Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
What is meant by the term averge bond enthalpy ? Why is there difference in bond enthalpy of O-H bond in ethanol (C_(2)H_(5)OH) and water ? |
|
Answer» Solution :All the similar bonds in a molecule do not have the same bond enthaipies ,e.g., in`CH_(4) (H-underset(H)underset(|)overset(H)overset(|)C-H)`, the four C-H bonds do not have the same bond enthapies because after breaking of the bonds one by one, the electronic environmentaroundcarbon changes . Hence, the actual bond enthalpy of C-H bond is TAKEN as the averae value. O-H bond in ethanol 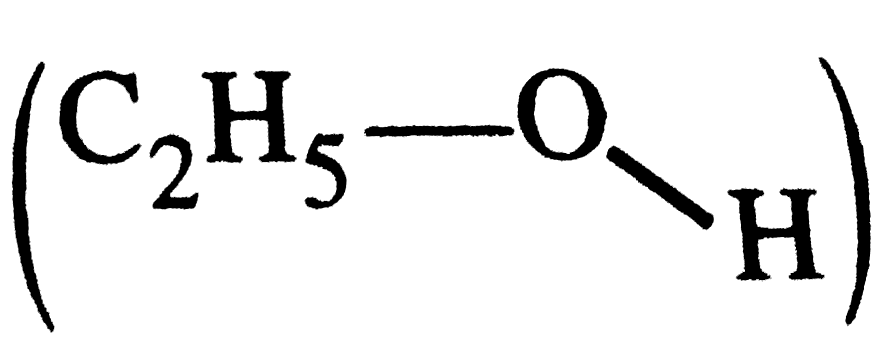 and that in WATER and that in WATER 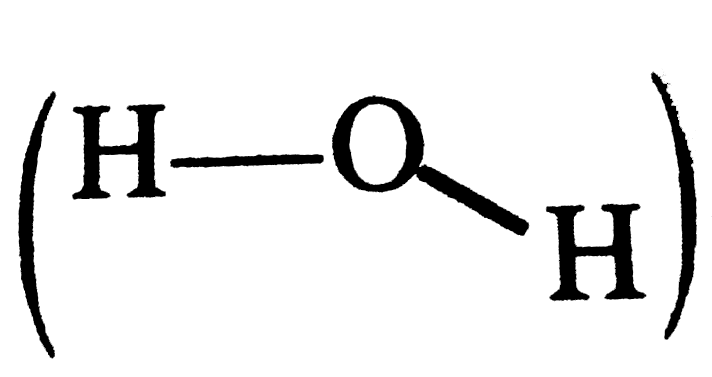 do nothave similar electronic do nothave similar electronicenviroment around oxygen atom. Hence, their O-H bond enthalpies are DIFFERENT |
|
| 3. |
Which of the following reactions due to the reaction gives - |
|
Answer» `2CuSO_(4)to4KtoCu_(2)I_(2)+2K_(2)SO_(4)+I_(2)` |
|
| 4. |
The self ionization constant for pure formic acid, K = [HCOOH_(2^(+))][HCOO^(-)] has been estimated as 10^(-6) a room temperature. What percentage of formic acid molecules in pure formic acid are converted to formate ion? The density of formic acid is 1.22 g//cm^(3). |
|
Answer» `0.037` weight of fromic acid in 1litre solution `= 1.22' 10^(3) g` `:' [HCOOH]= (1.22 XX 10^(3))/(46) = 26.5 M` `rArr K = [HCOOH_(2)^(+)] = 10^(-6)` `:' [HCOOH_(2)^(+)] = [HCOO^(-)]` `rArr [HCOOH]_("dissociated") = 10^(-3)` `:. (10^(-3))/(26.5) xx 100 = 0.0037` |
|
| 5. |
What happens when magnesium is burnt in air ? |
|
Answer» Solution :Magnesium burns in air with a dazzling light to form MGO and `Mg_(3)N_(2)`. `2Mg+O_(2) overset("on heating") to 2MgO` `3Mg+N_(2) overset("on heating")to Mg_(3)N_(2)` |
|
| 6. |
When the electron of a hydrogen atom jumps from n = 4 to n = 1 state, the number of spectral lines emitted is |
|
Answer» 15 |
|
| 7. |
Which of the following is true about the element with atomic number 18 ? |
|
Answer» It has a very LOW IONISATION POTENTIAL |
|
| 8. |
Which of the following compounds are exhibit tautomerism? |
|
Answer» |
|
| 9. |
The volume of CO_(2) liberated at S.T.P and the quantity of heat energy liberated from the combustion of 2.3 g of ethyl alcohol are C_(2)H_(5)OH(l) +3O_(2)(g) rarr 2CO_(2)(g)+3H_(2)O(l) +1367.2 |
|
Answer» Solution :MOLECULAR weight of `C_(2)H_(5)OH = 46` 46 grams of `C_(2)H_(5)OH` on combustion liberate `2 XX 22.4` lit of `CO_(2)` at S.T.P. and 1367.2 of energy. Volume of `CO_(2)`liberated by the combustion of 2.3 g. of `C_(2)H_(5)OH` `(2.3)/(46) xx 2 xx 22.4 = 2.24` lit Energy liberated by the combustion of 2.3 g `C_(2)H_(5)OH = (2.3)/(46) xx 1367.2 = 68.36 k.j` |
|
