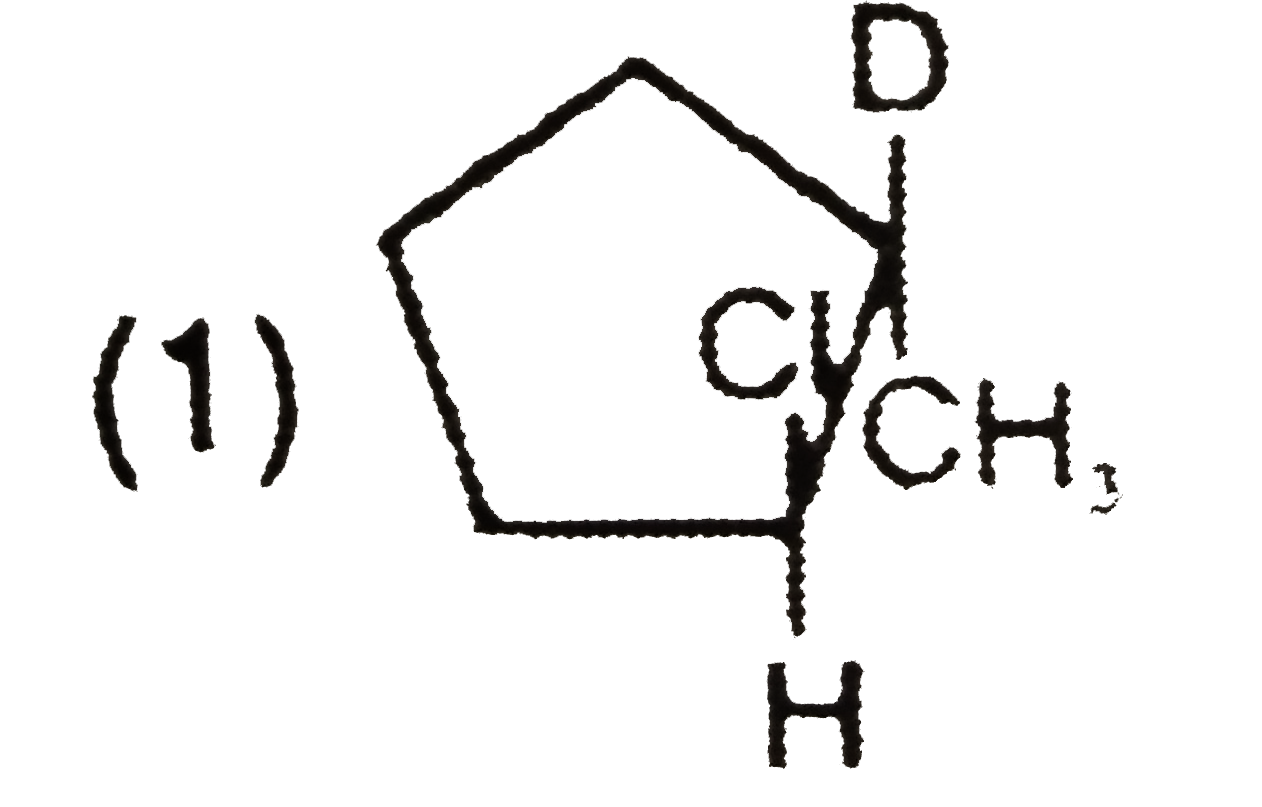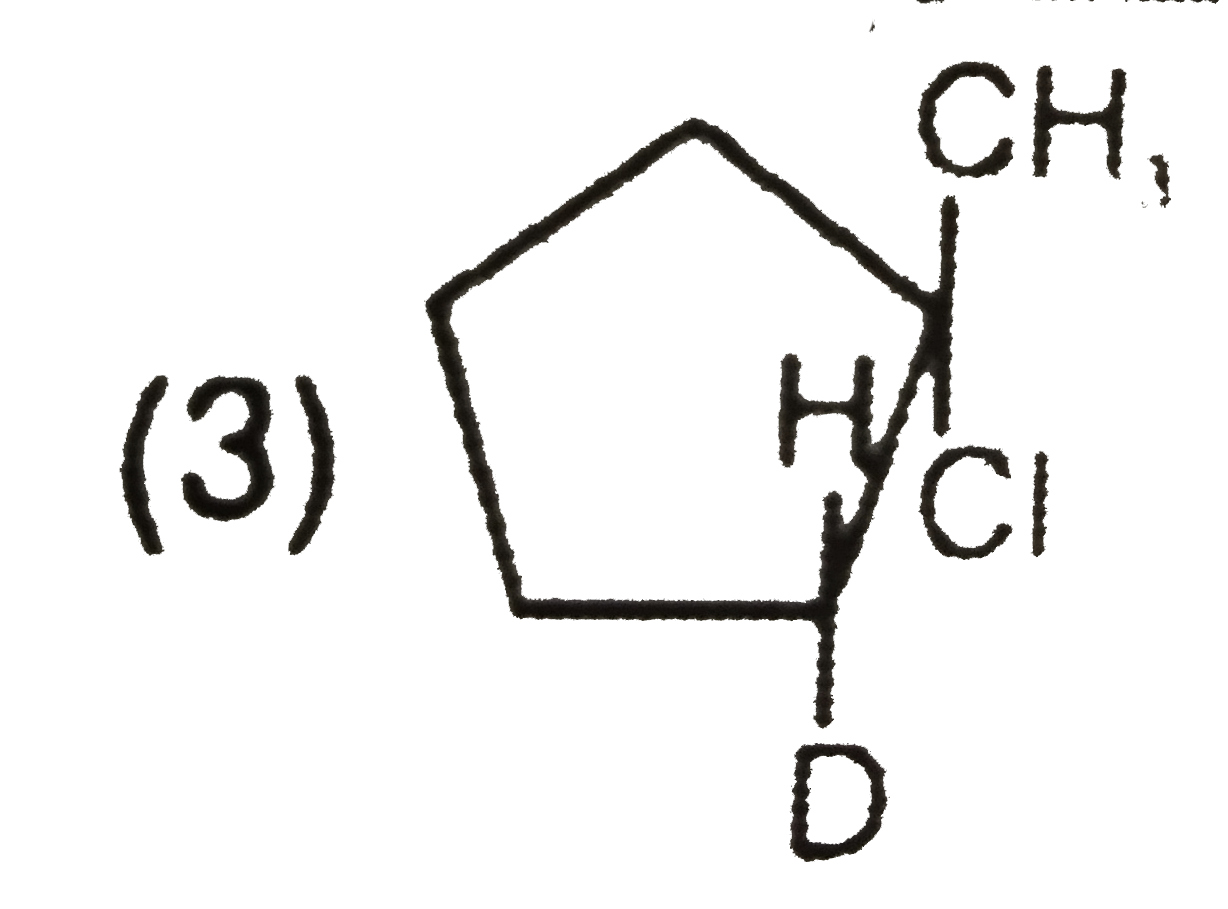Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
What is the K_(c) value of formation of HCl at 300 K ? Explain it. |
|
Answer» SOLUTION :`H_(2)(g)+Cl_(2)(g) hArr 2HCl (g) at 300 K ` `K_(c) = 4 xx 10^(31)` `K_(C) lt 10^(3)` , So [ PRODUCTS ] >> [Reactant] Reaction nearly goes to compeletion. So forward reaction is FAVOURED. |
|
| 2. |
What is the symbol for SI unit of mole ? How is the mole defined ? |
|
Answer» Solution :The symbol for SI of mole is mol. ONE mole is TERMED as the AMOUNT of a substance that CONTAINS as many PARTICLES and there are atoms in exactly 12 g ( 0.012kg) of the `""^(12)C`- isotope. `(1)/(12) g ` of `""^(12)C`-isotope `=1` mole |
|
| 3. |
Which of the following would be true for the reaction shown ? |
|
Answer» The rate of the reaction depends only on the ALKYL bromide concentration. |
|
| 4. |
Two grams of pure caustic soda is present dissolved in 1.5 Lit solution. If 10mL of this is diluted to 150mL. What is the normality of diluted base? |
|
Answer» Solution :Number of MOLES of caustic soda `("WEIGHT")/(GMW)=(2)/(40)=0.05` Normality `=(0.05)/(1.5)=0.0333eq L^(-1)` Equation for DILUTION `=V_(1)N_(1) -V_(2)N_(2)` Normality of dilute solution `=N_(2)=(V_(1)N_(1))/(V_(2))=(10xx0.0333)/(150)=0.00222eq L^(-1)` |
|
| 5. |
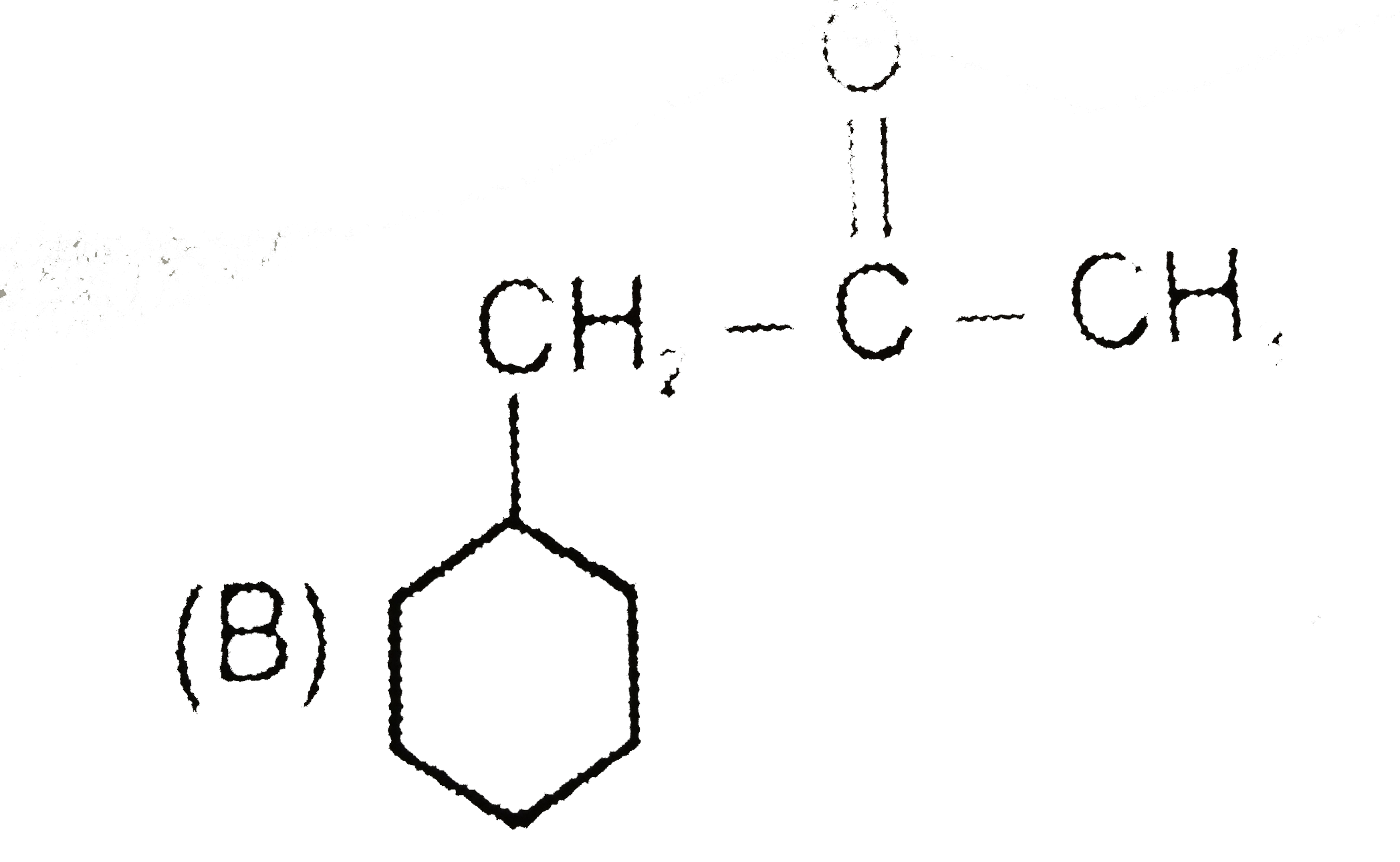
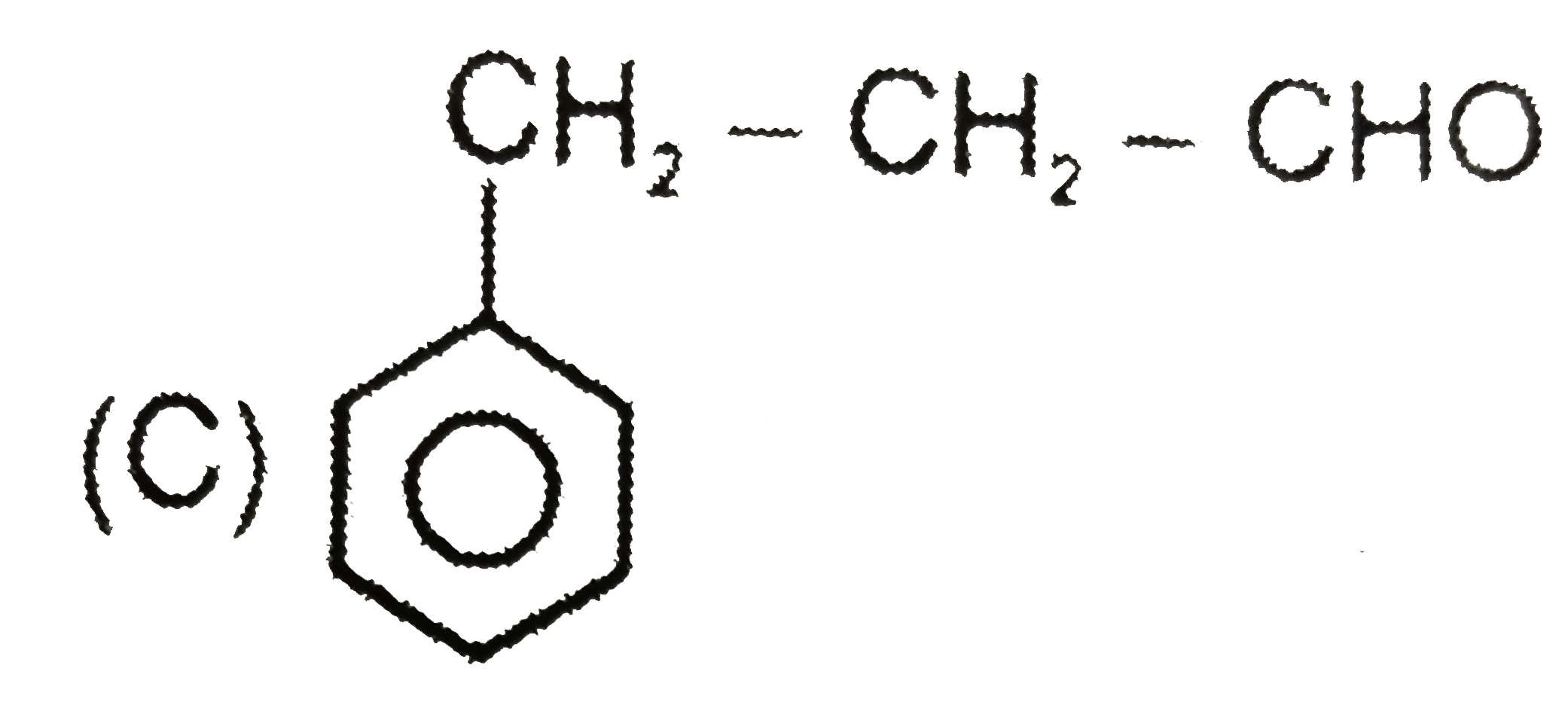
underset("(A)")(Ph-underset(OH)underset(|)(CH)-CHO) overset(HO^(Theta))underset(H_(2)O)(hArr) (B) overset(OH^(Theta))underset(H_(2)O)(hArr) (C) (A),(B) and (C) are structural isomers and isomerization is effectively carried out by trace of base. Give structure of (B) and (C) and also write base catalysed mechanism for this interconversion. |
