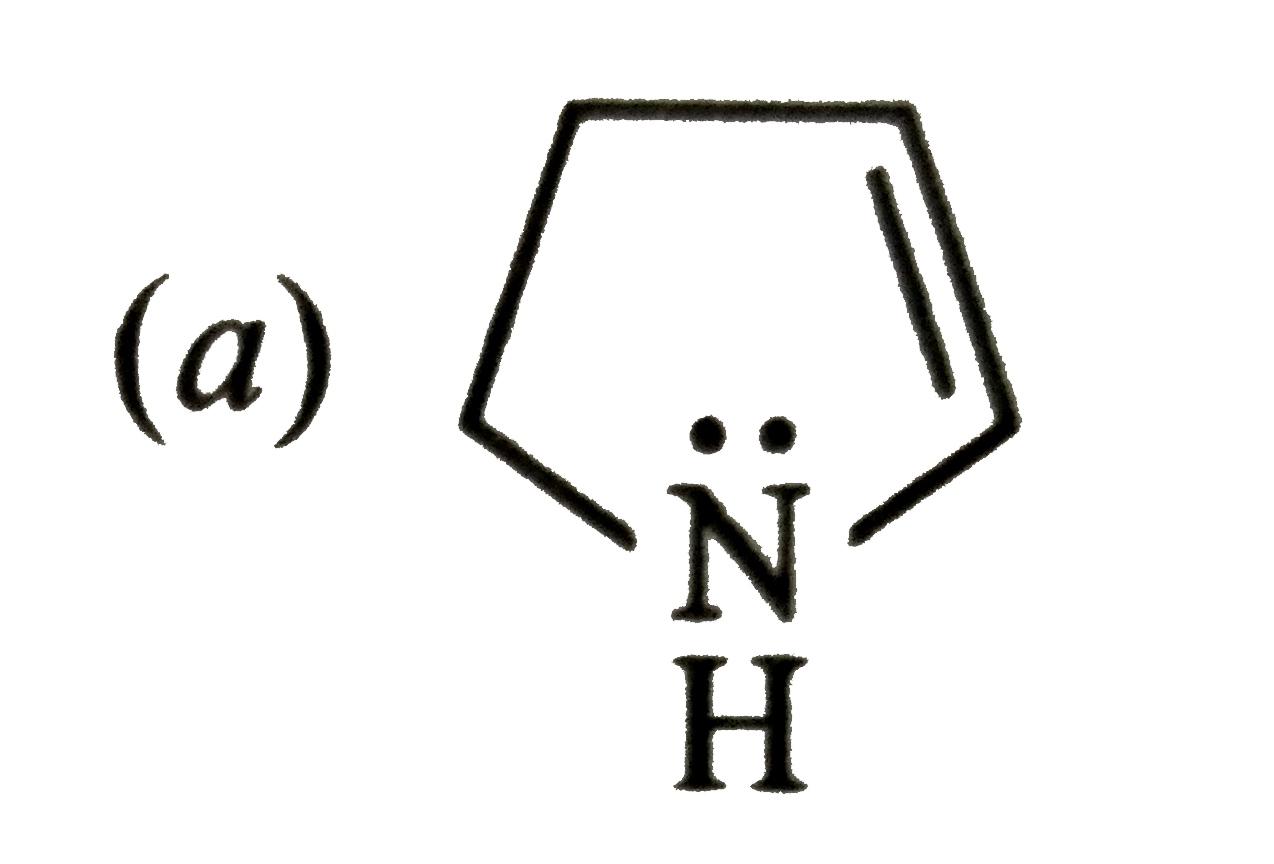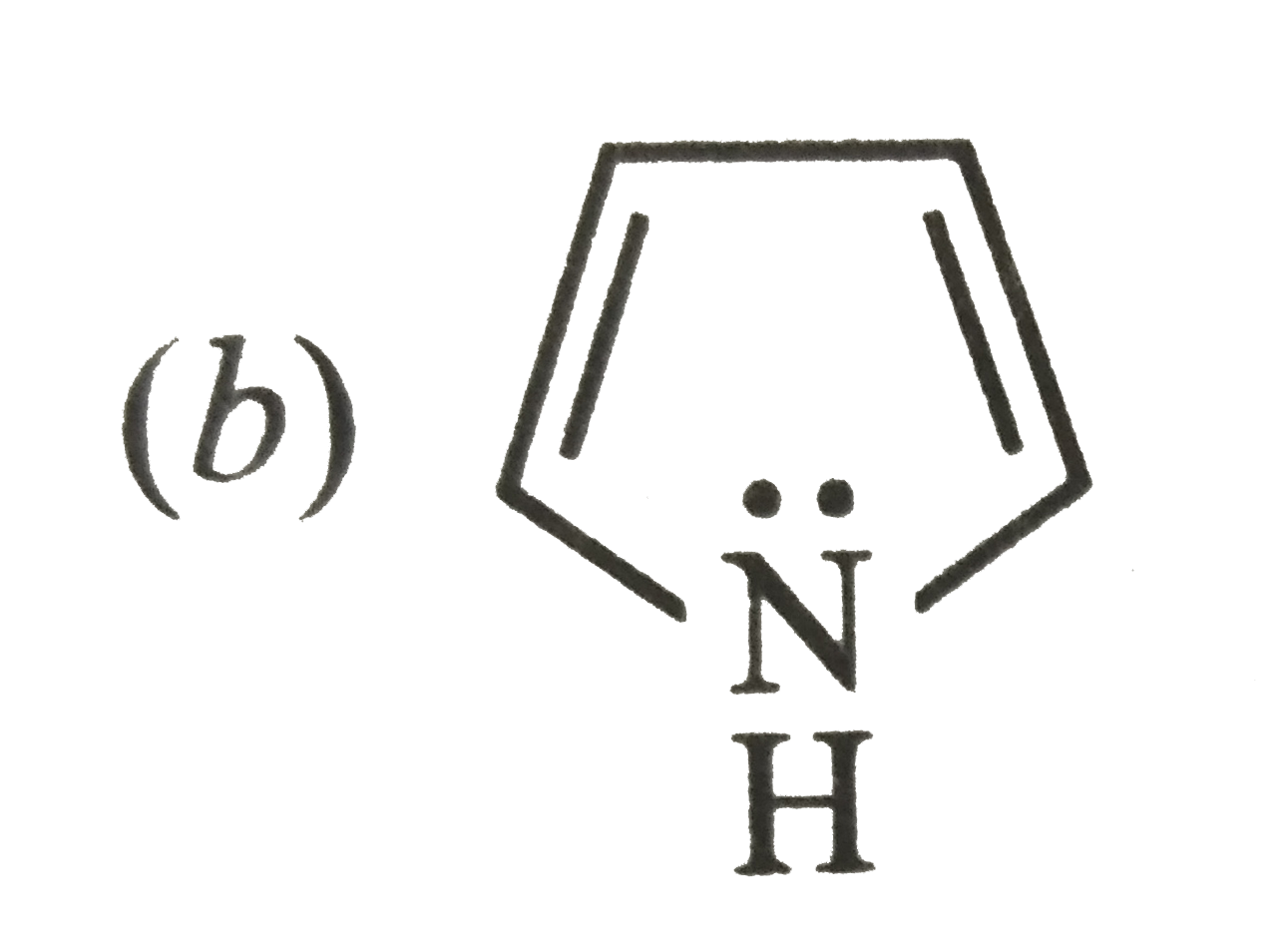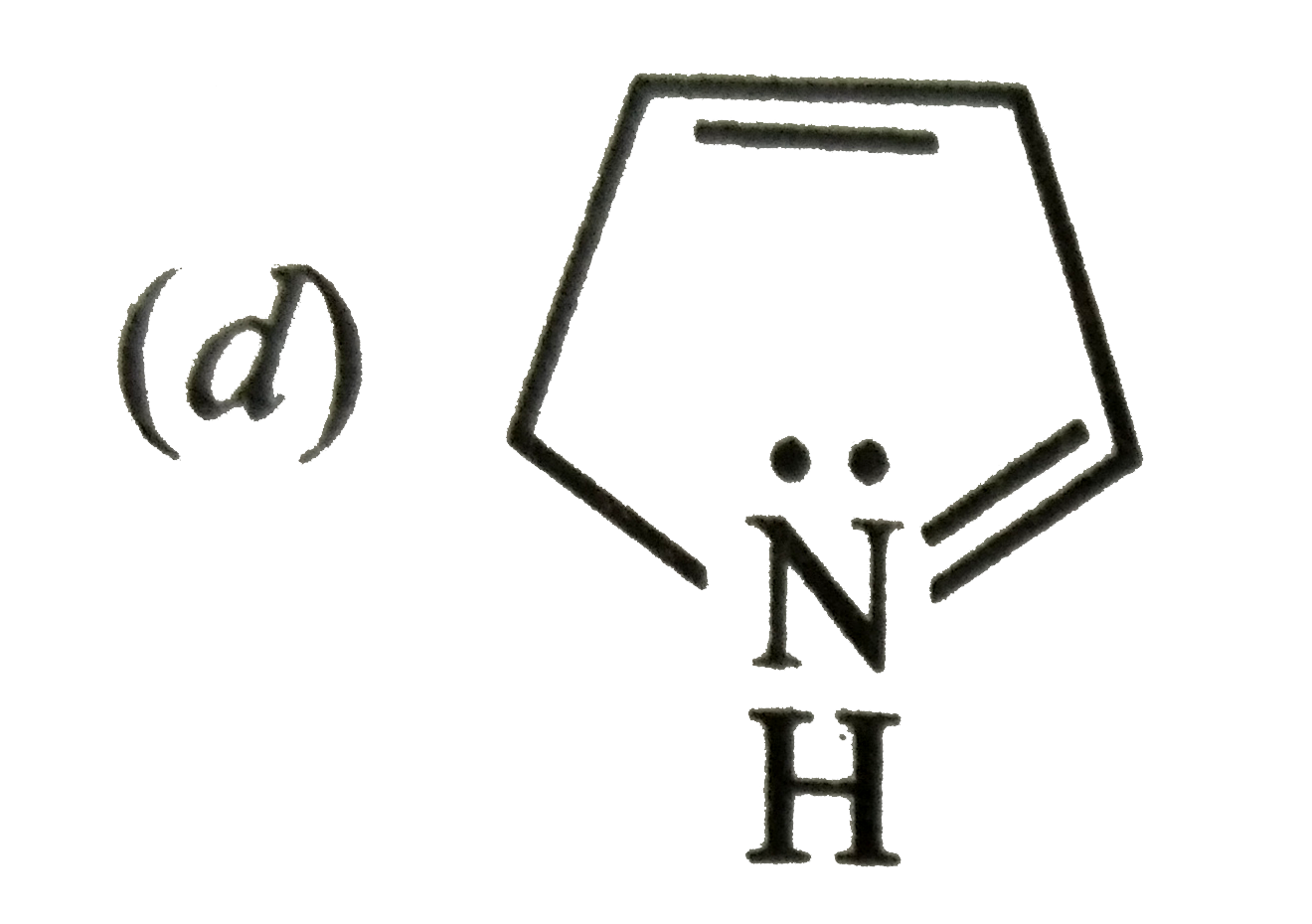Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which property of colloids is not dependent on thhe charge on colloidal particles |
|
Answer» Electro-osmosis |
|
| 2. |
The wrong statement about glycerol is |
|
Answer» It is a trihydric alcohol |
|
| 3. |
Which of the following have same number of electron in the outer most orbit? |
| Answer» Solution :As, Bi both are group-15 elements. | |
| 4. |
What is NTP or STP and SATP ? Mention its volume. |
|
Answer» Solution :STP means `0^(@)C` (273.15 K) temperature and 1 atm. or (101.32 kPa) PRESSURE. At STP molar volume of an ideal gas or a combination of ideal gases is 22.413996 L `mol^(-1)`. SATP means STANDARD AMBIENT Temperature and Pressure means 298.15 K and 1 bar `(10^(5)PA)` pressure. ..SATP is at 1 bar pressure and 298.25 K tempr-rature volume of ideal gas is 24.789 L `mol^(-1)`... |
|
| 5. |
Which one of the following is an ionic or saline hydride? |
| Answer» Answer :d | |
| 6. |
Two organic compounds have same empirical formula but different molecular formula, they must have |
|
Answer» different percentage composition |
|
| 7. |
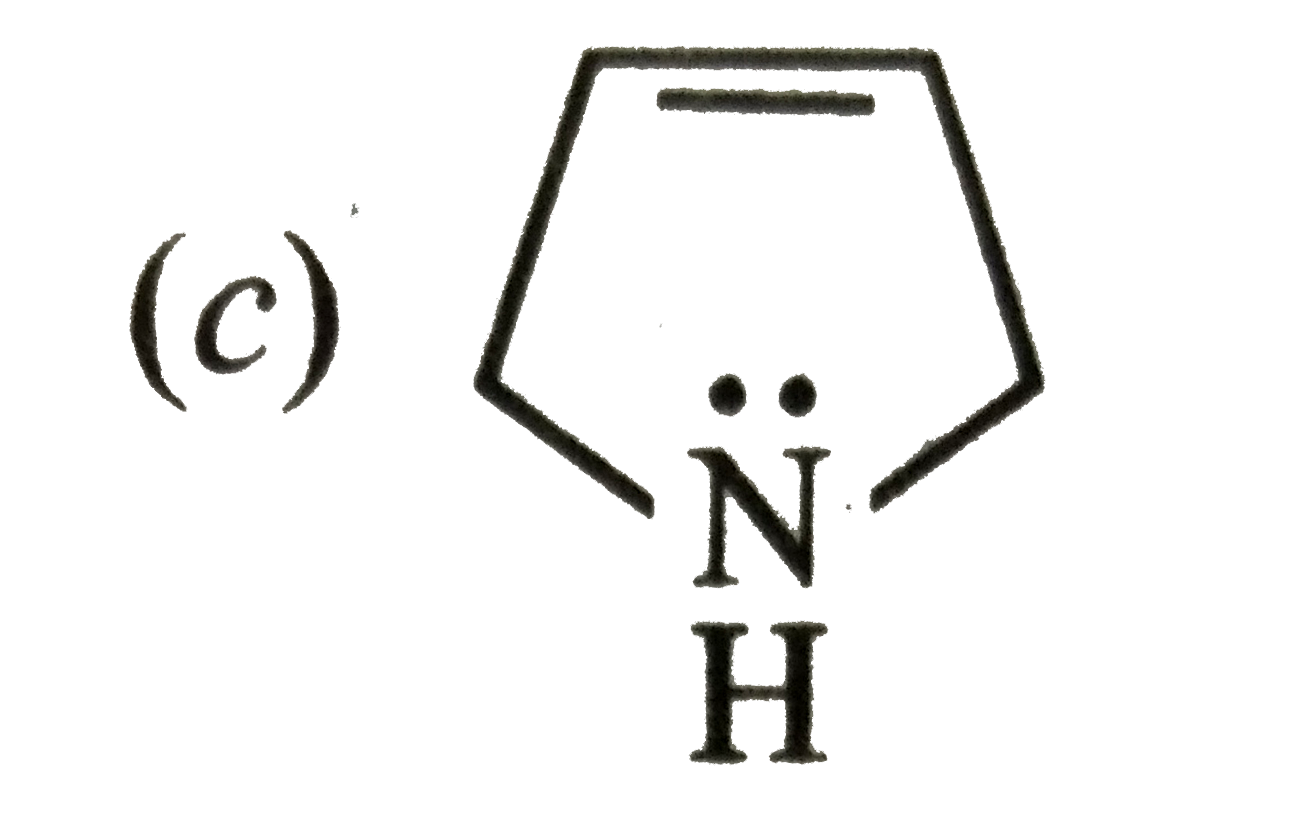
Which of the following is not a resonance structure of CH_(2)= CH-CHO ? |
|
Answer» `CH_(2) = CH- OVERSET(overset( :O: )(||))(C )-H` |
|
| 8. |
Write Lewis dot symbols for atoms of thefollowing elements : Mg, Na, B, O, N , Br |
Answer» SOLUTION :
|
|
| 9. |
Which ion is displaced by Na^+ ions when hard water is passed over Zeolite ? |
|
Answer» `OH^-` |
|
| 11. |
What are Lewis structures? Write the Lewis structure of H_(2), BeF_(2) and H_(2)O. |
|