| 10. |
Which of the following is the most stable ion ? |
|
Answer» `CH_(3) - CH_(2) - CH_(2) - overset(+)(CH_(2)` |
|
| 11. |
What would be the pH of 0.1 molar sodium acetate solution, given that the dissociation constant of acetic acid is 1.8xx10^(-5). |
|
Answer» `pH = 7 + (1)/(2) [ pK_(a)+log c] = 7 + (1)/(2) [- log (1.8xx10^(-5))+log 0.1] = 7 + (1)/(2) [ 4.76xx-1] = 8.88` |
|
| 12. |
Which of the following relations gives the value of n = |
|
Answer» `("MOLECULAR mass ")/("ATOMICMASS ")` |
|
| 13. |
Which of the following pairs of elements exhibit diagonal relationship ? |
|
Answer» Be and Mg |
|
| 14. |
Which of the following statement (s) about the Modern Periodic Table is are incorrect(i)The elements in the Modern Periodic Table are arranged on the basis of their decreasing atomic number(ii)The elements in the Modern Periodic Table are arranged on the basis of theirincreasing atomic masses(iii)Isotopes are placed in adjoining group (s) in the Periodic Table(iv) The elements in the Modern Periodic Table are arranged on the basis of their increasing atomic number |
|
Answer» (i) only |
|
| 15. |
Which of the following reactions are ot feasible? |
|
Answer» `HC-=CH+KOHto` |
|
| 16. |
What volume of 1N NaOH solution can completelyconvert a solution containing12g of NaH_(2)PO_(4) to Na_(3)PO_(4)? |
|
Answer» |
|
| 17. |
Which of the following correct regarding the -I effect of the substituents ? |
|
Answer» `-NR_(2) lt -OR lt -F` |
|
| 18. |
Which of the following alkaline earth metal does not react with cold water but water |
|
Answer» Mg |
|
| 19. |
When a substance A reacts with water, it produces a combustible gas B and s solution of substance C in water. When another substance D reacts with this solution of C, it produces the same gas B on warming but D can produces gas B on reaction with dilute sulphuric acid at room temperature . A imparts a deep golden yellow colour to a smokeless flame of Bunsen. A,B,C and D respectively are: |
|
Answer» `NA,H_(2),NAOH, Zn` Since Na gives golden yellow flame, K gives pink violet flame while Ca gives BRICK red flame, therefore, option (a) is correct. |
|
| 20. |
Where does electronic configuration takes place in an atom ? |
|
Answer» <P> SOLUTION :In ORBITALS LIKE s, p, d, F |
|
| 21. |
Which among these is not associated with aliphatic compounds |
|
Answer» theycontain (4n+2) electrons |
|
| 22. |
The sharp melting point of crystalline solids is due to …….. |
|
Answer» a REGULAR ARRANGEMENT of consitituent particles observed over a short DISTANCE in the crystal lattice . |
|
| 23. |
What is the composition of Zeigler Natta Catalyst? |
| Answer» SOLUTION :Tiethylaluminium and TITANIUM TETRACHLORIDE INERT SOLVENT. | |
| 24. |
