Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
(vii) H_(2)C_(2)O_(4) +KMnO_(4)+H_(2)SO_(4)to H_(2)SO_(4)+MnSO_(3)+CO_(2)+H_(2)O. |
Answer» Solution : Equalise the INCREASE/ decrease in O N by multiplaying Cu species by 5 and Mn species by 1 `5H_(2)C_(2)O_(4)+KMnO_(4)+H_(2)SO_(4)to K_(2)SO_(4)MnSO_(4)+5CO_(2)+H_(2)O` Balance all other atoms except H and O atoms `5H_(2)C_(2)O_(4)+2KMnO_(4)+3H_(2)SO_(4)to K_(2)SO_(4) +2MnSO_(4)+10CO_(2)+H_(2)O` Balance O ATOM by adding `H_2O` on the side falling SHORT of oxygen atoms. `5H_(2)C_(2)O_(4)+2KMnO_(4)+3H_(2)SO_(4)to K_(2)SO_(4)+2MnSO_(4)+10CO_(2)+H_(2)O+7H_(2)O` The balanced equation in `5H_(2)C_2O_(4)+2KMnO_(4)+3H_(2)SO_(4)to K_(2)SO_(4)+2MnSO_(4)+10CO_(2)+8H_(2)O`. |
|
| 2. |
Use the periodictable toanswerthe followingquestions (a)Identify anelement with fiveelectrons in the outersubshell. ( b)Identifythe elementthat wouldtend to lose twoelectrons. ( c) Identifythe elementthat wouldtend to gain twoelectrons ( d)Identifythe grouphavingmetal non - metalliquid as well as gas at room temperature |
|
Answer» Solution :(a) The generalelectronicconfigurationof the elementshavingfiveelecrons in theoutersubshell is `ns^(2) np^(5)` . Thiselectronicconfigurationis CHARACTERISTIC of element of GROUP 17, i.e.,halogensand theirexamplesare F, Cl, Br,I ,At , etc. ( b)the elements whichhave atendencyto losetwo elementmust havetwoelectrons in thevalenceshell.Thereforetheirgeneralelectronicconfigurationshouldbe `n s^(2)`. Thiselectronicconfigurationis characterisitic ofgroup 2elements i.e.,alkalineearthmetalsand theirexampleare Mg ,CA, Sr, BA, etc ( c)the elementwhichhavea tendencyto accepttwo electronsmust havesixelectronicin the valence shell. thereforetheirgeneralelectronicconfiguration is `ns^(2) np^(4)` . thiselectronicconfigurationcharacteristicofgroup16 elements and theirexampleare Oand S. ( d)A metalwhich isliquidat roomtemperatureis mercury. It istransitionmetaland belongs to group 12A non- metal whichis gas itroomtemperatureishydrogen( group 1) . nitrogen (group 15) , oxygen ( group 16) , fluorinechlorine( group 17)and inertgases( group 18) . A non- metalwhich is a liquid at roomtemperatureis bromine( group 17). |
|
| 3. |
