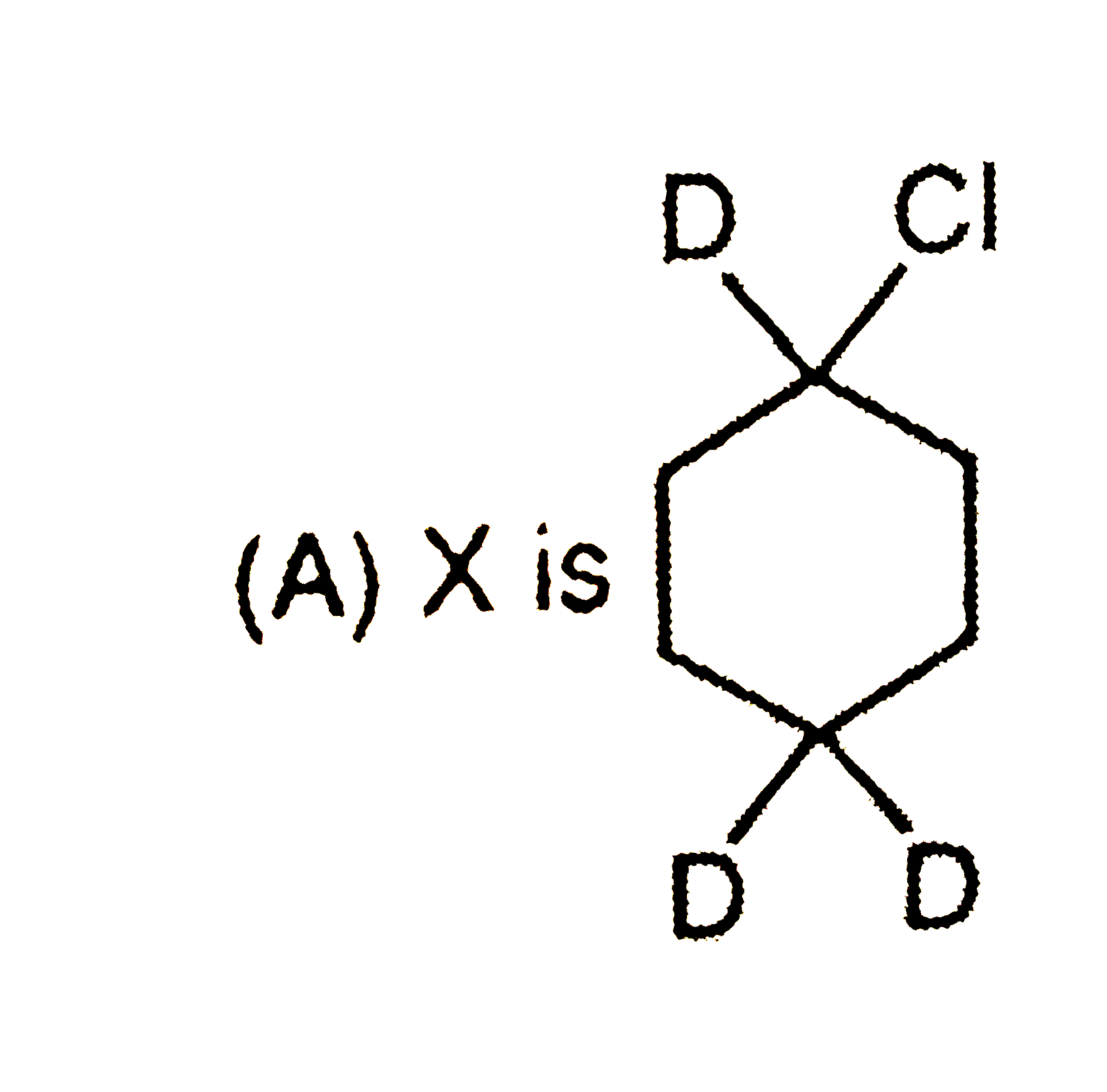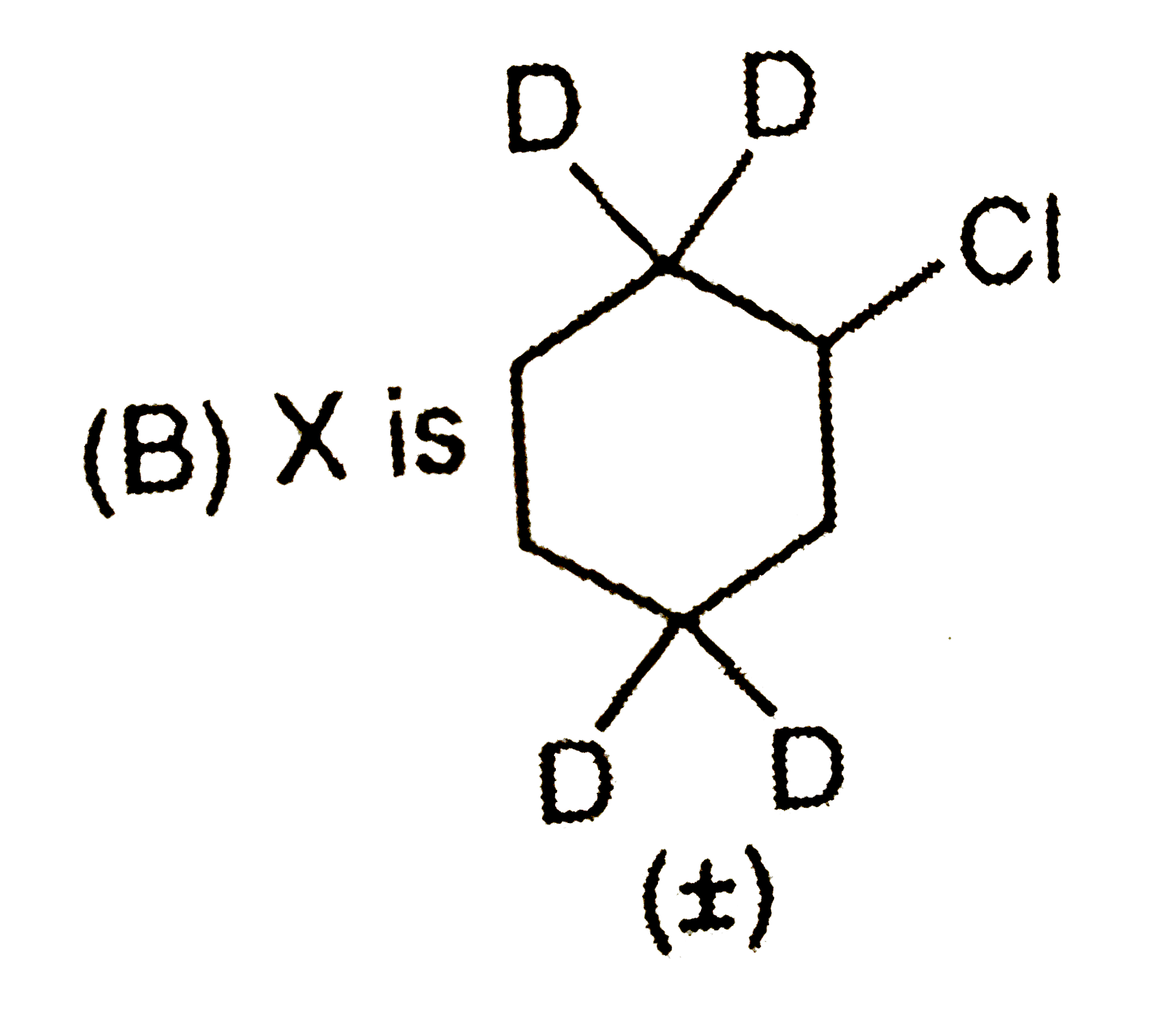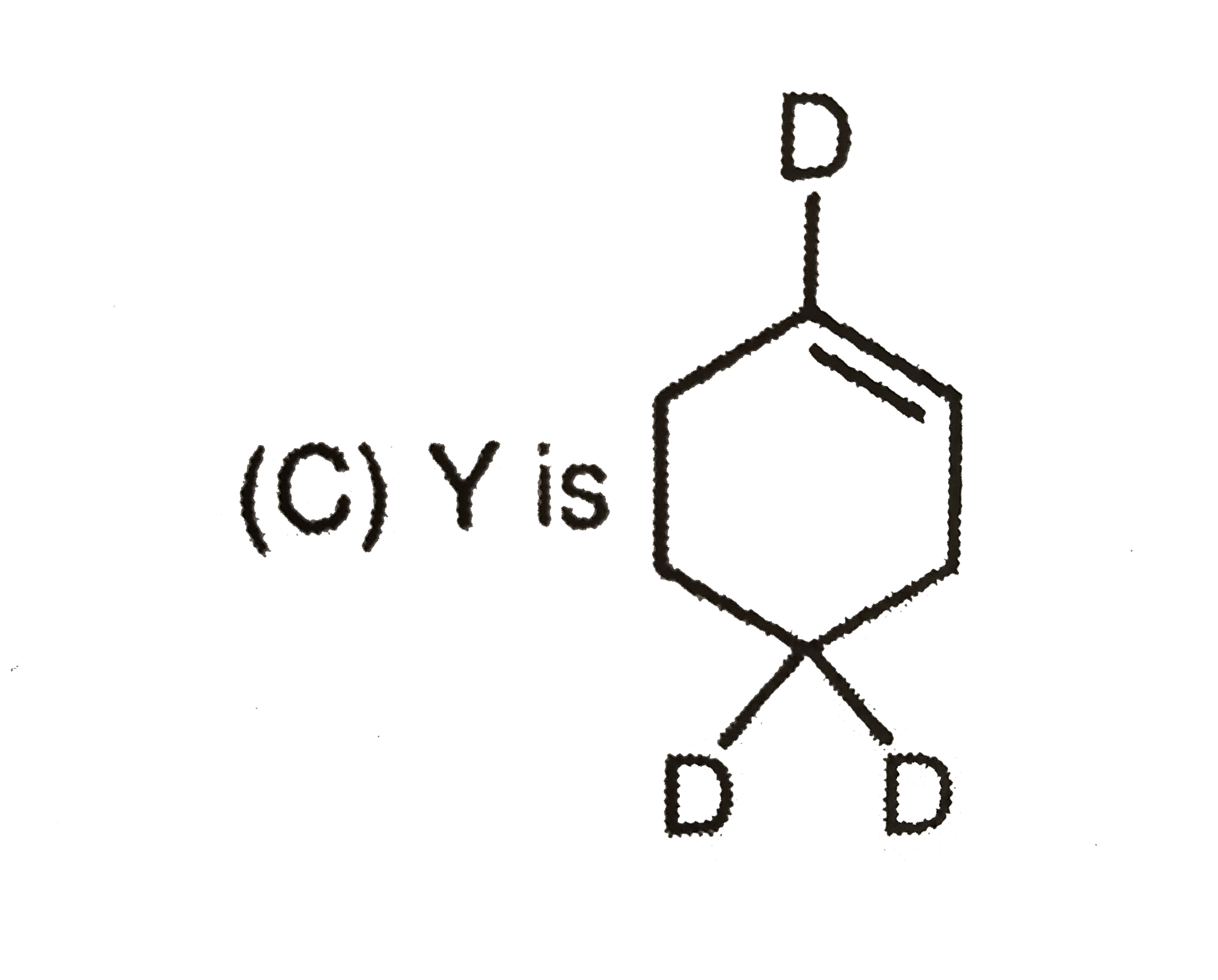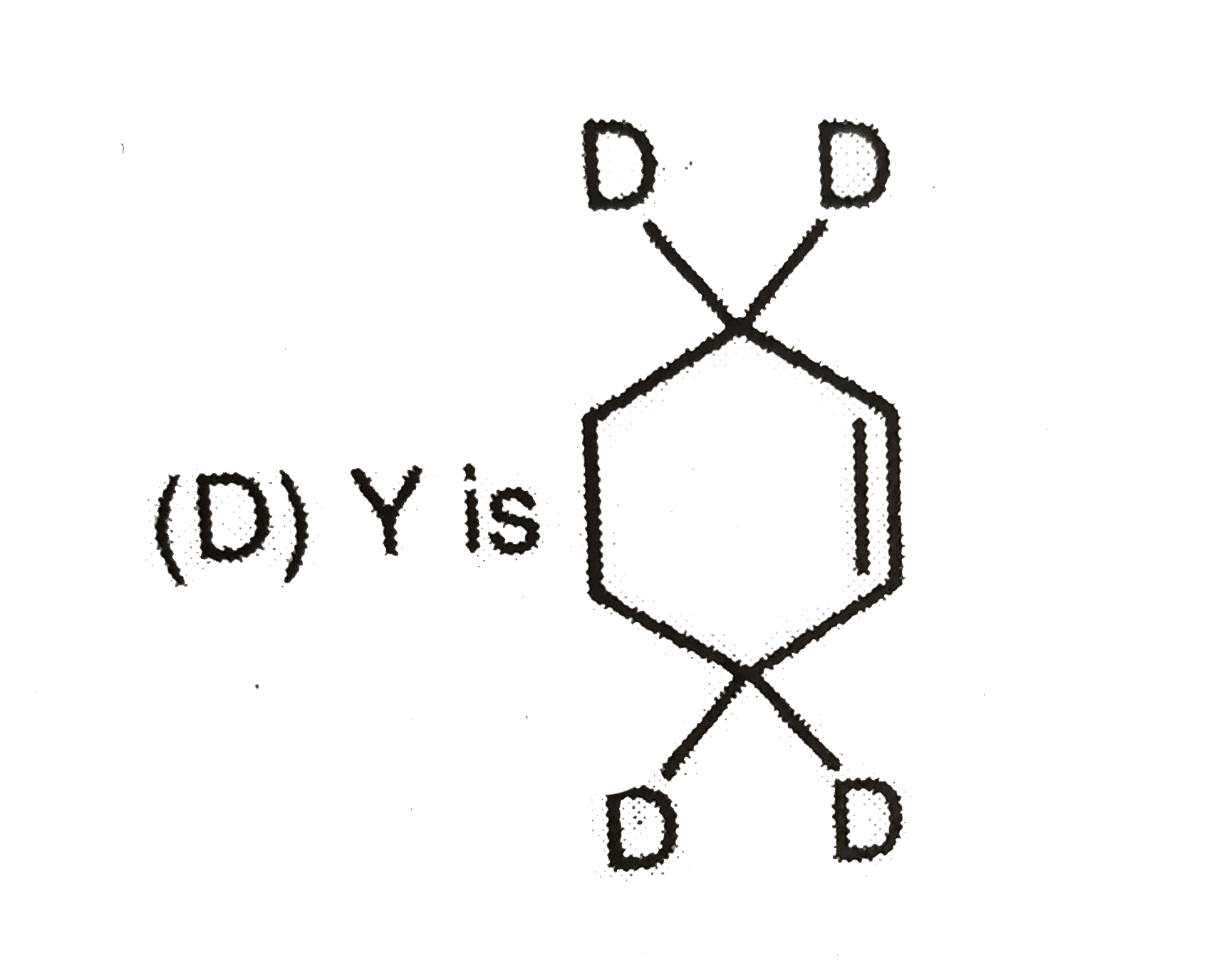Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
What is meant by a functional group? Identify the functional group in the following compounds (a) acetaldehyde (b) oxalic acid (c) dimethyl ether (d) methylamine |
|
Answer» Solution :(i) A funcnonal group is an atom or a specific COMBINATION of bonded atoms that react in a CHARACTERISTIC WAY, irrespective of the ORGANIC molecule in which it is PRESENT. (ii) 
|
|
| 2. |
When does the rate of backward reaction increase ? What is its consequence ? A+BhArrC+D |
|
Answer» Solution :(i) Initially only A and B are present. Soon, a few molecules of the products C and D are formed by the forward reaction. (ii) As the concentration of the products increases, more products collide and react in the BACKWARD direction. (iii) This leads to an INCREASE in the rate of backward reaction. As the rate of reverse reaction increases, the rate of the forward reaction decreases. (iv) Eventually, the rate of both reactions BECOMES equal. 
