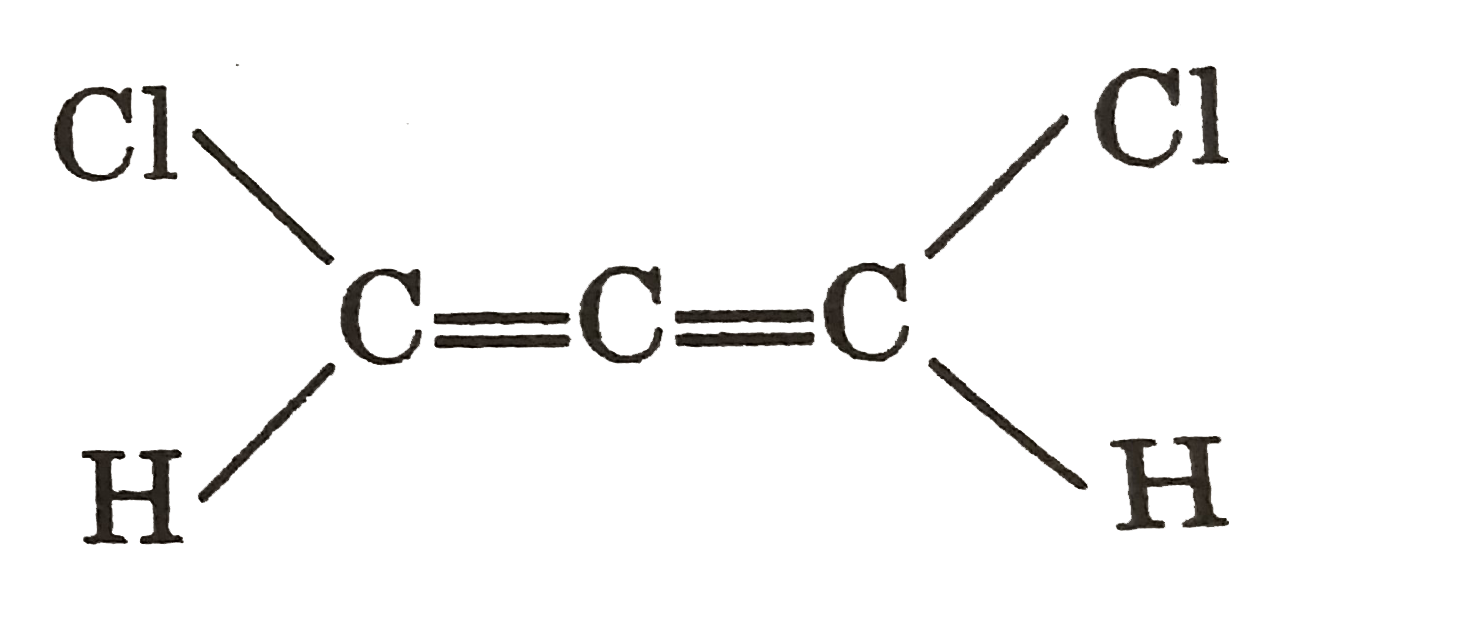Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which of the following statement about the electron is incorrect ? |
|
Answer» It is a NEGATIVELY charged particle |
|
| 2. |
Thermodynamics mainly deals with |
|
Answer» interrelation of various forms of energy and their transformation from one formto another. |
|
| 3. |
Which of the following ions exhibit resonance ? |
|
Answer» `NO_(3)^(-)` 
|
|
| 4. |
Which one is green house gas ? |
|
Answer» `CH_4` |
|
| 5. |
Which nitrate will decompose to give NO_(2) on heating? |
|
Answer» `NaNO_(3)` `2NaNO_(3)+ overset(Delta)to 2NaNO_(2)+O_(2)` |
|
| 6. |
Which of the following represents the correct IUPAC name for the compounds concerned? 2,4,7-Trimethyloctane or 2,5,7-Trimethyloctane |
|
Answer» |
|
| 7. |
Which of the following belongs to the homologous series of C_(5)H_(8)O_(2)N? |
|
Answer» `C_(6)H_(10)O_(3)N` |
|
| 8. |
Which among the following is not an exact differential? |
|
Answer» Q (dQ= heat absorbed) |
|
| 9. |
What is the oxidation number of phosphorous in calcium phosphide ? |
|