|
Answer» |
|
| 6. |
What is the effect of temperature on the reactions? (i) N_(2)(g)+3H_(2)(g)hArr2NH_(3)(g)+"Heat" (ii) N_(2)(g)+O_(2)(g)hArr2NO_(3)(g)-"Heat" |
|
Answer» SOLUTION :(i) The INCREASE in temperature will favour BACKWARD reaction because the reaction is exothermic. (II) The increase in temperature FAVOURS forward reaction because the reaction is endothermic. |
|
| 7. |
Which is the Acid in CH_3COOH_((aq)) + H_2O_((aq)) hArr H_3O_((aq))^(+) + CH_3COO_((aq))^(-) |
| Answer» SOLUTION :In FORWARD REACTION : `CH_3COOH` and In REVERSE reaction : `H_3O^(+)` ACID | |
| 8. |
Which of the following is represented incorrectly ? |
|
Answer» `C_6H_6 + Cl_2 UNDERSET("anhydrous" AlCl_3)overset("Sunlight")(rarr) C_6H_6Cl_6` |
|
| 9. |
True statement about silicone is |
|
Answer» Silicon is BONDED to another silicon |
|
| 10. |
The set representing the correct increasing order of first ionization enthalpy |
|
Answer» `Na lt K lt LI` `IP_(1)` values : 
|
|
| 12. |
What do youundersetand bythe term auto-protolysis of water ? What isitssignificance? |
|
Answer» `UNDERSET("Acid"_(1))(H_(2)O(l)) + underset("Base"_(2))(H_(2)O(l)) hArr underset ("Acid"_(2))(H_(3)O^(+) (AQ))+ underset("Base"_(1))(OH^(-) (aq))` Due to auto -protolysis, water is AMPHOTERIC in nature. It reacts with both acids and BASES e.g., `""underset("Acid"_(1))(H_(2)O(l)) + underset("Base"_(2))(NH_(3) (aq)) to underset("Acid"_(2))(NH_(4)^(+) (aq)) + underset("Base"_(1))(OH^(-)(aq))` `underset("Base"_(1))(H_(2)O(l)) + underset("Acid"_(2))(H_(2)O(l)) to underset ("Acid"_(1))(H_(3)O^(+) (aq))+ underset("Base"_(2))(HS^(-) (aq))` |