Answer» Solution :The outer shell electrons are SHOWN as dots surrounding the symbol of the atom. These symbols are known as Lewis symbols or Lewis structures. The Lewis STRUCTURE of (i) `H_(2) - H : H` (ii) `BeF_(2) - :underset(* *)overset(* *)F:Be:underset(* *)overset(* *)F:` 
|
|
| 12. |
The solution of KOH in ethyl alcohol contains |
|
Answer» `C_(2)H_(4)` |
|
| 13. |
When a mixture of NaBr and NaCl is separately digested with H_2SO_4 all the halogens are expelled and Na_2SO_4 is formed quantitatively with a particular mixture, it was found that the weight of Na_2SO_4 obtained was precisely the same as the weight of NaBr+NaCl mixture taken. Calculate the ratio of the weight of NaCl and NaBr in the mixture. |
|
Answer» SOLUTION :Let the weight of `NaCl` be x g and the weight of NABR be y g. ,brgt We have to CALCULATE `(x)/(y)` `2NaCl-=Na_2SO_4` `2xx58.5g of NaCl-=142g of Na_2SO_4` `x g of NaCl-=(142)/(2xx58.4)XX x g of Na_2SO_4` `2NaBr-=Na_2SO_4` `2xx103g of NaBr-=142 g of Na_2SO_4` `y g of NaBr-=(142)/(2xx103)xx x g of Na_2SO_4` `therefore(142X)/(2xx58.5)+(142x)/(2xx103)=x+y` `1.21x-x=y-0.69y` `0.21x=0.31y` `(x)/(y)=(0.31)/(0.21)=1.475` The ratio of weights of `NaCl:NaBr` is `1.476:1` |
|
| 14. |
Which are of the following is an exothermic reaction? |
|
Answer» `N_(2(g)) + O_(2(g)) + 180.8 k.J rarr 2NO_((g))` |
|
| 15. |
Which sequence for greenhouse gases is based on GWP? |
|
Answer» `CFC gt N_(2)O gt CO_(2) gt CH_(4)` |
|
| 16. |
Which of the following is incorrect? |
|
Answer» RMS VELOCITY depends upon molecular weight |
|
| 17. |
Which of the following would NOT be a product from the Friedel-Crafts alkylation of benzene with1-bromopropane? |
| Answer» Solution :Friedel craft.s REACTION with rearrangement of carbonium ion | |
| 18. |
Which of the following property is extensive property ? |