Answer» <P>`-5` `therefore3(+2)+2(x)=0` `therefore2x=-6" "thereforex=-3` |
|
| 10. |
When 20.0 g of ammonium nitrate( NH_(4)NO_(3)) is dissolvedin 125g of waterin a coffee-cup calorimeter, the temperature fallsfrom 296.5 K to 286.4K. Findthe value of q for the calorimeter. (Treat heat capacityof water as the heat capacityof the calorimeter and its contents) . |
|
Answer» `125xx ( 296.5 - 286.4) XX 4.18 J= 5282J = 5.282J` |
|
| 11. |
Which sequence for green house gases is truely based on GWP? |
|
Answer» `CFC GT N_2 O gt CO_2 gt CH_4` |
|
| 12. |
Write the relatioship between K_(sp) and S of BaSO_(4) and AgCl. |
|
Answer» Solution :Both `BaSO_(4)` and AgCl belong to AB type of salts. (a) Let the solubility of AgCl be S MOL `DM^(-3)` `AgCl_((s))hArrAg_((aq))^(+)+Cl_((aq))^(-)` `[Ag^(+)]=S" mol "dm^(-3)and[Cl^(-)]=S" mol "dm^(-3)` `thereforeK_(sp)=[Ag^(+)][Cl^(-)]=S*S=S^(2)` (B) Let the solubility of `BaSO_(4)` be S mol `dm^(-3)` `BaSO_(4(s))hArrBa_((aq))^(2+)+SO_(4(aq))^(2-)` `[Ba^(2+)]=S" mol "dm^(-3)and[SO_(4)^(2-)]=S" mol "dm^(-3)` `thereforeK_(sp)=[Ba^(2+)]*[SO_(4)^(2-)]=S*S=S^(2)` |
|
| 13. |