|
| 13. |
Write note on reverse osmosis and explain its application in desalination of sea water. |
|
Answer» Solution :Let us CONSIDER the experimental setup discussed in the osmosis . The pure water moves through the semipermeable membrane to the NaCl solution due to osmosis . This process can be reversed by applying pressure greater than the osmotic pressure to the solution side . Now the pure water moves from the solution side to the solvent side and this process is CALLED reverse osmosis . It can be defined as a process in which a solvent passes through a semipermeable membrane in the opposite direction of osmosis when SUBJECTED to a hydrostatic pressure greaterthan the osmotic pressure. Application of Reverse osmosis in water purication: Reverse osmosis is used in the desalination of sea water and also in the purication of drinking water. A simple set up usedin both the process is shown in the figure . When a pressure higher than the osmotic pressure it APPLIED on the solution SIDED (sea water )the water molecules moves from solution sideto the solvent side through semi-permeable membrane (Opposite to the Osmotic flow ) the pure water can be collected . ere are different types of semi-permeable membrane used in this process . The membrane used for reverse osmosis has to withstand high pressures . Genrally cellulose acetate or polymide membrane are commonly used in commercial system The selection of membrane used for reverse osmosis will be decided based on the nature of the input water. ` (##SUR_CHE_XI_V02_C09_E05_009_S01.png" width="80%"> |