|
Answer» BOILING point |
|
| 19. |
What is Psi ? |
| Answer» Solution :According to quantum mechanism `Psi` is wave function of molecular ORBITALS and represent the AMPLITUDES of electron wave. | |
| 20. |
What happens when methane reacts with conc. HNO_(3) at high temperature? |
|
Answer» NITROMETHANE is FORMED |
|
| 21. |
What is the maximum concentration of equimolar solutions of ferrous sulphate and sodium sulphide so that when mixed in equal volumes , there is no precipitation of iron sulphide ?(For iron sulphide , K_(sp)=6.3xx10^(-18) ) |
|
Answer» Solution :(i)Concentrationin x M Ferrous sulphate solution : `{:(FeSO_4 to , Fe^(2+) +,SO_4^(2-)),(x,xM,xM):}` (ii) Concentration in x M sodium sulphide : `{:(Na_2S to , 2Na_((AQ))^(+)+,SO_(4(aq))^(2-)),(x,"2X M","x M"):}` FeS is form by same concentration of solution(i) and (ii) in it. `[Fe^(2+)]=x/2 , [S^(2-]=x/2` `{:(FeS_((s))hArr , Fe^(2+) +, S^(2-)),(,x/2M,x/2M):}` `therefore K_(sp) (FeS)=6.3xx10^(-18)=(x/2)(x/2)` `therefore x^2=4xx6.3xx10^(-18) = 25.2xx10^(-18)` so `x=sqrt(25.2xx10^(-18))=5.02xx10^(-9)` M so , concentration `x=[Na_2S]=[FeSO_4]` `=5.02xx10^(-9)` M |
|
| 22. |
You have a mixture of three liquids A, B and C. There is a large difference in the boiling points of A and rest of the two liquids i.e., B and C. Boiling point of liquids B and C are quite close. Liquid A boils at a higher temperature than B and C and boiling point of B is lower than C. How will you separate the components of the mixture. Draw a diagram showing set up the apparatus for the process. |
|
