Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which substance possess B_3N_3H_6 molecular formula ? |
|
Answer» Boric ACID |
|
| 2. |
Write the relation between Delta H and Delta U. |
|
Answer» Solution :`Delta H = Delta U + Delta n. RT` where R is GAS constant= 8.314 J/K mole T is the temperature in kelvin `Delta n` = Number of MOLES of GASEOUS products - Number of moles of gaseous reactants. |
|
| 3. |
What is the oxidation number of the metals [Fe(CN)_6]^(4-) |
|
Answer» Solution :Fe in `[Fe(CN)_6]^(4-)` . LET the oxidation number of Fe in `[Fe(CN)_6]^(4-)` be x and CN be -1 i.e., `oversetx(Fe)OVERSET(-)((CN))_6` `therefore` SUM of oxidation number of all the atoms in `[Fe(CN)_6]^(4-)=x+6(-1)=-4,x-6=-4,x=+2` Thus, the oxidation number of Fe in `[Fe(CN)_6]^(4-)` is +2. |
|
| 4. |
What are electrophiles? Give an example. |
|
Answer» SOLUTION :Electrophiles: are reagents that are ATTRACTED towards NEGATIVE charge on rich center. They are either positively charged ions or electron deficient neutral MOLECULES. Example, `CO_2 AICI_3, BF_3 FeCI_3, NO^(+), NO_(2)^(+)`.etc. |
|
| 5. |
When one mole of an ideal gas is compressed to half of its initial volume and simultaneously heated to twice its initial temperature, the change in entropy of gas (Delta S) is |
|
Answer» `C_(p, m) ln 2` `=1 XX C_(v) ln 2 + 1 xx R "ln " (1)/(2) = ln = (C_(v)- R)` |
|
| 6. |
When sodium metal is dropped in liquid NH_3 it forms Na^(+) and gets ammoniated. Which of the following forces are responsible for the formation of ammoniated sodium ion. |
|
Answer» ION-induced dipole |
|
| 7. |
When NaNO_(3) is heated in a closed vessel, O_(2) is liberated and NaNO_(2) is left behind. At equilibrium: NaNO_(3(g)) hArr NaNO_(2(g)) + (1)/(2) O_(2(g)) |
|
Answer» addition of `NaNO_2` favours reverse reaction |
|
| 8. |
Which of the following reagent(s) make distinction of acetate from oxalates and formates? |
|
Answer» `BaCI_(2)` |
|
| 9. |
Verify the law of reciprocal proportions from the following data : KCl contains 52.0 % potassium , KI contains 23.6 % potassium while Icl contains 78.2 % iodine. |
|
Answer» Solution :Let 1 g of potassium (K) be the FIXED WEIGHT In KCl, 52.0 g of K are COMBINED with Cl = 48.0 g 1.0 g of K are combined with `Cl = (48.0)/(52.0)g = 0.092 g` In KI, 23.6 g of K are combined with `I = 76.4 g` 1.0 g of K is combined with `I = (76.4)/(23.6)g = 3.24 g` Ratio by weight of I and Cl combined with 1.0 g of K in the two halides `= (3.24)/(0.92)` In ICI, ratio by weight of I and Cl `= (78.2)/(21.8)` COMPARING the two ratios : `(3.24)/(0.92):(78.2)/(21.8)or 3.52:3.59` Since the two ratios are nearly the same, this verifies the Law of Reciprocal Proportions. 