|
| 14. |
There is an increase of bond length of the carbon- halogen bond from CH_3 F "to" CH_3 I because |
|
Answer» the size of halogens increases from F to I. |
|
| 15. |
The straight chain polymer is formed by |
|
Answer» hydrolysis of `(CH_(3))_(4)SiCl_(3)` by addition CONDENSATION polymerisation |
|
| 16. |
What happens when (i) sodium metal is dropped in water? (ii) sodium metal is heated in free supply of air? (iii) sodium peroxide dissolves in water? |
| Answer» SOLUTION :`2NA + O_2 to UNDERSET("SOD. PEROXIDE")(Na_2O_2)` | |
| 17. |
Which of the following pairs have the same number of atoms ? |
|
Answer» 16 g of `O_(2(g))` and 4 g of `H_(2(g))` No. of atoms in 32 g of `O_(2) = (32)/(32) xx N_(A) xx 2 = 2N_(A)` (D) No. of atoms in 12 g of `C=(12)/(12) xx N_(A) xx 1 = N_(A)` No. of atoms in 23 g of `N_(A) = (23)/(23) xx N_(A) xx 1 = N_(A)` |
|
| 18. |
Which of the following statement are correct ? |
|
Answer» The electronic CONFIGURATION of Cr is `[Ar] 3D^(5) 4s^(1)` (Atomic No. of cr = 24) |
|
| 20. |
underset(Br)underset(|)CH_(2)-underset(Br)underset(|)CH_(2)overset(A)rarrCH=CH where Ais |
|
Answer» Zn |
|
| 21. |
The value of K_c for the reaction 2HI_((g)) hArr H_(2(g)) + I_(2(g)) is 1xx10^(-4) At a given time, the composition of reaction mixture is (HI] = 2 xx 10^(-5) mol, [H_2] = 1 xx 10^(-5) mol and [I_2] = 1 xx 10^(-5) mol In which direction will the reaction proceed ? |
|