Answer» Solution :Since the boiling point of liquid A is MUCH higher than those of liquids B and C WHOSE boiling points are quite close, therefore, separate liquid A by simple distillation USING the apparatus shown in Fig. Since the boiling points of liquids B and C are quite close but are much lower than that of liquid A, therefore, mixture of liquids B and C will distill together LEAVING behind liquid A. On further heating liquid A will distil over. Now place the mixture of liquids B and C in a flask fitted with a fractionating column as shown in Fig. Since the b.p. of liquid B is lower than that of liquid C, therefore, on FRACTIONAL distillation, first liquid B will distill over andthen liquid C. |
|
| 23. |
Which of the following are incorrect statements ? |
|
Answer» `BH_(3)` is stable compound |
|
| 24. |
Weight percentage of silica in portland cement is |
|
Answer» 12 |
|
| 25. |
Which of the following is correctly matched? |
|
Answer» tartaric ACID-geometrical ISOMERISM  Lactic acid is optically active due to the presence of CHIRAL CENTRE. |
|
| 26. |
Which of the following is true about the number of molecules in A and B? |
|
Answer» Flask `A` contains EIGHT TIMES more molecules than flasks `B`. `n_(A) prop N`, `:. (N_(A))/(N_(B))=8` (`n=` number of moles)(`N=` number of molecules) |
|
| 27. |
Which of the following mixture of gases is called water gas? |
|
Answer» `CO_(2(G))+H_(2(g))` |
|
| 28. |
Which of the following statement is "NOT" correct about 6.8% strengh of H_2O_2 |
|
Answer» Its normality is 4N volume strength =`11.2 XM` |
|
| 29. |
Which ofthe following compounds will not be soluble in sodium bicarbonate ? |
|
Answer» 2,4,6-Trinitrophenol |
|
| 30. |
Write Friedel -Craft's reation . |
Answer» SOLUTION :
|
|
| 31. |