|
|
| 3. |
What do you mean by Alkane ? Explain the formation of alkane from methane, substituting the hydrogens by -CH_(3) groups ? |
|
Answer» Solution :* Alkanes are saturated open chain hydrcarbons containing carbon-carbon single bonds. * Methane is the first member of this family. Methane is a gas found in coal mines and marshy PLACES. If you replace one hydrogen atom of methane by carbon and JOIN the required NUMBER of hydrogens to satisfy the tetravalence of the other carbon atom, this hydrocarbon with molecular formula `C_(2)H_(6)` is known as ethane. Thus you can consider `C_(2)H_(6)` as derived from `CH_(4)` by REPLACING one hydrogen atom by `-CH_(3)` group. Go on constructing alkanes by doing this theoretical exercise i.e., replacing hydrogen atom by `-CH_(3)` group. `underset("Methane "(CH_(4)))(H-underset(H)underset(|)overset(H)overset(|)(C)-H) overset("Replace any H by "-CH_(3))rarr underset("Ethane "(C_(2)H_(6)))(H-underset(H)underset(|)overset(H)overset(|)(C)-underset(H)underset(|)overset(H)overset(|)(C)-H)` The next molecules will be `C_(3)H_(8), C_(4)H_(10)`...... `CH_(4)underset(+CH_(3))overset(-H)rarr H_(3)C-CH_(3) underset(+CH_(3))overset(-H)rarr CH_(3)-CH_(2)-CH_(3)`  `underset("ISOBUTANE")(H_(3)C-underset(CH_(3))underset(|)(CH)+ CH_(3))+H_(3)C-underset("n-butane")(CH_(2)-CH_(2)-CH_(3))` |
|
| 4. |
Which among the following method is the best for the preparationof BeF_(2)? |
|
Answer» `(NH_(4))_(2) BeF_(4) OVERSET(Delta)to` `(NH_(4))_(2)BeF_(4) overset(Delta)to BeF_(2)+2NH_(4)F` |
|
| 5. |
What is meant by BOD? |
| Answer» Solution :BOD is the amount of dissolved oxygen REQUIRED by microorganism to oxidise organic organic matter PRESENT in polluted water. It is expressed in ppm. This gives a REALISTIC MEASURE of the QUALITY of water. | |
| 6. |
Vinegar obtained from sugarcane has |
|
Answer» `CH_(3)COOH` |
|
| 7. |
What isthe wavelengthelectron? What itindicate ? |
|
Answer» SOLUTION :`LAMBDA = 896.7 nm = 8.967 XX 10^(8) m` it is morethanordinaryobject so electronhas WAVENATURE. |
|
| 8. |
What is multiple bond ? Give difference between single bond, double bond and triple bond. |
Answer» SOLUTION :If ELECTRON pair between TWO atom is more than one it is called multiple BOND.
|
|
| 9. |
What is correct for resonance structure ? |
|
Answer» Solution :The CANNONICAL forms have no real existence. The molecule does not exist for a certain fraction of time in one cannonical forms. There is no such equilibrium between the cannonical forms as we have between tautomeric FORM in tautomerism. The molecule as such has a single structure which is the RESONANCE hybrid of the cannonical form and which cannot as such be DEPICTED by a single Lewis structure. |
|
| 10. |
What is meant by dead burnt plaster? |
| Answer» SOLUTION :It is anhydrous CALCIUM sulphate `(CaSO_(4))` | |
| 12. |
The space lattice given in the figure to : |
|
Answer» FLUORITE STRUCTURE |
|
| 13. |
Which compound is reducing agent in the following equation ? CH_(3)CHO_((s))+Ag_(2)O_((s))toCH_(3)COOH+2Ag |
| Answer» Answer :C | |
| 14. |
Which elements are from the following ? Isoelectric Na, Na^(+) , F^(-) , F ? |
| Answer» SOLUTION :`F^(-)" and " Na^(+)` are ISOELECTRIC elements they have 10 ELECTRONS. | |
| 15. |
Which observation will be correct about the major products X and Y of the following reaction. |
|
Answer»
|
|
| 16. |
What per cent of its temperature pressure and volume are to 110^(@)C, 0.7 atm 1.0litre repectively ? . |
|
Answer» |
|
| 17. |