Answer» Solution :Given for , `[HI]=2xx10^(-5)` mol `[H_2] = 1xx10^(-5)` mol `[I_2]= 1xx10^(-5)` mol At a given TIME, the reaction quotient Q for the reaction will be given by the expression `Q=([H_2][I_2])/[HI]^2` `=(1xx10^(-5) xx 1xx10^(-5))/(2xx10^(-5))^2=1/4=0.25 =2.5xx10^(-1)` If the value of reaction quotient is more than the value of `K_c`.i.e., `1 xx 10^(-4)` the reaction will PROCEED in the reverse reaction. |
|
| 22. |
Volume of a solution changes with change in temperature, then, what will the molality of the solution be affected by temperature ? Give reason for your answer. |
|
Answer» Solution :No, because mass remains unaffected with temperature hence MOLALITY of solution does not CHANGE. Molality, `m=("MOLES of SOLUTE")/("weight of solvent( in G)")xx1000` |
|
| 23. |
The unpaired electrons in Al and Si are present in 3p orbital. Which electron will experience more effective charge from the nucleus? |
| Answer» SOLUTION :Silicon has GREATER NUCLEAR charge `(+14)` than aluminium `(+13)`. Hence, the unpaired 3p ELECTRON in case of silicon will experience more effective nuclear charge. | |
| 24. |
Which of the following statements is not true about the stability of carbanions? |
|
Answer» Stability of carbanions becauses with increase in s-character of orbital |
|
| 25. |
Which one of the following has Delta S^(0) greater than zero? |
|
Answer» `CaO _((s)) + CO _(2 (s)) hArr CaCO _(3 (s))` |
|
| 26. |
Which method of purification is represented by the following equation Ti(g) +2I_(2)(g) overset(523K)to Til_(4)(g) overset(1700K)to Ti(s) + 2I_(2)(g) |
|
Answer» Cupellation |
|
| 27. |
Under what conditions can a weak electrolyte have a high degree of ionization? |