|
|
| 10. |
Write the condensed formulae for each of the following compounds : (i) Isopropyl alcohol(ii) Methyl t-butyl ether (iii) 2-Chloro-1, 1, 1-trifluoroethane(iv) 2-Methylbuta-1, 3-diene(v) But-2-en-1-ol |
|
Answer» Solution :(i) `(CH_(3))_(2)CHOH`(ii) `CH_(3)-O-C(CH_(3))_(3)`(III) `F_(3)C-CH_(2)Cl` (iv) `{:(""CH_(3)),("|"),(CH_(2)=C-CH=CH_(2)):}` (V) `CH_(3)CH=CHCH_(2)OH` |
|
| 11. |
What is meant by elastic collision? |
|
Answer» Solution :Elastic collision means that the total ENERGY of molecules before and after collision remains same. There may be exchange of energy between COLLIDING molecules. Their individual ENERGIES may change, but the average energy remains CONSTANT. |
|
| 12. |
Which of the following compounds will decarboxylate to form ketone ? |
|
Answer» `CH_(3)CH_(2)COCH_(2)COOH` 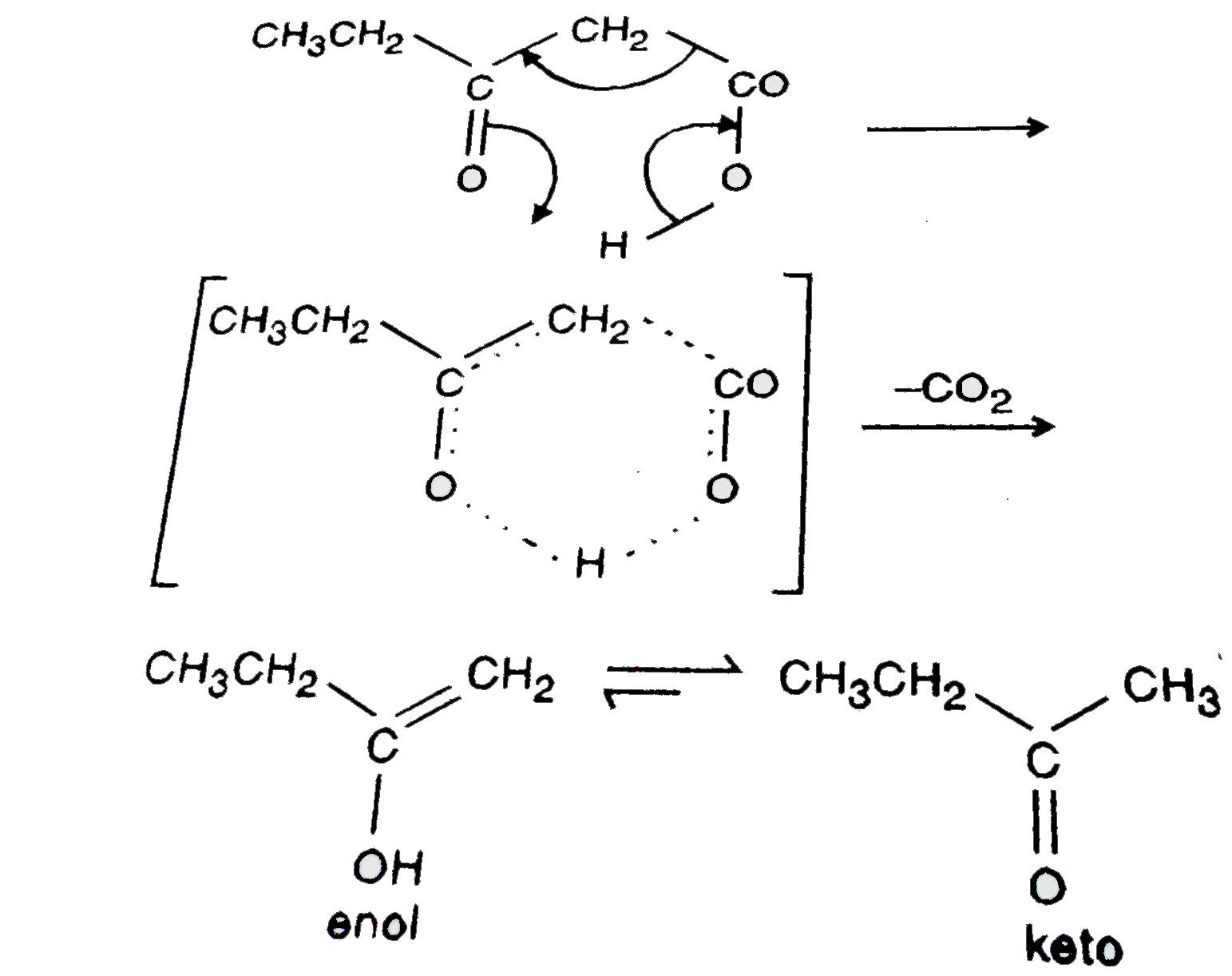
|
|
| 13. |
Which of the following CORRECTLY represents the reactions occuring in Daniel cell ? |
|
Answer» `CH_(4(g)) + O_(2(g)) RARR CO_(2(g)) + 2H_(2)O_((l))` |
|
| 14. |
Which one of the following gives onlyh one monochloro derivative? |
|
Answer» neo-Pentane |
|
| 15. |
Which of the following statement about IV group elements ( C to Pb) is not correct |
|
Answer» The thermal stability of hydrides decreases from `CH_(4)" to "PbH_(4)` as bond enthalpy for M - H bond decreases |
|
| 16. |
The volume of liberated at STP from 20ml of Vol H_(2)O_(2) is |
|
Answer» 20 ml |
|
| 17. |
Which of thefollowingsentences is not true ? |
|
Answer» Lightisa BEAM of PHOTON |
|
| 18. |
What is the molecular formula of carvone ? |
|
Answer» `C_(8)H_(10)O` |
|
| 19. |
Which of the following compounds can prefer E^(1)cb mechanism over E^(2) ?. |
|
Answer»

|
|
| 20. |
Which one of the following can exist in optically active forms? |
|
Answer» cis-1,3-Dichlorocyclohexane |
|
| 21. |
Which allotropes of phosphorous is most stable ? |
|
Answer» BLACK P |
|
| 22. |
Which one is a wrong statement ? |
|
Answer» The VALUE of m for `d_z^2`is ZERO. 