The value of DeltaG^(@) for a reaction is 7.97 KJ//"mole". Its equilibrium constant K_(c) at 298K is 4 xx 10^(-x) What is x? |
|
Answer» `LOG K = -1.4= -2+0.6=log(4 XX 10^(-2)) IMPLIES K=2` |
|
| 5. |
Which of the following functional group can not be reduced to alcohol using sodium borohydride in the anolic solution ? |
|
Answer» RCHO |
|
| 6. |
The set representing the correct order of first ionisation potential is… |
|
Answer» `K gt Na gt Li` |
|
| 7. |
Which one of the following is true? |
|
Answer» Lithium on DIRECT combination with nitrogen to FORM `Li_3N` |
|
| 8. |
Which one is the most acidic compound ? |
|
Answer» `NiCl_2` |
|
| 9. |
The solubility of AgCl in 0.1M NaCI is (K_(sp) " of AgCl" = 1.2 xx 10^(-10)) |
|
Answer» `0.1 M ` ` "" s "" 0.1` `KSP= 5 xx 0.1 rArr S =1.2 xx 10 ^(-9) ` |
|
| 10. |
Two rigid adiabatic vessels A and B which initially, contain two gases at different temperature are connected by pipe line with value of negligible volume. The vessel 'A' contain 2 moles Ne gas (C_(p.m) = (5)/(2)R) at 300K, vessel 'B' contain 3 moles of SO_(2) gas (C_(p.m) = 4R) at 400K. The volume of A & B vessel is 4 and 6 litre respectively. The final total pressure (in atm) when value is opened and 12 Kcal heat supplied through it to vessels. [Use: R= 2 cal/mol, K and R= 0.08L. atm/mol K as per desire] |
|
Answer» 3.5 atm `2 (3)/(2) R (T - 300) = 3 xx 3R (400-T)` On solving T= 375 Again temperature will be INCREASES to `T^(1)` due to the supply of 12K. Cal of heat as follows: `2 xx (3)/(2) R (T^(1) - 375) + 3 xx 3R (T^(1) - 375) = 12000` On solving `T^(1) = 875` So, `P = (5 xx 0.08 xx 875)/(10) = 35` atm |
|
| 11. |
Which of the following is correct representatin of the reaction when BF_(3) reacts with ammonia? (i) F - underset(F)underset(|)overset(F)overset(|)(B) + underset(H)underset(|)overset(H)overset(|)( :N) - H to [F - underset(F)underset(|)overset(F)overset(|)(B) to underset(H)underset(|)overset(H)overset(|)( :N) - H] (ii) F - underset(F)underset(|)overset(F)overset(|)(B) + underset(H)underset(|)overset(H)overset(|)( :N) - H to [F - underset(F)underset(|)overset(F)overset(|)(B) to underset(H)underset(|)overset(H)overset(|)(N) - H] |
|
Answer» (i) is INCORRECT and (II) is CORRECT |
|