| Answer» Solution :On DILUTION, i.e., at a very low CONCENTRATION, a weak electrolyte has a high DEGREE of IONIZATION. | |
| 28. |
Give any two characteristics of transition elements. |
|
Answer» SOLUTION :They MOSTLY from COLOURED inos. (2) They exhibit VARIABLE valence (OXIDATION state) |
|
| 29. |
Which of the following statement(s) is/are correct in the case of heavy water ? |
|
Answer» Heavy water is usedas a MODERATOR in nuclear reactor |
|
| 30. |
Which orbital gives an electron the greatest probability of being found close to the nucleus |
|
Answer» 3p |
|
| 31. |
The estimated van't Hoff factor for acetic acid solution in benzene is …………… |
|
Answer» Solution :(i) van.t HOFF factor is defined as the ratio of the actual MOLAR MASS to the abnormal (calculated) molar mass of the solute. (ii) `i=("Noraml molar mass")/("OBSERVED(abnormal") molar mass")` `i=("Observed colligative property")/("Calculate colligative property")` van.t Hoff factor (i) for ACETIC acid `= (60)/(120) =0.5` (iii) van.t Hoff factor (i) for NaCl `= (117)/(58.85) =2` |
|
| 32. |
Write structural formula for the following compounds (i) m-dinitrobenzene (ii) p-dichlorobenzene (iii) 1,3, 5- Trimethylbenzene |
Answer» SOLUTION :
|
|
| 33. |
Whichhas a largerradius: (i) Mg orCa (ii)S or Si |
|
Answer» Solution :(i) Ca(withina GROU sizeincreasesfrom top TOBOTTOM ) (II)s (alongaperiodsizedecreasesfrom LEFT to right ) |
|
| 34. |
Which of the following represents the vartiation of electronegivity with atmoic number Z of f group 13th elements ? Boron and its compounds |
|
Answer»
|
|
| 35. |