|
|
| 23. |
Which of the following do not represent ground state electronic configuration of an atom ? |
|
Answer» `1s^(2)2s^(2)2p^(6)3s^(2)3p^(6)3d^(8)4s^(2)` |
|
| 24. |
What is the mass percentage of carbon in ethane. |
|
Answer» |
|
| 25. |
Write the relation between Delta G^(@) and equilibrium constant. |
|
Answer» `Delta G^(@) = -2.303 RT LOG K` This equation is called "Van't Hoff's reaction isotherm.". When the reaction attains equilibrium, `Delta G = 0` and `(p_(C ).p_(D))/(p_(A).p_(B)) = K`. The above equation becomes, `0 = Delta G^(@) + RT ln K` or `Delta G^(@) = -RT ln K` `:. Delta G^(@) = -2.303 RT log K` `Delta G^(@)` is the standard free energy change of the reaction, R is the gas constant, T is the temperature in absolute scale and `K_(p)` is the equilibrium constant In general, for `aA + bB hArr c C + d D` `Delta G^(@) = -2.303 RT log K_(p)` where `K_(p) = (p_(C ).p_(D))/(p_(A).p_(B))` |
|
| 26. |
Which of the following is the correct IUPAC name ? |
|
Answer» 3-ethyl-4, 4-dimethylheptane According to alphabetical order methyl is written after ethyl. To write prefixes di of two `-CH_(3)` |
|
| 27. |
What do you understand by isoelectronic species ? Name a species that will be isoelectronic with each of the following atoms or ions. (i) F^(-)"" (ii) Ar"" (iii) Mg^(2+) "" (v) Rb^(+) |
|
Answer» Solution :Number of electrons in ATOM or IONS are same than it is called .isoelectric species.. e.g. `O^(2-) , F^(-) , Na^(+) , Mg^2+)`ions having 10 electrons and isoelectric species. Radius of isoelectric species are different because nuclear charge are different. In isoelectric species, nuclear charge increases radius decreases so radius increases like `Mg^(2+) lt Na^(+) lt F^(-) lt O^(2-) `because nuclear charge decreases like 12, 11, 9, 8. 
|
|
| 28. |
The region which contains water vapour is |
|
Answer» STRATOSPHERE |
|
| 29. |
What is the role of builder in synthetic detergents? |
| Answer» Solution :It REMOVES hardness PRODUCING ions VIZ., `Ca^(2+)` and `MG^(2+)` ions | |
| 30. |
Which one of the following is not a transuranic element ? |
|
Answer» Californium |
|
| 31. |
Which of the following pairs of d-orbitals will have electron along the axes? |
|
Answer» `d_(z^2), d_(XZ)` |
|
| 32. |
What is dead burnt plaster ? Why is it so called ? |
|
Answer» Solution :When gypsum or PLASTER of Paris is heated above 393 K , it loses all the water of CRYSTALLIZATION and anhydrous calcium sulphate is obtained . `Caunderset("Gypsum")(SO_(4).2H_(2)) O overset( gt 393K)(to) underset("Dead burnt plaster")(CaSO_(4)) + 2H_(2)O` `2 underset("Plaster of Paris")(CaSO_(4).2H_(2)) O overset(gt 393 K)(to) 2 CaSO_(4) + H_(2)O` Anhydrous calcium sulphate does not absorb water to form gypsum and HENCE loses the properties of setting with water . That is why anhydrous calcium sulphate is called dead burnt plaster . |
|
| 33. |
Which of the following lattices has the highest packing efficiency ? (i) Simple cubic (ii)body-centred cubic (iii) hexagonal close-packed lattice.. |
| Answer» SOLUTION :packing efficiencies are : simple CUBIC = 52.4% , BODY - centred cubic = 68% , HEXAGONAL close-packed = 74 % thus, hexagonal close - packed lattice has the highest packing EFFICIENCY. | |
| 34. |
Which conformation of ethane has the lowest potential energy? |
|
Answer» Eclipsed |
|
| 35. |
Which of the following is called Lucas reagent? |
|
Answer» CONC. `H_2SO_4` + ANHYDROUS `CuSO_4` |
|
| 36. |
Water softened by permutitt process contains |
|
Answer» <P>Dissolved SODIUM salts |
|
| 37. |
Which of the following sets of ions represents the collections of isoelectronic species? |
|
Answer» `K^(+), CA^(2+), Sc^(3+), Cl^(-)` 
|
|
| 38. |
Which of the following is a wrong statement ? |
|
Answer» The reacting species in SULPHONATION is `H_2SO_4` |
|
| 39. |
Which one is not correct for the reaction (CN)_(2) + H_(2)O rarr HCN + HOCN? |
