Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which are correct statement ? |
|
Answer» `CH_3cOONH_4` have greater degree of hydrolysis in 0.2M solution in comparison of 0.4 M solution (b)anions come fromacids which are more acidic than `H_2O` undergo hydrolysis. (C ) AMONG two acids , anioncomes fromweaker acid (LESS `K_a) ` undergo more hydrolysis. (d)` HSO_4^(-) `comes fromstronger acid `H_2SO_4` . so it does not undergo hydrolysis. |
|
| 2. |
To decrease the volume of a gas by 5 tiumes at constant temperature, pressure should be …………. . |
|
Answer» DECREASED by 5 TIMES |
|
| 3. |
What are isoelectronic ions ? Give examples. |
|
Answer» SOLUTION :Ions of different elements having the same number of electrons are called iso electronic ions. De- Broglie combined the following two equations of ENERGY of which one represents wave CHARACTER(hu)and the other represents the particle NATURE `(mc^(2))` . |
|
| 4. |
Which metal is protected by a layer of its own oxide? |
|
Answer» AL |
|
| 5. |
Uncertainity in the position of an electron (mass = 9.1 xx 10^(-31) kg) moving with a velocity 300 ms^(-1) accurate upto 0.001% will be (h = 6.63 xx 10^(-34) Js) |
|
Answer» |
|
| 6. |
What will be the strength of 20 vol of H_(2)O_(2) in terms of gram per litre? |
|
Answer» `60.71 GL^(-1)` 1 L of this `H_(2)O_(2)` will give 20 L of oxygen at sTP. `underset(2xx34)(2H_(2)O_(2))to underset(22.4" L at STP")(O_(2))+H_(2)O` 22.4 L of `O_(2)` is PRODUCED from 68 g of `H_(2)O_(2)` 20 L of `O_(2)` is produced from `=(68xx20)/(22.4)=60.71 g L^(-1)`of `H_(2)O_(2)`. |
|
| 7. |
Which of the following is a reducing agent and undergoes hydrolysis ? |
| Answer» Answer :D | |
| 8. |
Which of the following is an example for polyhalo compounds? |
|
Answer» Vinyl IODIDE |
|
| 9. |
The salt which is added to table salt to keep it dry and free flowing is - |
|
Answer» KCL |
|
| 10. |
Which one of the following pairs of atoms/ions have identical ground state configuration ? |
|
Answer» `Li^(+)` and `He^(+)` |
|
| 11. |
When HCl gas is passed through a saturated solution of common salt, pure NaCl is precipitated because |
|
Answer» HCL is HIGHLY ionised in solution |
|
| 12. |
Which one is not included in branch of chemistry ? |
|
Answer» ORGANIC |
|
| 13. |
What weight of slaked lime will be required to decompose completely 4 grams ammonium chloride |
|
Answer» `2.77g` |
|
| 14. |
The undesirable material that enters into the environment by natural source or due to human activity is called _______ |
|
Answer» |
|
| 15. |
Why are potassium and caesium rather than lithium used in photoelectric cells ? |
| Answer» Solution :The IONISATION enthalpy of lithium `(520 kJ mol^(-1))` is much higher than those of POTASSIUM `(419 kJ mol^(-1))`and caesium `(376 kJ mol^(-1))`. Due to high ionisation enthalpy, lithium does not emit electrons on EXPOSURE to light. On the other hand, potassium and caesium do so. Therefore, K and CS rather than Li are used in photoelectric cells. | |
| 16. |
Which of the following cannot give precipitate with cuprous ammonium chloride? |
|
Answer» `CH_(3)-C-=C-CH_(3)` |
|
| 17. |
Whenphenol reqacts with Zn dust under dry distillationconditions it gives ………………. |
| Answer» SOLUTION :BENZENE | |
| 19. |
The recommended concentration of fluoride ion in drinking water is up to 1 ppm as fluoride ion is required to make teeth enamel harder by converting [3Ca(PO_4)2. Ca(OH)_2] to : |
|
Answer» `[CaF_2]` |
|
| 20. |
Which of the following is not an electromagnetic radiation ? |
|
Answer» Gamma RAYS |
|
| 21. |
Which of the following cannot be regarded as molecular solid ? |
|
Answer» SIC ( Silicon carbide ) |
|
| 22. |
What indicate these is solid dash model of organic molecule? |
