Ethyl-4-methylpentanal (xiii) 7-Ethyl-6-methylundec-6-enoic acid (xiv) 1-(2-chlorophenyl)-2-(4-chlorophenyl)ethane (xv) 1-(2-methylcyclohexy)-4-nitrobenzene (XVI) 1-Bromobut-2-ene (xvii) 2-Methylcyclohexanone. |
|
| 50. |
Which one of the following compounds will show geometrical isomerism ? |
|
Answer» A. 2-Butene |
|