|
Answer» It is an auto REDOX change |
|
| 40. |
Phosphorus on reaction with NaOH produces PH_(3) and NaH_(2)PO_(2). This reaction is an example of |
|
Answer» oxidation |
|
| 41. |
What is organic chemistry? |
| Answer» Solution :The carbon FORMS covalent bonds with other carbon and also with other elements like hydrogen oxygen, nitrogen, SULPHUR, PHOSPHORUS and halogen. The resulting compound are studies under a separate branch of chemistry called ORGANIC chemistry | |
| 42. |
Which of the following is not an example of friedel-craft reaction. |
|
Answer»
|
|
| 43. |
When an electric discharge is passed through hydrogen gas, the hydrogen molecules dissociate to produce excited hydrogen atoms. These excited atoms emit electromagnetic radiation of discrete frequencies which can be given by the general formula bar(v) = 109677 [(1)/(n_(i)^(2))- (1)/(n_(2)^(f))] What points of Bohr's model of an atom can be usedto arrive at this formula ? Based on these points derive the above formula giving description of each step and each term. |
|
Answer» Solution :Two important points of Bohr's model that can be used to derive the given formula are: (i) Electrons revolve AROUND the nucleus in stationary states, i.e., orbits with fixed values of energy (quantized values). (ii) Energy is EMITTED or absorbed only when electron jumps from one orbit to another Derivation. Step (i) The energy of the electron in the nth stationary state is given by `E_(n) = - (2pi^(2) me^(4))/(n^(2)h^(2))` where m = mass of electron, e= charge on electron, h = Planck's constant Step (ii). When th electron jumps from outer stationay state `n_(2)` to inner stationary state `n_(1)`, the different of energy `(Delta E)` is emitted, i.e., `Delta E = E_(2) - E_(1) = - (2pi^(2) me^(4))/(n_(2)^(2) h^(2)) - (-(2pi^(2) me^(4))/(n_(1)^(2) h^(2))) = (2pi^(2) me^(4))/(h^(2)) ((1)/(n_(1)^(2)) - (1)/(n_(2)^(2)))` Step (iii), `Delta E = hv = h(c)/(LAMDA) = hc BAR(v)` `:. bar(v) = (Delta E)/(hc) = (2pi^(2) me^(4))/(ch^(3)) ((1)/(n_(1)^(2)) - (1)/(n_(2)^(2)))` Substituting the value of the constant, `pi, m, e, c and h` in CGS units, we get `bar(v) = 109677 cm^(-1) ((1)/(n_(1)^(2)) - (1)/(n_(2)^(2)))` |
|
| 44. |
Which of the following can act both as oxidising agents and reducing agents? |
|
Answer» `HNO_(2)` |
|
| 45. |
Write the structural formula of the following compounds : (i) 2- Chloro- 2- Methylpropane (ii) 1- Bromo-2 , 2-Dimethylpropane |
|
Answer» SOLUTION :(i) 2 - Chloro-2- Methylpropane <BR> `CH_(3)- overset(CH_(3))overset(|)underset(Cl) underset(|)(C)- CH_(3)` (ii) 1- Bromo-2 , 2- DIMETHYLPROPANE `CH_(3) - overset(CH_(3))overset(|)underset(CH_(3))underset(|)(C) - CH_(2) Br` |
|
| 47. |
Write the position isomers of (a) C_(3)H_(6)Cl_(2) (b) C_(3) H_(5) Cl |
|
Answer» Solution :The number of POSITION isomers for the formula `C_(3)H_(6)CI_(2)`. Is four. `(1)underset(1,1-"Dichloropropane")(CH_(3)-CH_(2)-CHCI_(2))` `(2) underset(2,2-"Dichlorophane")(CH_(3)-C CI_(2)-CH_(3))` (3) `underset(1,2-"Dichloroprophane")(CH_(3)-CHCI-CH_(2)CI)` (4) `underset(1,3-"Dichloropropane")(CH_(2)CI-CH_(2)CI)` (B) The number of position isomers for the formula `C_(3)H_(5)CI` is three. (1) `underset(3-"Chlorpropane")(CH_(2)=CH-CH_(2)CI)` (2) `underset(2-"Chloropropane")(CH_(2)=C CI-CH_(3))` (3) `underset(1-"Chloropropane")(CHCI=CH-CH_(3))` |
|
| 48. |
Which of the following statement is not correct about the characteristics of cathode rays |
|
Answer» They START from the cathode and move towards the anode. |
|
| 49. |
When zeolite, which is hydrated sodium aluminium silicate, is treated with hard water, the sodium ions are exchanged with |
|
Answer» `H^(+)` IONS |
|
| 50. |
When SO_2is passed in acidified potassium dichromate solution, the oxidation state of S is changedfrom: |
|
Answer» `+ 4` to 0 |
|