What will be happened when organic compound fusion with sodium metal? |
| Answer» Solution :The conversion of N, S, X in COVALENT bond to IONIC bond COMPOUND | |
| 14. |
The velocity of an electron in a certain Bohr.s orbit of H-atom bears the ratio 1:275 to the velocity of light. Then find the quantum number (n) of orbit ? |
|
Answer» |
|
| 15. |
Which of the following alkenes is most reactive towai cationic polymerization? |
|
Answer» |
|
| 16. |
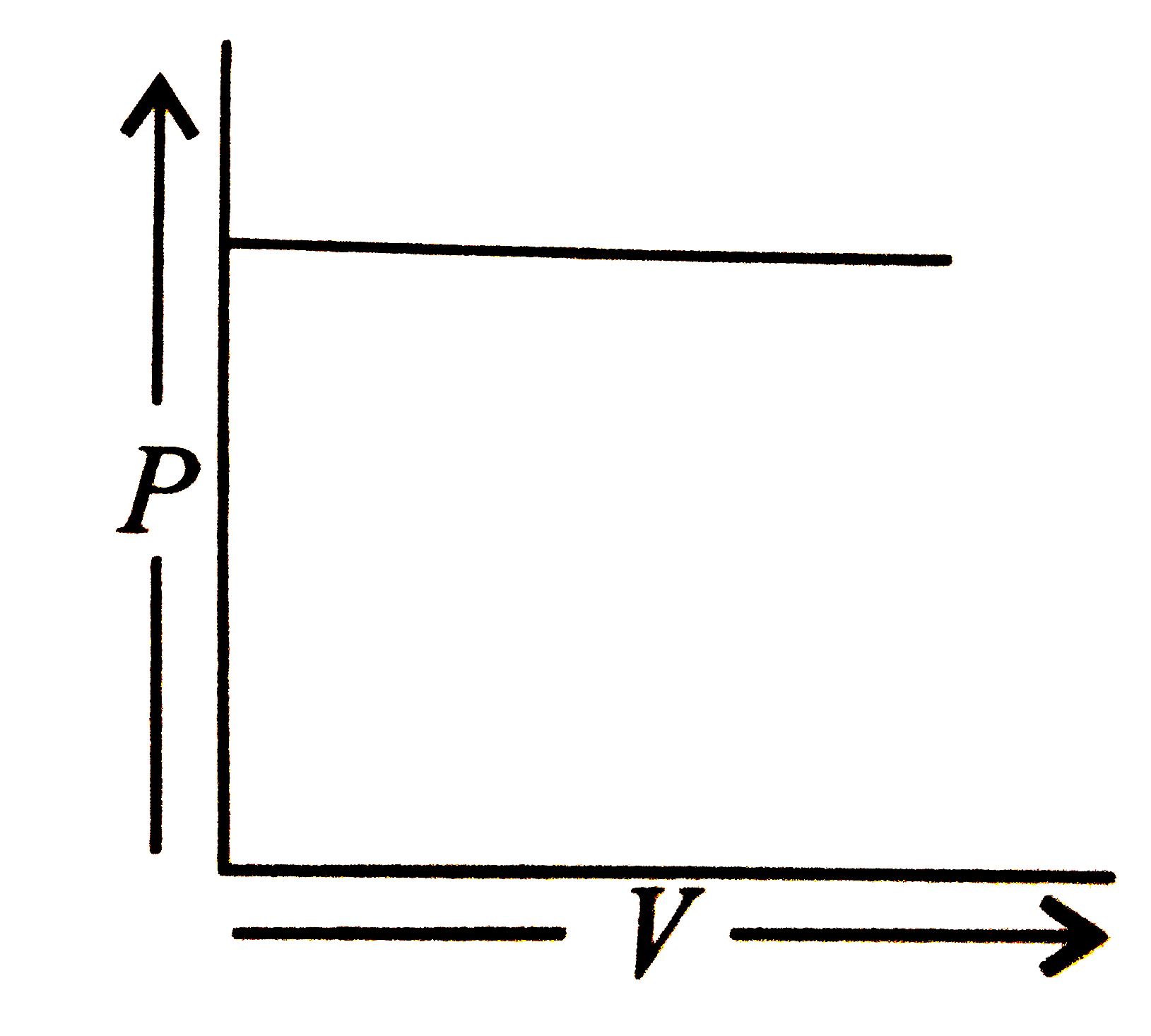
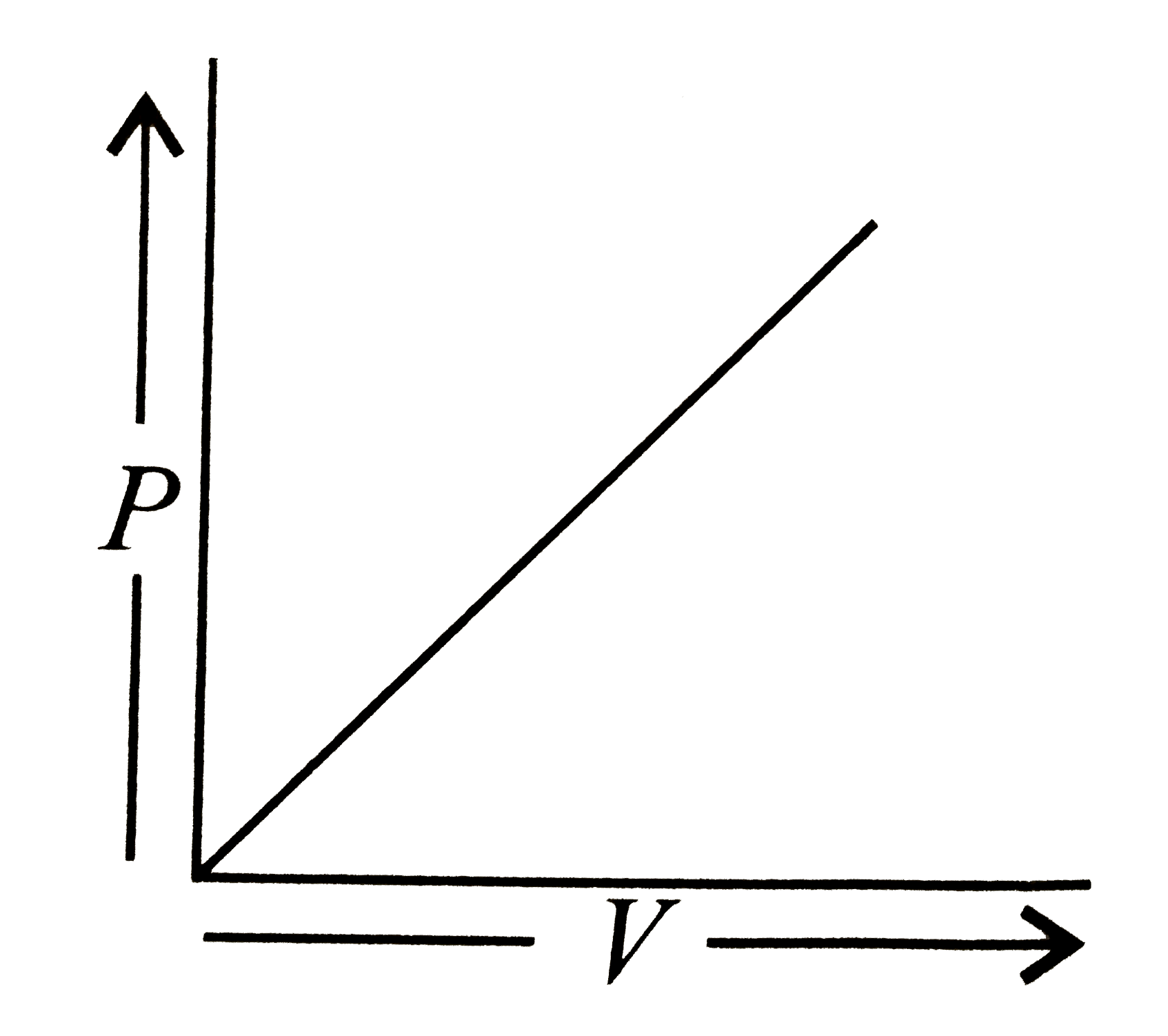
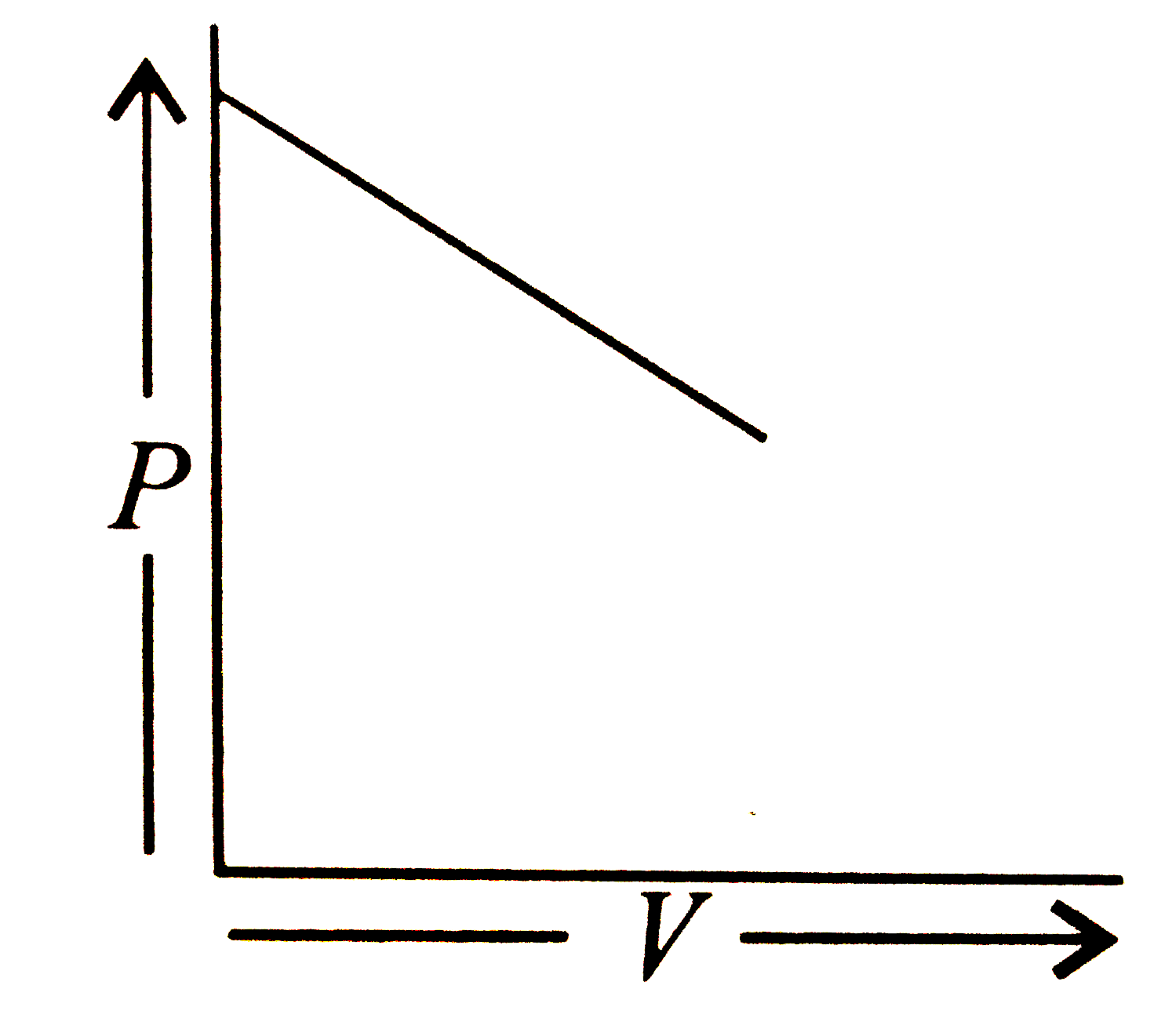
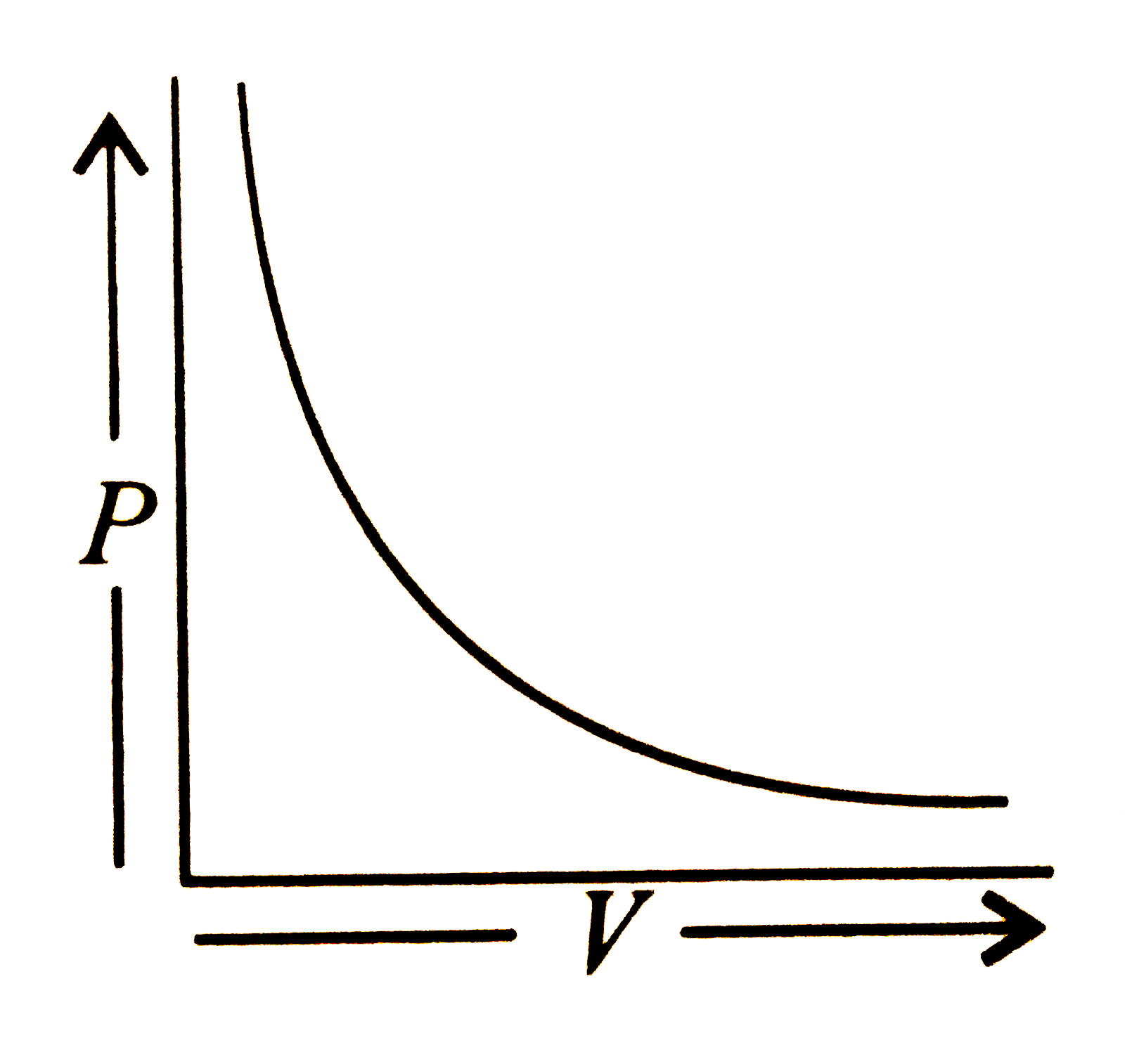
Which of the following is according to Boyle's law? |
|
Answer»
|
|
| 17. |
Which oneof the following binary liquid mixtures exhibits positive deviation from Raoults law? |
|
Answer» Acetone + CHLOROFORM |
|
| 18. |
Which one of the following is not used for the purification of solid impurities ? |
|
Answer» Distillation |
|
| 19. |
The structure of sodium chloride crystal is: |
|
Answer» body centred cubic LATTICE |
|
| 20. |
Which of the following is/are correct |
|
Answer»
But in option (C) and (D) `CH_(3)-OVERSET(O)overset(||)(C)_(o+)` is ELECTROPHILE. |