The volume to be excluded due to only two molecules of a gas in collision with a fixed point of impact is (0.09)/(N_A) litre (NA = Avogadro number). If the value of 'a' is 3.6 atm L^2 mol^(-2) then the value of Boyle's temperature is, 10^x K. What is the value of 'x'? (R = 0.08 L atm K^(-1)m^(-1)) |
|
Answer» SOLUTION :`B = (0.09)/2 " litre mol"^(-1) implies T_b = a/(bR) = (3.6)/((0.09)/2 R)` `= (7.2)/(0.09 xx 0.08) = (72000)/(9 xx 8) = 1000 K`. |
|
| 36. |
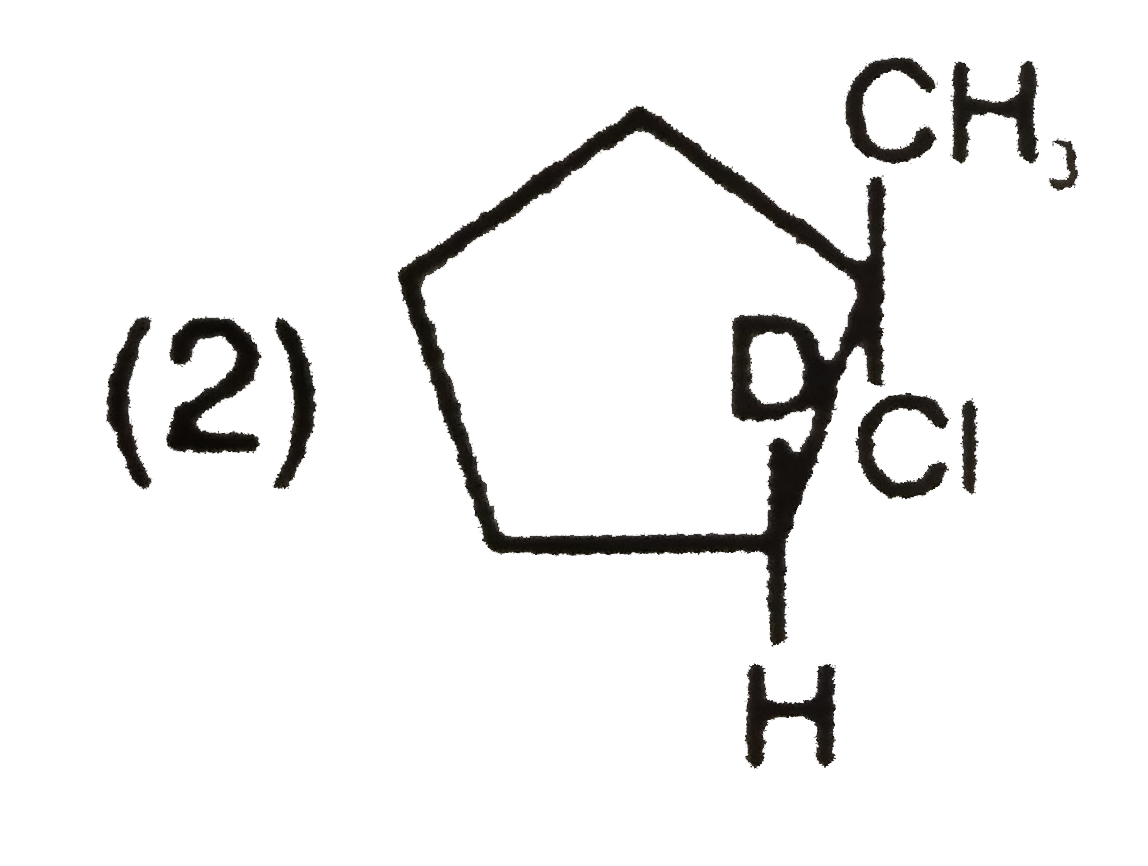
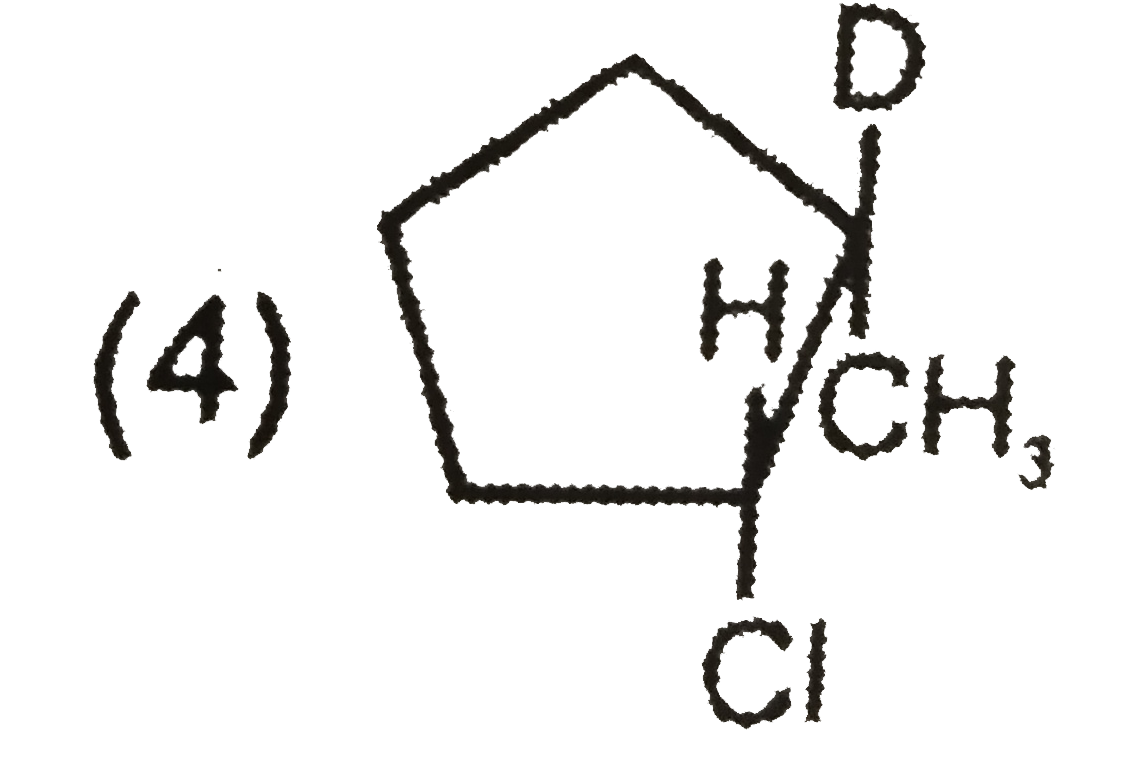
What is the major product expected from the following reaction ? Where D is an isotope of hydrogen. |
|
Answer»
|
|
| 37. |
Which of the following acts as a sink for CO? |
|
Answer» PLANTS |
|
| 38. |
Whichof the followingthe ratioof the sizesofcationof anionis smallest ? |
|
Answer» `NACI` Thus`Na^(+)` has smallerradiusthan `K^(+)` and `Mg^(2+)` has smallerradiusthan `CA^(2+)`. Incontraston movingacrossa period , ionicradiusdecreases .Thus`Mg^(2+)` has smallerradiusthan`Ca^(2+)` .In other words `Mg^(2+)` has thesmallestradiusamongst `Na^(+) , K^(+) , Mg^(2+) ` and `Ca^(2+)` Amongst halogens `C1^(-)` has lergestradiusthan`F^(-)`. thus`MgC1_(2)` has thesmallestratioof the sizeof cationto anion. |
|
| 39. |
Which of the following statements regarding (I) cis-2-pentene &(II) trans-2-pentene is correct |
|
Answer» I & II are a pair of diastereoisomers |
|
| 40. |
Write the IUPAC names of the compound: CH_3 - CH = CH -CH_2 - CH = CH- undersetoverset(|)(C_2H_5)(C)H - CH_2 - CH = CH_2 |
Answer» SOLUTION :
|
|
| 41. |