Answer» SOLUTION :In these FORMULA, the solid WEDGE  is used to indicate a BOND PROJECTING out of the plane of paper, towards the observer. The dashed wedge is used to indicate a BOND PROJECTING out of the plane of paper, towards the observer. The dashed wedge  is used to depict the bond projecting out of the plane of the paper and away from the observer is used to depict the bond projecting out of the plane of the paper and away from the observer
|
|
| 23. |
Which is the IUPAC name of |
|
Answer» 4-ethyl-2-methylaniline |
|
| 24. |
What is the effect on equilibrium and on the value of equilibrium constant on adding catalyst ? |
| Answer» Solution :No EFFECT on EQUILIBRIUM constant. However, forward and backward reactions become faster to the same EXTENT and equilibrium is attained QUICKLY. | |
| 25. |
There is change in volume of an ideal gas when pressure is changed at constant temperature. The change in free energy associated with this can be calculated by which of the following equation ? |
|
Answer» `Delta G = RT "In" (P_2)/( P_1)` |
|
| 26. |
Which type of reaction occurred between H_(2)SO_(4)-HNO_(3) in nitration ? |
| Answer» Solution :Acid-BASE TYPE of reaction in which `H_(2)SO_(4)` protn donor acid and `HNO_(3)` PROTON acceptor base. | |
| 27. |
When two atoms of chlorine combine to form one molecule of chlorine gas, the energy of the molecule is |
|
Answer» GREATER than that of SEPARATE atoms |
|
| 28. |
What is the characteristic of R_(f) value ? |
| Answer» Solution :In EVERY compound, the `R_(F)` VALUE is definite and constant. So, the `R_(f)` value give identification of compound LIKE other consant value | |
| 29. |
Which of thefollowingelementswill gainoneelectronmore readily comparison to other elements of theirgroup ? |
|
Answer» S (g ) |
|
| 30. |
Write the bond -line formula for CH_(3)underset(CH_(3))underset(|)(CHCH_(2))CH_(2)CH_(2)OH |
Answer» Solution :Firstfor the sake of practice , we outline the CARBON skeleton , including the OH group ,as follows :  Then we write the bond -LINE formula as  As you gain EXPERIENCE you will likely skip the intermediate STEPS shown above and proceed directly to writing bond - line formulas . |
|
| 31. |
Which of these elements have almost same electronegativity? (i) C,I (ii) P, H (iii) N, Cl (iv) Br,S |
|
Answer» |
|
| 32. |
Write short notes on ortho, para directors in aromatic electrophillic substitution reactions. |
| Answer» Solution :`{:(A,-,CH_(2)=CH_(2),-,,"ETHENE"),(B,-,UNDERSET(Cl)underset(|)(C )H_(2)-underset(Cl)underset(|)(C )H_(2),-,1,2-"dichloro ethane"),(C,-,HCHO,,-,"formaldehyde"),(D,-,CH-=CH,-,,"ACETYLENE"):}` | |
| 33. |
Which islargest in size Cu^(+),Cu^(2+)or Cu and why ? |
|
Answer» |
|
| 34. |
The sigma bond formed between two carbon of ethene is result of overlapping of ……….. hybridized orbital. |
| Answer» SOLUTION :`SP^(2) - sp^(2)` | |
| 36. |
Which gases are responsible for greenhouse effect ? List some of them. |
| Answer» Solution : `CO_2`is the main GAS responsible for green-house effect. Other greenhouse gases are methane, nitrous oxide, water VAPOURS. chlorofluorocarbons (CFC's) and OZONE. | |
| 37. |
van der Waals' constant 'b' for oxygen is 32 cm^(3)//mol. Assume b is four times the actual volume of a mole of ''billiard-ball'' O_(2) molecules and compute the diameter of an O_(2) molecule. |
| Answer» | |
| 38. |
What are conjugate acid-base pairs? Give one example. |
|
Answer» SOLUTION :ACID - base PAIRS which differ by ONE proton are called conjugate acid - base pair. `NH_(3)+H_(2)OtoNH_(4)^(+)+OH^(-)` Conjugate base of acid `H_(2)O" is "OH^(-),H_(2)O+H_(2)OtoH_(3)O^(+)+OH^(-)` Conjugate acid of base `NH_(3)" is "NH_(4)^(+),NH_(3)+NH_(3)toNH_(4)^(+)+NH_(2)^(-)`. |
|
| 39. |
What is milk of lime? How CO_(2) reacts with it? |
|