|
| 21. |
Which type of force present between HCl and O_(2) molecules ? |
|
Answer» London |
|
| 22. |
Which of the following products may be formed by the reaction of diborane with ammonia/ |
|
Answer» `(BN)_(X)` |
|
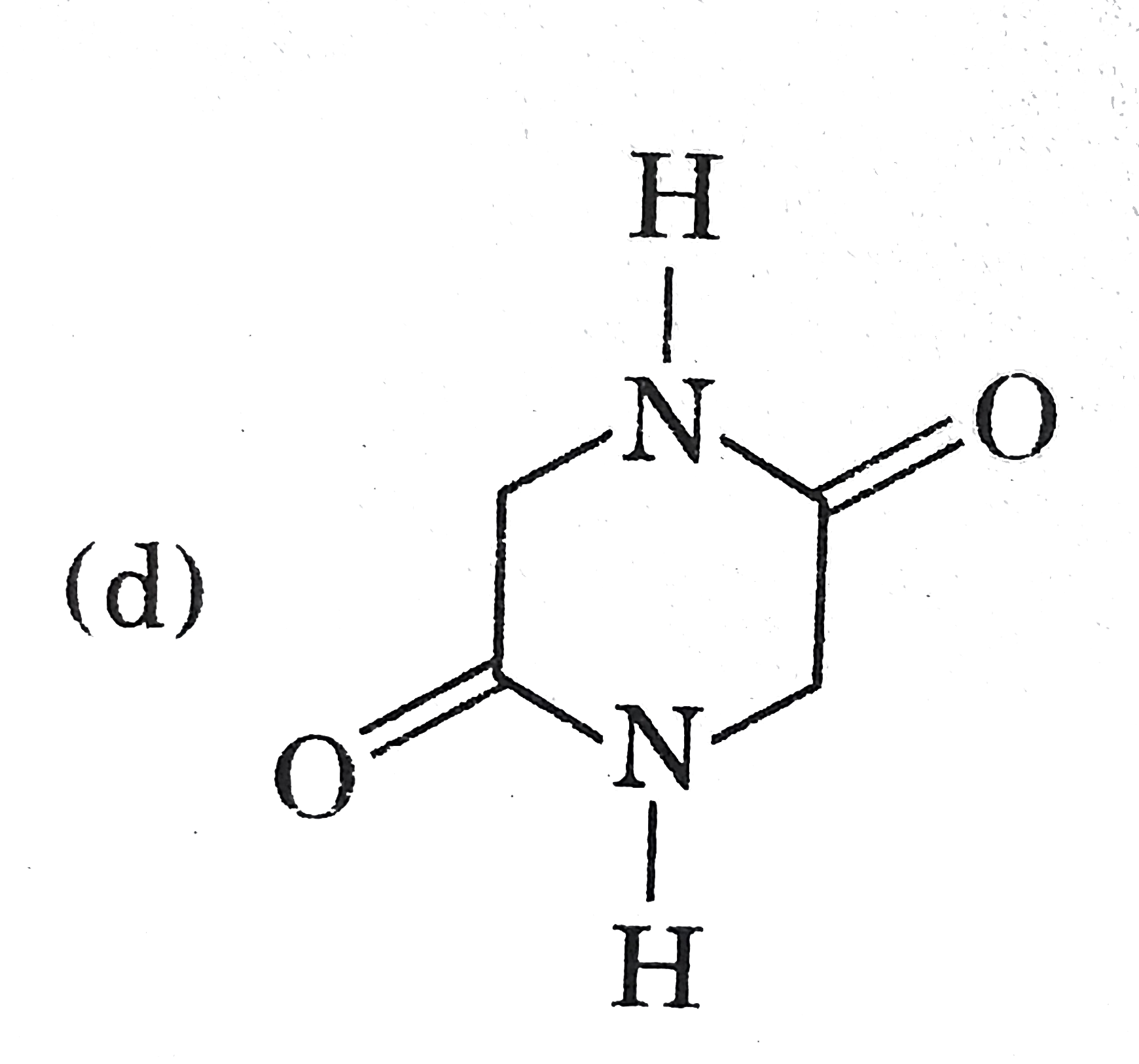
| 23. |
Which compound (s) will show the geometrical isomerism? |
|
Answer»
|
|
| 24. |
Which of the following is not an non-ideal solution showing negative deviation? |
|
Answer» PHENOL and ANILINE |
|
| 25. |
Which of the following compounds has correct IUPAC name? |
|
Answer» `CH_(3) - CH_(2) - CH_(2) - COO- CH_(2) CH_(3) RARR` butanal |
|
| 26. |
What is Troposphere ? |
| Answer» SOLUTION :Throughout our life we REMAIN immersed in the ocean of air which is a mixture of GASES. We spend our life in the LOWERMOST layer of the atmosphere called .troposphere.. | |
| 27. |
What would be the consequence of presence of following in water ? (i)Sulphates gt 500 ppm (ii)Fluorides between 2 ppm -10 ppm gt 10 ppm (iii)Nitrates gt 50 ppm |
|