The stability of the following alkali metal chlorides follows the order : |
|
Answer» `LiCl gt KCl gt NaCl gt CS Cl` |
|
| 18. |
What mass of NaCl would contain the same total number of ions as 245 g of MgCl_(2) ? |
|
Answer» |
|
| 19. |
Write the electronic configurations and the names of the elements having the atomic numbers 5, 9, 10, 19 and 20. |
Answer» SOLUTION :
|
|
| 20. |
What are ideal gases? |
|
Answer» SOLUTION :i) A gas that follows Boyle.s law, CHARLES. law and Avogadro law strictly is called an ideal gas. It is assumed that intermolecular forces are not present between the molecules of an ideal gas. ( ii) Ideal GASES are the gases that obey gas laws or gas EQUATION PV = nRT. (iii) Real gases do not obey gas equation. PV = nRT. (iv) The DEVIATION of reaf gases from ideal behaviour is measure in terms of a ratio of PV to nRT. This is termed as compression factor (Z). `Z= (PV)/(nRT)` |
|
| 21. |
Which of the following is the strongest acid ? |
|
Answer» `CH_(3)underset(F)underset(|)(C)HCOOH` |
|
| 22. |
Which of the following statements regarding Be is /are true? (I) It forms covalent compounds almost exclusivlty , evan with the highly eletronegative elements (II) It does not form ionic compounds containing simple Ber^(+) ion , but does readily acheve a maximum coordination mumberof fopur through formation of complex ions such as and BeF_(4)^(2-)[Be(H_(2)))_(4)]^(2+). (III) it forms a series of organo derivatives BeR_(2) and [BeR_(3)],which contain covalent Be-C bonds. (IV) Its halides are covalent polymers that are readilty hyrolysed or cleaved by donors. |
|
Answer» All are correct |
|
| 23. |
Which of the following name is correct :- |
|
Answer» 4-Isopropyl-3-methylhexane |
|
| 25. |
The schrodinger wave equation for hydrogen atom is Psi_2 = (1)/(4sqrt(2pi))((1)/(a_0))^(3//2) (2-(r)/(a_0))e^(-r//a_0) where a_0 is Bohr.s radius. If the radial node in 2s be at r_0 would be equal to |
|
Answer» `(a_0)/(2)` |
|
| 26. |
Which is/are correct statement? |
|
Answer» No corrosion takes PLACE in vaccum |
|
| 27. |
What is hydride gap ? |
| Answer» Solution :The REGION of the periodic table from groups 7-9 which does not FORMHYDRIDES is called the hydride GAP. | |
| 28. |
What is the relation of positive ion with their parent atom ? |
|
Answer» DOUBLE |
|
| 29. |
Which indicator should preferably be used for titration of NH_(4)OHwith HCl solution? |
| Answer» SOLUTION :METHYL ORANGE. | |
| 30. |
Which of the following can exist in three geometrical isomeric forms? |
|
Answer»
no. of GEOMETRICAL isomers = `2^(n-1) + 2^((n-2)/(2))` For b,c , No. of geometrical isomers = 3 |
|
| 31. |
Which of the following pairs of transition metal ions are the stronger oxidising agents in aqueous solutions ? |
|
Answer» `V^(2+)andCr^(2+)` `Mn^(3+)andCo^(3+)` can act as a STRONGER oxidising agents because their least oxidation state is +2. So they can get reduced from +3 oxidation state to +2oxidation state hence they act as strong oxidising agents. |
|
| 32. |
What is Octet rule ? Briefly explain its significance and limitations. |
| Answer» SOLUTION :ELEMENT s having 8 electrons in the outermost shell are stable. STABILITY of atoms is explained. Some molecules defy OCTET rule and are still stable., | |
| 33. |
Which of the following statements about a compound is incorrect ? |
|
Answer» A molecule of a compound has atoms of different elements. |
|
| 34. |
Which of the following is not correct |
|
Answer» low ionisation potential is a favourable condition for the fonnation of cation |