The sinle quantum number by Neil.s Bohr and the appearance of several spectral lines in a particular series of Hspectrum, suggest that there more than one quantum to explain all the quantized properties of an electron in a particular energy level inside the atom. If fact when the properties of the electron is transformed from Cartesian co-ordinate to polar co-ordinates then it becomes the function of r, theta and phi suggesting three independet quantum numbers. In addition to this a fourth quantum number is required to consider the spinning behaviour of an electron. The principal quantum number n suggest the orbit number in which electron revolves, Azimuthal quantum number 1 suggest the shape of orbitals, magnetic quantum number m, gives orientation of orbital in presence of external magnetic field, while spin quantum number s, gives direction of an electron about its own axis n != 0 values of "1" ranges in between 0 to (n-1), while m depends upon 1. Total number values of m = 21+1, and "m" ranges in between -1 to +1 including 0. For each electron spin will be +-(1)/(2) The two unpaired electron present in C-atom are different w.r.t their |
|
Answer» n |
|
| 32. |
Write the application of equilibrium constant? |
|
Answer» Solution :1. predict the direction in which the net reaction will take place 2. predict the EXTENT of the reaction and 3. calculate the equilibrium concentrations of the reactants and PRODUCTS. |
|
| 33. |
Which of the following statements is false regarding alkali metals |
|
Answer» ALKALI METALS are SOFT and can be cut with the help of knife |
|
| 34. |
The sinle quantum number by Neil.s Bohr and the appearance of several spectral lines in a particular series of Hspectrum, suggest that there more than one quantum to explain all the quantized properties of an electron in a particular energy level inside the atom. If fact when the properties of the electron is transformed from Cartesian co-ordinate