What is the effect of pressure on gas dissolve in liquid ? |
| Answer» SOLUTION :As PRESSURE INCREASE SOLUBILITY increase. | |
| 25. |
Write the IUPAC names of (i) CH_(2) = CHCl (ii) CH_(2) = CH- CH_(2) Br |
|
Answer» Solution :`(i) CH_(2) = CHCL` : CHLOROETHENE (ii) `CH_(2) = CH - CH_(2) Br : 3`- BROMO- prop-1-ene |
|
| 26. |
Using s, p, d, f notations, describe the orbital with the following quantum numbers (a) n=2, l = 1 (b) n = 4, l = 0 (c) n = 5, l = 3 (d) n = 3,l = 2. |
Answer» SOLUTION :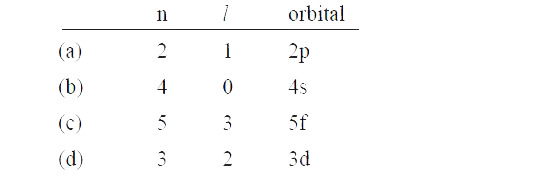
|
|
| 27. |
Which of the following compound is/are react with Na metal & liberate hydrogen gas |
|
Answer» `CH_(3)-OH` |
|
| 28. |
Which can be purified by crystalization ? |
|
Answer» PHENOL |
|
| 29. |
What is the shape of methane molecule? |
|
Answer» Squareplaner |
|
| 30. |
What will be the molality of the solution made by dissolving 10g of NaOH in 100g of water? |
|
Answer» 2.5 m |
|
| 31. |
Which of the following graphs is correct for a reaction? |
|
Answer»
`:.` exothermic `implies` ,+, ve slope ENDOTHERMIC `implies` .-. ve slope |
|
| 32. |
Which of the following reagents are used to give blue ring test of solution of Na_(2)S_(2)O_(3)? |
|
Answer» `(NH_(4))_(2)MoO_(4)` |
|
| 33. |
What will be the conjugate bases for the Bronsted acids :HF, H_2SO_4 and HCO_3^- |
| Answer» SOLUTION :CONJUGATE BASE : `F^(-) , HSO_4^(-)` and `CO_3^(2-)` | |
| 34. |
Write the dimeric structure of Aluminium chloride. |
Answer» SOLUTION : 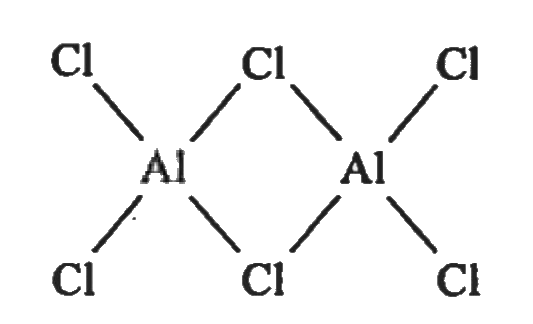
|
|
| 35. |
What is the relation between standard free energy change and equilibrium constant ? |
|
Answer» `DeltaG^(@)=+"RT LN K"` |
|
| 36. |
Which of the following pair contains with same structures. |
|
Answer» Borazine & BENZENE |
|
| 37. |
Which of the following metals are not detected by flame test? |
|
Answer» Be and MG |
|
| 38. |
Which of the following is environmental friendly reaction |
|
Answer» A + B `RARR` C (wanted) |
|
| 39. |
What happen when diborane is heated with ammonia ? |
| Answer» | |
| 40. |
What weight of non-volatile solute (urea) NH_(2) CO NH_(2) needs to be dissolved in 100 g of water in order to decrease the vapour pressure of water by 25%. What will be the molality of solution? |
|
Answer» |
|
| 41. |
Which of the following dienophiles is the most reactive in normal Diels-Alder reactions? |
|
Answer»
|
|
| 42. |
Which of the group given below is most appropriate for the hydride compounds ? |
|
Answer» s-block - METALLIC hydride - `TiH_2` |
|
| 44. |
Which of the following is a primary pollutant? |
|
Answer» Sulphuric ACID |
|
| 45. |
The stablest radical among the following is |
|
Answer» `C_(6)H_(5)-OVERSET(*)(C)H-CH_(3)` |
|
| 46. |