Answer» Solution :(i)Sulphates GT 500 PPM have laxative effect (ii)Fluorides gt 10 ppm cause brown MOTTLING of TEETH Fluorides gt 10 ppm are harmful to bones and teeth (iii) Nitrates gt 50 ppm cause methemoglobinemia (blue baby syndrome) |
|
| 28. |
Which of the following has highest conductivity in aqueous solution ? |
|
Answer» `LI^(+)" ion"` |
|
| 29. |
When n=3 and l=1, the designation given to the orbital is |
|
Answer» 4s |
|
| 30. |
Which of the following are the ores of lead ? |
|
Answer» GALENA Cassiterite, `SnO_(2)` Anglesite, `PbSO_(3)` Cerussite `PbCO_(3)` (white LEAD ore) |
|
| 31. |
Which of the following statements is false regarding alkali metals ? |
|
Answer» ALKALI metals are soft and can be cut with the help of knife |
|
| 32. |
Write the IUPAC names of the following substituted benzene derivatives: |
| Answer» SOLUTION :(a) 1, 4-Dimethylbenzene (B) 2, 4,6-Trinitrophenol (C ) 3-Phenyl-2-propenal | |
| 33. |
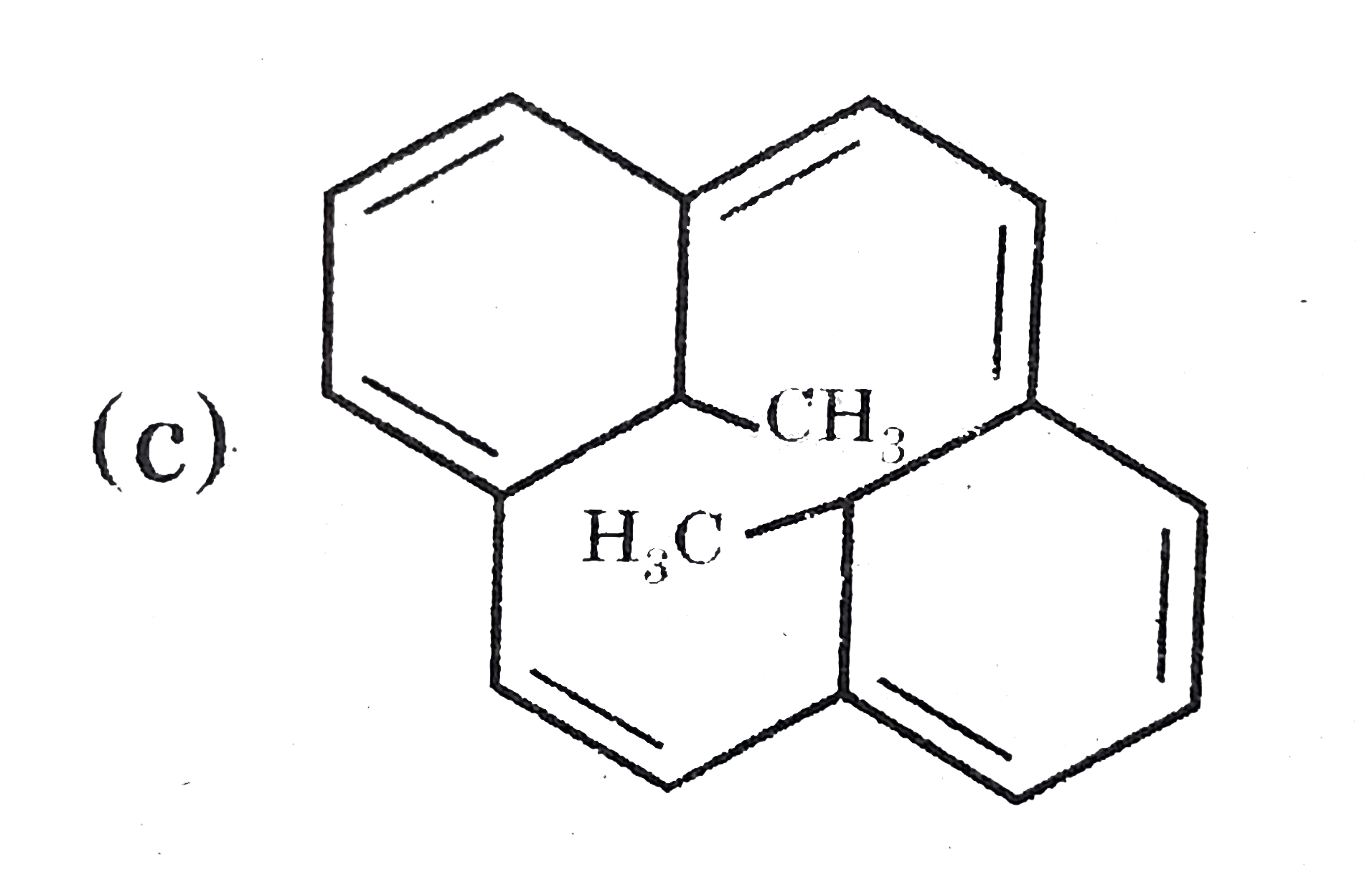
Which of the following structures is not correctly named according of IUPAC ? |
|
Answer»
|
|
| 34. |
Usings,p,dfnotationsdescribethe orbitalwith thefollowingquantumnumber (a )n=2,l =1 ( b)n=4,l =0 (c ) n=5,l =3(d )n=3 , l= 2 |