| 12. |
The structures of beryllium chloride in the solid state and vapur phase, respectively, are: |
|
Answer» CHAIN and chain |
|
| 13. |
What is the coordination number in a square close packed structure in htwo dimensions ? |
|
Answer» 2 |
|
| 14. |
Whch one of the following substances is used to reactivate the de activated filters in activated carbon method for defluoridation ? |
|
Answer» Defluoron -I |
|
| 15. |
What is called Bond Length? Name the techniques through which the length of a bond can be determined |
|
Answer» Solution :The distance between the nuclei of two COVALENTLY bonded atoms is called bond length, For e.g., in a covalent MOLECULE A - B, the bond length is EQUAL to the sum of the radii of bonded atoms,i,e .. `gamma_(A)+gamma_(B)` = bond lenght The length of a bond can be determined by spectroscopic, x-ray DIFFRACTION and electron- diffraction techniques |
|
| 16. |
Two bubbles of different radii are connected by a hollow tube, then |
|
Answer» SMALLER bubble gets smaller, larger gets larger |
|
| 17. |
Which of the following is independent of temperature of a gas |
| Answer» Answer :C | |
| 18. |
Which of the following statements is correct about inductive effect |
|
Answer» Implies the TRANSFER of lone pair of ELECTRONS from more ELECTRONEGATIVE atoms to lesser electronegative atom in a molecule |
|
| 19. |
Which of the following will have functional group isomerism? |
|
Answer» Methanol  `:.` FUNCTIONAL GROUP ISOMERISM |
|
| 20. |
Which has maximum molar heat of combustion? |
|
Answer» n-Butane |
|
| 21. |
The transition elements have a general electronic configuration: |
|
Answer» `ns^(2)np^(6)ND^(1-10)` |
|
| 22. |
Which of the following carbocation will be most stable ? |
|
Answer» `Ph_(3)C_(-)^(+)` |
|
| 23. |
What is the relationship betwee the eV and the wavelength in metre of the energetically equivalent photons? |
|
Answer» |
|
| 24. |
The uncertainty in the position of a moving bullet of mass 10g is 10^(-5).Calculate the uncertainty in its velocity? |
|
Answer» SOLUTION :Acooding to uncertainty PRICIPLE, `/_\x.m/_\v=h/(4PI)or /_\v=h/(4pim/_\x),h=6.626xx10^(-34)KGM^(2)kgm^(2)s^(-1),m=10g=10^(-2)kg` `/_\x=10^(-5)m,/_\v=((6.626xx10^(-34)kgm^(2)s^(-1)))/(4xx3.143xx(10^(-2)kg)xx(10^(-5),m))=5.27xx10^(-28)mv` |