Write the IUPAC names of the given poly functiona compounds (a) HOCH_(2)CH_(2)CH_(2)COOH (b) (CH_(3))_(2) CH overset(overset(O)(||))(C )CH_(2)COOCH_(3) (c ) CH_(3)CH(NH_(2))CH_(2)CH_(2)CH(OH)CH_(2)CH_(3) (d) CH_(3)OCH_(2)COCH_(3) (e ) CH_(2)CHBrCH_(2)CONHCH_(2)CH_(3) |
|
Answer» (B) Methyl-4-methyl-3-oxopentanoate; (C ) 6-Amino-3-heptanol; (d) Methoxypropanone; (E ) Bromo-N-ethylbutanamide |
|
| 47. |
Which hybridization is possible with d orbitals ? And which are not possible ? Explain. |
|
Answer» Solution :The elements PRESENT in the THIRD period contain d orbitals in addition to s and p orbitals. The hybridization is possible between less energy difference containing nearer orbital. (i) The energy of the 3d orbitals are comparable to the energy of the 3s and 3p orbitals. So, hybridization involving 3s, 3p and 3d orbitals is possible. (ii) The energy of the 3d orbitals are comparable of the energy of the 4s and 4P orbitals. So, hybridization involving 3d, 4s and 4p orbitals is possible. SINCE the difference in energies 3p and 4s orbitals is SIGNIFICANT, no hybridization involving 3p, 3d and 4s orbital is possible. |
|
| 48. |
Write note on Metallic or non stoichiometric hydrides. |
|
Answer» Solution :The Metallic hydride are formed by many d -block and f-block elements. However, the metals of group 7, and 9 do not form hydride. Even from group 6, only chromium forms CrH. These hydrides conduct heat and electricity THOUGH not as efficiently as their parent metals do. Unlike SALINE hydrides, they are almost always non-stoichiometric, being deficient in hydrogen. For example : `LaH_(2.87), YbH_(2.55),TiH_(1.5-1.8),ZrH_(1.3-1.75), VH_(0.56), NiH_(0.6-0.7), PdH_(0.6-0.8)` etc. In such hydrides, the law of constant composition does not hold good. In these hydrides, hydrogen occupies interstitial SPACE in the metal lattice producing distortion without any change in its type. Consequently, they were termed as interstitial hydrides. The except for hydrides of Ni, Pd, CE and Ac, other hydrides of this class have lattice different from that of the parent metal. The PROPERTY of absorption of hydrogen on transition metals is widely used in catalytic reduction / hydrogenation reactions for the preparation of large number of compounds. Some of the metals (e.g., Pd, Pt) can accommodate a very large volume of hydrogen and, therefore, can be used as its storage media. This property has high potential for hydrogen storage and as a source of energy. |
|
| 49. |
Which one of the following is used for producing pesticides? |
|
Answer» `CHI_3` |
|
| 50. |
What is inversion temperature? |
|
Answer» SOLUTION :The TEMPERATURE below which a GAS obey Joule-Thomson effect is called inversion temperature `(T_i). TI=(2a)/(Rb)` |
|