Which of the following is the use of H_(2)O_(2) i) hair bleach, disinfectant and antiseptic. ii) In the synthesis of hydroquinone, tartaric acid, cephalosporin. iii) Bleaching agent for textile, paper pulp, leather, oils, fats etc. (iv) Treatment of domestic and industrial effluents. |
| Answer» Answer :D | |
| 42. |
Write the order of oxygen-oxygen bond energies of O_2 , O_3 and H_2 O_2 molecules. |
|
Answer» Solution :BOND order in `O_(2)` is 2. Bond order in `O_(3)` is 1.5. Bond order in `H_(2)O_(2)` is 1. Descending order of O-O bond energies: `O_(2)gtO_(3)gtH_(2)O_(2)` |
|
| 43. |
Volume of 0.1 M NaOH needed for the neutralisation of 20 mL of 0.05 M oxalic acid is |
|
Answer» 10 mL |
|
| 44. |
What are the atomicity values of (a) monoclinic sulphur, (b) phosgene, (c ) phenol and (d) fructose. |
|
Answer» |
|
| 45. |
Which of the following alkali metal carbonates gives CO_(2) on heating as well as on treating with acids ? |
|
Answer» `Na_(2)CO_(3)` `Li_(2)CO_(3)+2HClrarr2LiCl +CO_(2)+H_(2)O` |
|
| 46. |
Which 'H' atom is most acidic in the following compounds. |
|
Answer» |
|
| 47. |
What are the general molecular formula and functional group of the following compounds? (i) Hydrazines (ii) Hydrazo compound (iii) Imide (iv) Phenols (v) Amine (vi) Nitroalkane |
Answer» SOLUTION :
|
|
| 48. |
Which of the following is a network solid ? |
|
Answer» `SO_2` (SOLID ) |
|
| 49. |
Which of the following set of quantum numbers is correct for the 19^"th" electron of chromium |
|
Answer» `{:(N,L,m,s),(3,0,0,1//2):}` |
|
| 50. |
There is no difference between a 2p and a 3p orbital regarding |
|
Answer» VALUE of n |
|
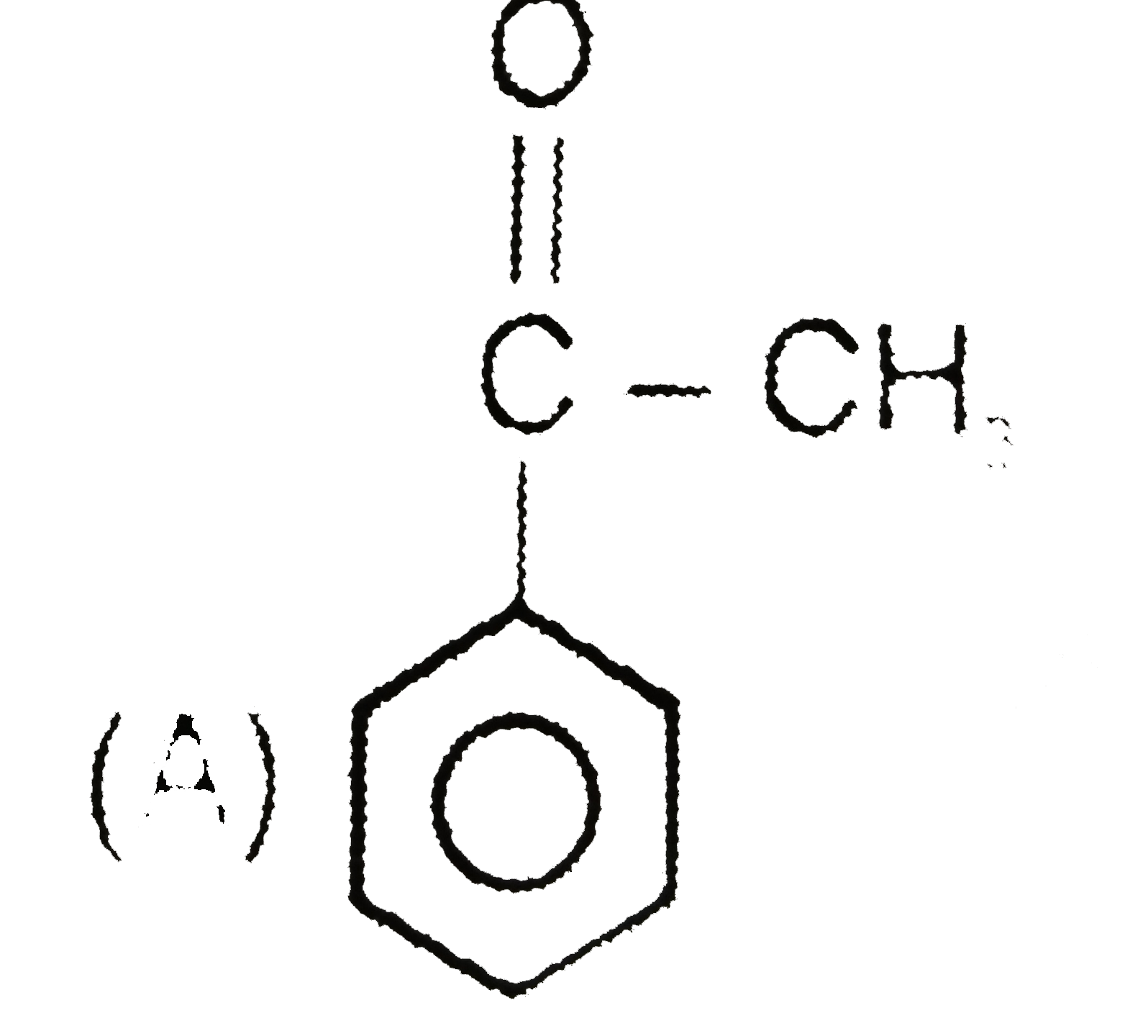




 increases the stability of carbanions.
increases the stability of carbanions.