to polar co-ordinates then it becomes the function of r, theta and phi suggesting three independet quantum numbers. In addition to this a fourth quantum number is required to consider the spinning behaviour of an electron. The principal quantum number n suggest the orbit number in which electron revolves, Azimuthal quantum number 1 suggest the shape of orbitals, magnetic quantum number m, gives orientation of orbital in presence of external magnetic field, while spin quantum number s, gives direction of an electron about its own axis n != 0 values of "1" ranges in between 0 to (n-1), while m depends upon 1. Total number values of m = 21+1, and "m" ranges in between -1 to +1 including 0. For each electron spin will be +-(1)/(2) d-orbital may contain maximum of 10 electron as for d-orbital |
|
Answer» l=2 |
|
| 35. |
Which of the following statement is/are correct |
|
Answer» The weight of `CaCO_(3)` in the ORIGINAL MIXTURE is `0.5 g`. `((1 "MOL" CO_(2))/(44 g CO_(2)))((1 "mol" CaCO_(3))/("mol"CO_(2)))` `((100 g CaCO_(3))/("mol" CaCO_(3)))` `= (0.22 xx 100)/(44) = 0.5 g CaCO_(3)` b. Moles of `CaCO_(3)=` moles of `Ca` `= ((0.22)/(44)) = 0.005 "mol"` Weight of `Ca = 0.005 xx 40 = 0.2 g Ca` d. % of `Ca = (0.2)/(1.0) xx 100 = 20% Ca` ltbgt Hence, (c ) wrong. |
|
| 36. |
Which is true regarding nitrogen ? |
|
Answer» Less electronegativity |
|
| 37. |
Which group of the periodic table contains only metals |
| Answer» ANSWER :d | |
| 38. |
Which of the statements are correct about above reaction? |
|
Answer» (i) and (V) both are nucleophiles. |
|
| 40. |
What happens when calcium oxide is heated with a ammonium chloride? |
| Answer» Solution :It liberates AMMONIA gas `CaO + 2NH_(4) CI OVERSET(Delta)(to) CaCI_(2) + 2NH_(2) uarr + H_(2) O` | |