|
| 25. |
Which chemistry is involed in production of chemicals without damaging the environment ? |
|
Answer» Yellow CHEMISTRY |
|
| 26. |
What effect doesbranching of an alkene chain has on its boiling point ? |
| Answer» Solution :As the branching INCREASES , the surface area of an alkeneapproaches that of a sphere. Since, a sphere has minimum surface area, therefore, VAN der Waals FORCES of attraction are minimum and HENCE the BOILING point of the alkane decreases with branching. | |
| 27. |
Which of the following order is incorrect w.r.t. property indicated ? |
|
Answer» `UNDERSET(("ACID STRENGTH"))("Formic acid " gt " Acetic acid") gt "Propionic acid"` |
|
| 28. |
Which of the following does not result in an increase in the entropy? |
|
Answer» CRYSTALLISATION of SUCROSE from SOLUTION |
|
| 29. |
Which of the following is not the consequence of H-bonding? |
|
Answer» Glycerol is more soluble in WATER than ethanol. |
|
| 30. |
What is pK_(w) ? What is its value of 25^(@)C ? |
| Answer» SOLUTION :`pK_(w) = - LOG K_(w) = - log 10^(-14) = 14`. | |
| 31. |
Which postulate of kinetic theory can be used to justfy Dalton's law of partial pressures ? |
| Answer» SOLUTION :No, TEMPERATURE is LOWERED during ADIABATIC EXPANSION. | |
| 32. |
Which one is true from the following ? |
|
Answer» `(p_(1)T_(1))/(V_(1))=(p_(2)T_(2))/(V_(2))` |
|
| 33. |
Write short note on the following bond parameter . Bond enthalpy |
| Answer» Solution :Bond enthalpy : The bond enthalpy is DEFINED as the minimum AMOUNT of energy required to BREAK one mole of a PARTICULAR bond in molecules in their gaseous state . The unit of bond enthalpy is KJ `mol^(-)`. | |
| 34. |
Which of these is least likely to act as a Lewis base ? |
|
Answer» `PF_(3)` |
|
| 35. |
What is laughing gas ? How is it prepared ? |
| Answer» SOLUTION :`N_(2)O`. It is PREPARED by HEATING ammonium nirate, i.e., `NH_(4)NO_(3)overset(Delta)toN_(2)O+2H_(2)O`. | |
| 36. |
When acidified k_(2)Cr_(2)O_(7)O_(7)spitopm os added tp Sm^(2+) salts then Sn^(2+) salt then Sn^(2+) changes to |
| Answer» ANSWER :C | |
| 37. |
Which of the following is not an non-ideal solution showing positive deviation? |