Answer» SOLUTION :
|
|
| 35. |
Which of the following pair of oxides is responsible for acid rain? |
|
Answer» `SO_(3)+NO_(2)` |
|
| 36. |
When sodium hydroxide is added drop wise to a mixture of two compounds a white precipitate appears but on adding excess of NaOH the amount of precipitate decreases . Then the mixture contains |
|
Answer» NaOH and `FeSO_(4)` |
|
| 37. |
Which of the following process is feasible at all temperatures? |
|
Answer» `DeltaH GT 0, DeltaS gt 0` |
|
| 38. |
Two liquids (A) and (B) can be separated by the method of fractional distillation. The boiling point of liquid (A) is less than boiling point of Liquid (B). Which of the liquids do you expect to come out first in the distillate ? Explain |
|
Answer» Solution :The less boiling point containing liquid (A) comes first in the distillate. Fractional distillation is used to separate the components of a mixture if their boiling points differ by `20^(@)` or less. In this method, a FRACTIONATING column is used in between the flask and the CONDENSER. The purpose of the fractionating column is to provide hurdles for the ascending vapours and to provide hurdles for the ascending vapours and to provide large surface area for condensing the high boiling liquid. As a result, the vapour of the LOW boiling liquid (A) will move up while these of the high boiling liquid will condense adn back into the flask. Therefore, in this method liquid (A) with lower boiling point will distil first and liquid (B) with HIGHER boiling point afterwards |
|
| 39. |
What happened in hyperconjugation? |
| Answer» Solution :The `SIGMA` ELECTRONS of C-H BOND is delocalized and the stability of MOLECULE or ion will be INCREASES | |
| 40. |
Which one of the following shows geometrical isomerism? |
|
Answer» n-Butane |
|
| 41. |
Which of the following pairs of substance do not exhibit tautomerisms? |
|
Answer» propane-2-ol and prop-1-en-ol (A) `underset("Propane-2-ol")(CH_(3)-underset(underset(OH)(|))(CH)-CH_(3))|underset("prop-1-en-ol")(HOCH=CH-CH_(3))` These two are not TAUTOMERS (B)`underset("pent-2, 4-dione KETO form")(H_(3)C- OVERSET(overset(O)(||))(C )-CH_(2)-overset(overset(O)(||))(C )-CH_(3))|underset("pent-3-en-4-ol-2-ono Enol