| 41. |
Write the Van't Hoff equation of osmotic pressure. |
|
Answer» Solution :Van.t Hoff equation states that for DILUTE solutions, the osmotic pressure is DIRECTLY proportional to the molar CONCENTRATION of the solute and the temperature of the solution. `pi=CRT, ` where `pi=` Osmotic pressure, C = concentration, T =Temperature, R=gas CONSTANT |
|
| 42. |
Water is considered as pure if it has BOD less than …ppm |
|
Answer» |
|
| 43. |
Which of the following electrodes will act as anodes when connected to standard hydrogen electrode? |
| Answer» SOLUTION :All ELECTRODES which have negative electrode potential are stronger reducing agent than `H_(2)` gas and `FE^(2+//Fe(E^(@)=-0.44v)` acts as anodes i.e OPTION (a) and (b) are correct | |
| 44. |
Which of the product is obtained in the following reaction : |
|
Answer» |
|
| 45. |
Which of the following has square pyramidal structure? |
|
Answer» `BrF_(5)` |
|
| 46. |
What are the products formed in the following reaction : C_(6)H_(5) - COO - CH_(3) underset(2. H_(3)O^(+))overset(LiAlH_(4))rarr |
|
Answer» `C_(6)H_(5)CH_(2)OH " and " CH_(3)OH` |
|
| 47. |
Which of the following statements is not true for a lyophilic sol |
|
Answer» it can be EASILY solvated |
|
| 48. |
Write with a balanced chemical equation , how gypsum is used for the conversion of ammonia into ammonium sulphate without using H_(2)SO_(4). |
|
Answer» SOLUTION :A suspension of gypsum when saturated with ammonia and CARBON dioxide forms ammonium sulphate `2NH_(3) + CaSO_(4) + CO_(2) + H_(2) O to (NH_(4))_(2)SO_(4) + CaCO_(3)` |
|
| 49. |
Two electrons occupying the same orbital are distinguished by …………. . |
|
Answer» azimuthal QUANTUM NUMBER For the first electron MS `= +½` For the SECOND electron ms `= -½` |
|