|
| 35. |
Which of the following conditions is not correct for resonating structures? |
|
Answer» the CONTRIBUTING structure MUST have the same number of UNPAIRED electrons. |
|
| 36. |
What is isomerism? Why ? |
| Answer» Solution :ISOMERISM of n-alkane in presence of anhydrous `AlCl_(3)` and HCl GAS, because ACCORDING to isomerism more STABLE chain isomer is obtained. | |
| 37. |
What is/are true about resonance ? |
|
Answer» The resonance structures are hypothetical |
|
| 38. |
Which of the following specleshave the sameshape ? |
|
Answer» `CO_(2)` |
|
| 39. |
The substance used as a adsorbentt in the column |
| Answer» Answer :C | |
| 40. |
Write the Born-Haber cycle for NaCl. |
Answer» Solution :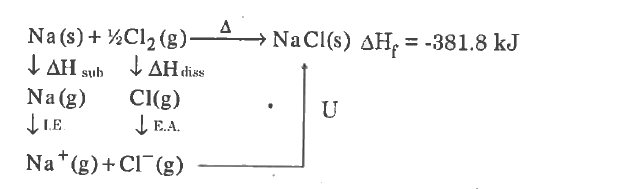 When U= Littice energuy `: Delta H_(s)` = Enthaply of SUBLIMATION,= Enthalpy of ionization, `Delta H_(4)=` enthalpy of dissociatio, `Delta H_(i)=DeltaH_(EA)` enthalpy for ELECTRON affinity. The only one step form formation of NaCl from sodium cholorine is associated with the energy called `Delta H_(f)` = = enthalpy of formation. `:. Delta H_(f)= U+DeltaH_(s)+DeltaH_(i)+DeltaH_(d)DeltaH_(Ea)` `:.` Lattice energy `U=DEltaH_(f)=[DeltaH_(s)+DeltaH_(i)+DeltaH_(d)+DeltaH_(Ea)]` |
|
| 41. |
What are quantum numbers? |
|
Answer» SOLUTION :The electron in an atom can be characterized by a set of four quantum numbers, namely principal quantum NUMBER (n), azimuthal quantum number (I), magnetic quantum number (m) and spin quantum number (s). (ii) When Schrödinger equationis solved for a wave FUNCTION `Psi` the solution CONTAINS the first three quantum numbers n, I and m. (iii) The FOURTH quantum number arises due to the spinning of the electron about its own axis |
|
| 42. |
Which of the following compound(s) on reductive ozonolysis (O_3 / /Zn-H_2O) would produce octane-2,4,5,7- tetraone ? |
|
Answer»

|
|
| 43. |
Which among the following compounds does not show hydrogen bonding? |
|
Answer» Ammonia |
|
| 44. |
underset(("Hydrated salt"))((A))underset("heated")overset("Strongly")rarr ubrace(B+C)_("Transparent glassy bead") H_(2)BO_(3) overset("Strongly")underset("heated")rarr C+D How many X-O-X linkages are present in structure of A(X = central atom)? |
|
Answer» 4 |
|
| 45. |
Which of the following oxides is acidic in nature ? |
|
Answer» `B_2O_3` `underset"Acidic"(B_2O_3)""underset"Amphoteric" (Al_2O_3 " and " Ga_2O_3) "" underset"Basic"(In_2O_3" and " Tl_2O_3)` |
|
| 46. |
Which of the following relates to photons both as wave motion and as a stream of particles |
|
Answer» Interference |
|
| 47. |
underset(("Hydrated salt"))((A))underset("heated")overset("Strongly")rarr ubrace(B+C)_("Transparent glassy bead") H_(2)BO_(3) overset("Strongly")underset("heated")rarr C+D Find the number of tetrahedral and trigonal planar units in structure of A: |
|
Answer» 2,1 |
|
| 48. |
When you freeze water in your freezer to make ice cubes,the amount of order in the molecules ofwater increases. However , second law of thermodynamics says that the amountof order in the isolated system can only stay constant or decrease with time. How can thus making of iceviolate second law ? |
|
Answer» because water expands during ice formation |
|
| 49. |
underset(("Hydrated salt"))((A))underset("heated")overset("Strongly")rarr ubrace(B+C)_("Transparent glassy bead") H_(2)BO_(3) overset("Strongly")underset("heated")rarr C+D Identify C : |
|
Answer» `(BN)_(X)` |
|
| 50. |
Uup means having atomic number of element is….. |
|
Answer» 105 |
|