Answer» Solution :The aqueous solution of CALCIUM hydroxide is known as LIME water and a SUSPENSION of slaked lime in water is known as MILK of lime. When carbon dioxide is passed through lime water, it turns milky due to the formation of calcium carbonate `Ca(OH)_(2) +CO_(2) to CaCO_(3) + H_(2)O` |
|
| 40. |
Which of the following acts as Lewis acid ? |
|
Answer» H |
|
| 41. |
What is Z in the following reaction? CH_(3)-CH_(2)-CO_(2)^(Theta)Na^(oplus)underset(Delta)overset(NaOH//CaO)rarrZ |
|
Answer» propane |
|
| 42. |
What is an ionic bond ? |
| Answer» Solution :An ionic BOND is the electrostatic force of attraction between oppositely charged ions. Or A chemical bond FORMED by COMPLETE transter of one or more electrons from outermost orbit of ELECTROPOSITIVE atom to OUTER most orbit of electronegative atom. | |
| 43. |
Which of the following use for mesurement of mass of D_2 and ND_3 ? |
|
Answer» Ferometer |
|
| 44. |
Vapour pressure increases with increase in : |
|
Answer» CONCENTRATION of solution CONTAINING non-volatile solute |
|
| 45. |
Which one of the following is a benzenoid aromatic compound ? |
|
Answer» Furan |
|
| 46. |
Which one is the most stable carbonium ion? |
|
Answer» `F_(3)C- CH_(2)^(+)` |
|
| 47. |
Which of the following are correct ? |
|
Answer» `CH_3-O-CH_2^(o+)` is more stable than `CH_3-CH_2^(o+)`  `CH_(3)O overset(+)(C)H_(2)` is stabilized by +I effect of the two `CH_(3)` groups while 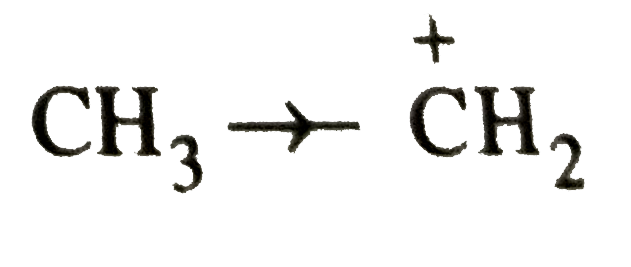 is stabilized by only weak +I- effect of the `CH_3` GROUP. Thus, option (a) is CORRECT.  is stabilized by +I-effect of the ethyl group. SINCE +I-effect of the ethyl group is weaker than +I-effect of the two `CH_3` groups, therefore, `(CH_3)_2CH^+` is more stable than `CH_3CH_2-overset+CH_2` group. Thus , option (b ) is wrong. is stabilized by +I-effect of the ethyl group. SINCE +I-effect of the ethyl group is weaker than +I-effect of the two `CH_3` groups, therefore, `(CH_3)_2CH^+` is more stable than `CH_3CH_2-overset+CH_2` group. Thus , option (b ) is wrong. is stabilized by strong resonance effect while, is stabilized by strong resonance effect while, 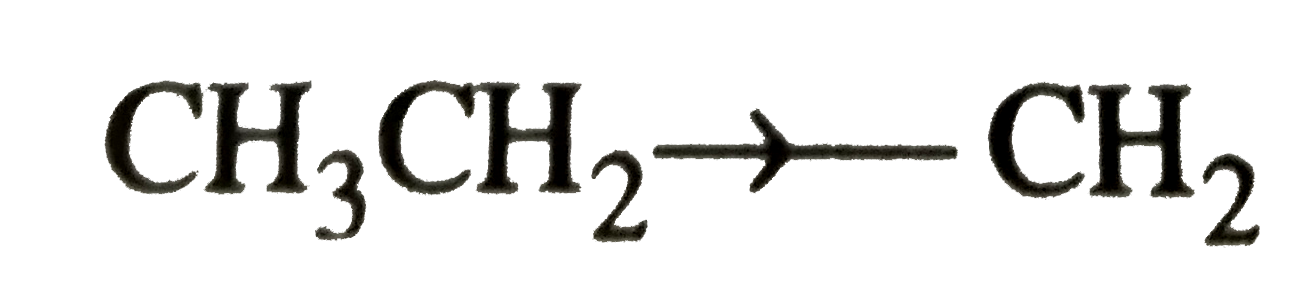 is stabilized by weak +I-effect of the `CH_2CH_3` group. The option (c ) is correct. In `CH_2=CH^+` , +ve charge is present on the more electronegative sp-hybridised carbon while in `CH_3-overset+CH_2`, +ve charge is present on the more electronegative sp-hybridized carbon while in `CH_3-overset+CH_2`. , +ve charge is present on the less electronegative `sp^2`-hybridized carbon, therefore , `CH_2=overset+CH` is less stable than `CH_3-overset+CH_2` . Thus option (d) is wrong. is stabilized by weak +I-effect of the `CH_2CH_3` group. The option (c ) is correct. In `CH_2=CH^+` , +ve charge is present on the more electronegative sp-hybridised carbon while in `CH_3-overset+CH_2`, +ve charge is present on the more electronegative sp-hybridized carbon while in `CH_3-overset+CH_2`. , +ve charge is present on the less electronegative `sp^2`-hybridized carbon, therefore , `CH_2=overset+CH` is less stable than `CH_3-overset+CH_2` . Thus option (d) is wrong.
|
|
| 48. |
What changes in pressure is sufficient to increase the products of this C_((s)) + CO_(2(g)) hArr 2CO_((g))reaction ? |
| Answer» Solution :Here, in this forward REACTION, the gaseous mole INCREASE (2 out of 1 mole). So if the pressure DECREASE than the reaction take place in such direction where the MOLES of gas increases. | |
| 49. |
Which of the following posses 1^(@), 2^(@), 3^(@) and 4^(@) carbon? |
|
Answer» 2, 3-dimethyl hexane |
|
| 50. |
Two beaker A and B present in a closed vessel. Beaker A contains 152.4 g aqueous soulution of urea, containing 12 g of urea. Beaker B contain 196.2 gglucose solution, containing 18 g of glocose. Both solution allowed to attain the equilibrium. Determine mass % of glocose in its solution at equilibrium allowed to attain the equilibrium : |
|
Answer» 6.71 |
|