|
Answer» Benzene & acetone |
|
| 38. |
The shape of CH_(3)^(-) is......................while that of CH_(3)^(+) is................. . |
|
Answer» |
|
| 39. |
What is the role of gypsum in cement ? |
| Answer» Solution :Gypsum decreases the rate of SETTING of cement . It increase the time required for setting of cement . Various silicates presentin the form of gels in the cement are SLOWLY converted to a hard MASS in the presence of added gypsum . | |
| 40. |
Which orbitals of the following are overlapped and form sigma bond ? 1s - 1s, 2s - 2s, 1s - 2s, 2p_(x) - 2p_(x) , 2p_(y)- 2p_(y) , 2p_(z) - 2p_(z) |
|
Answer» SOLUTION :`SIGMA` -bond is FORM by FOLLOWING overlapping 1s - 1s, 2s - 2s , 1s-2s, `2p_(z) - 2p_(z) ` `sigma`-bond is not formed by overlapping of `2p_(y)- 2p_(y) and 2p_(x) - 2p_(x)`. |
|
| 41. |
Which of the following is the electron deficient molecule ? |
|
Answer» `C_(2)H_(6)` |
|
| 42. |
Write the IUPAC names of the products obtained by the ozonolysis of the following compouds : l-phenyl but-1-ene |
Answer» SOLUTION :
|
|
| 43. |
What is the formula of gypsum ? What happens when it is heated ? |
| Answer» Solution :`CaSO_(4) . 2H_(2)O` . When heated to 393 K , it GIVES Plaster to Paris `(CaSO_(4). 1//2 H_(2)O)` but at 473 K it gives DEAD burnt plaster `(CaSO_(4))` . | |
| 44. |
The trans-alkenes are formed by the reduction of alkynes with : |
|
Answer» `H_(2)-Pd//c BaSO_(4)` |
|
| 45. |
Which of the following process used in water gas shift reaction |
|
Answer» MERCK's process |
|
| 46. |
Which of the following types of reactions majorly occur when a reactant has a double bond? |
|
Answer» Addition |
|
| 47. |
Which of the following statements concerning the quantum numbers are correct ? |
|
Answer» Angular quantum num ber DETERM ines the three DIM ensional shape of th e orbital. |
|
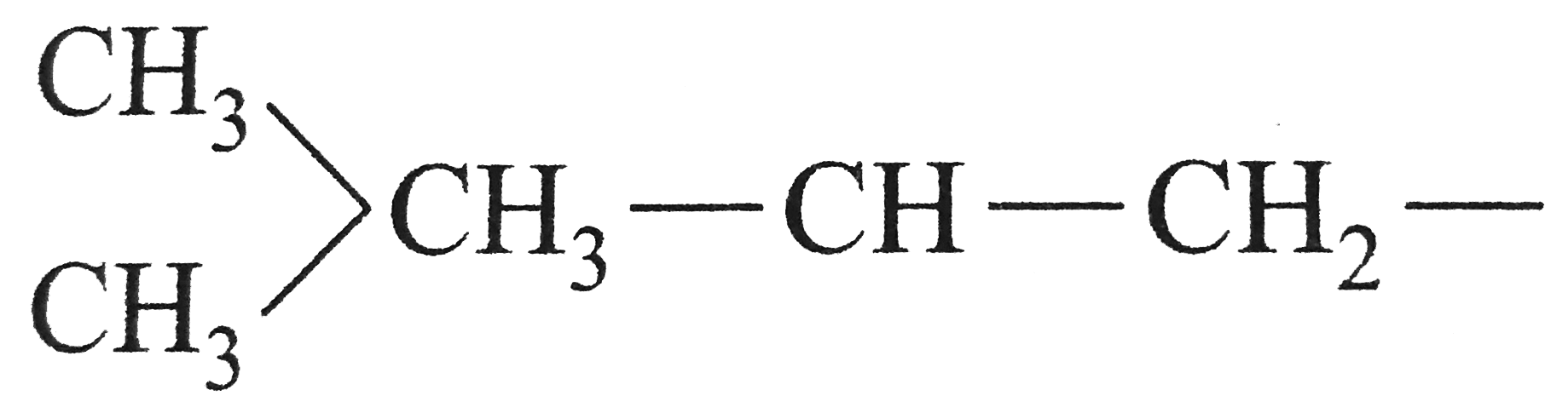
| 48. |
The structure of isobutyl group in an organic compound is |
|
Answer» `CH_(3)-UNDERSET(CH_(3))underset(|)(CH)-CH_(2)-CH_(3)` 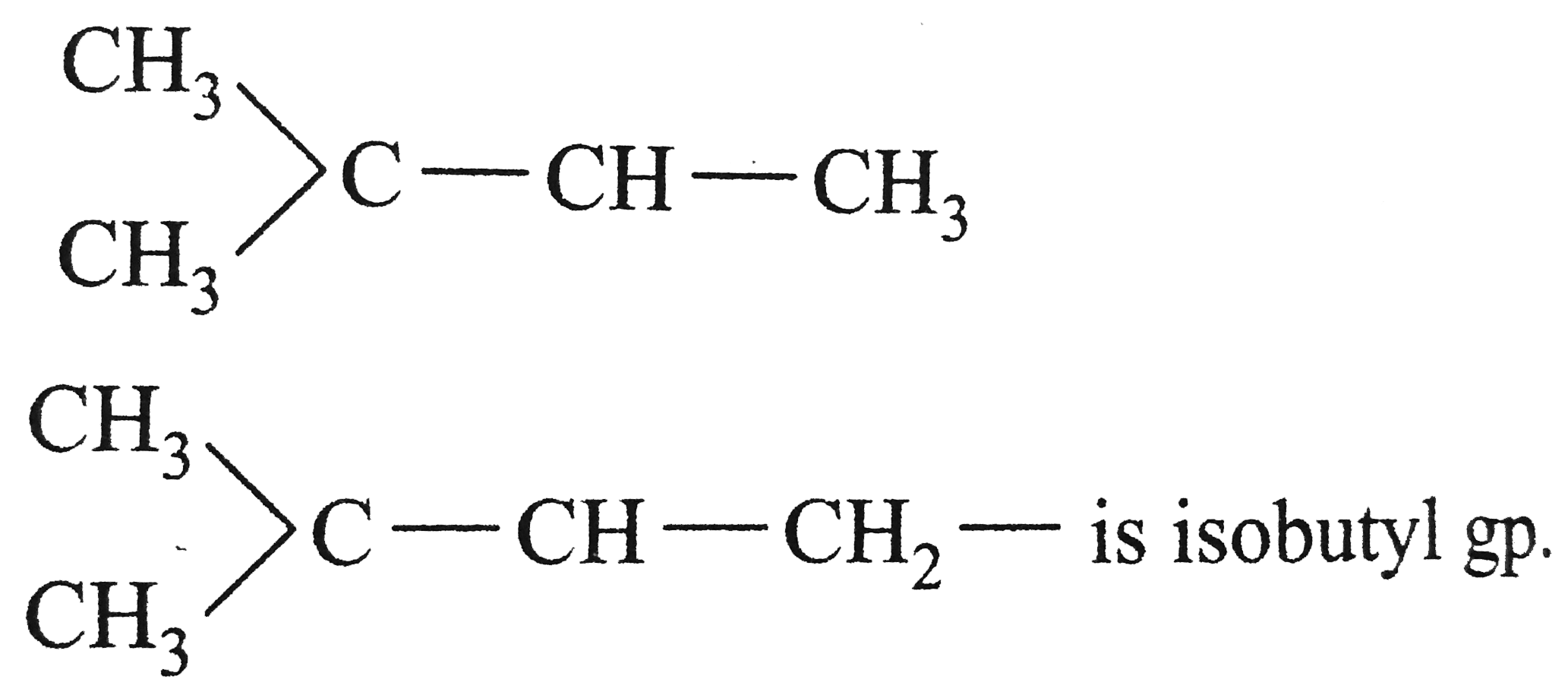 is isobutyl GP. is isobutyl GP.
|
|
| 49. |
The root mean square velocity of an ideal gas at constant pressure varies with density (d) as: |
|
Answer» `d^(2)` |
|