form")(H_(3)C - overset(overset(O)(||))(C )-HC= overset(overset(OH)(|))(C )-CH_(3))` (C ) `underset("3-methyl pentan-2-one")(H_(3)C- overset(overset(O)(||))( C)-overset(overset(CH_(3))(|))(CH)CH_(2)CH_(3))|underset("3-methyl pentan-3-ol")(H_(3)C-CH_(2)-underset(underset(CH_(3))(|))overset(overset(OH)(|))(C )-CH_(2)CH_(3))` These two are not tautomers (D) `underset("eth-1-en-ol Enol form")(H_(2)C=CHOH)| underset("Ethanal Keto form")(CH_(3)-underset(underset(O)(||))(C )-H)` |
|
| 42. |
What is meant by Biochemical Oxygen Demand (BOD)? What is its significants? |
| Answer» Solution :The amount of oxygen REQUIRED by bacteria to BREAK down the organic matter present in a certain VOLUME of a sample of water is CALLED Biochemical Oxygen Deman (BOD) The amount of BOD in the water is a measure of the amount of organic material in the water, in terms of hou MUCH oxygen will be required to break it down biologically. | |
| 43. |
Which of the following statement (s) is (are) not correct ? |
|
Answer» The shape of an ATOMIC orbital depends on the azimuthal quantum number |
|
| 44. |
XeF_(2) is isostructural with |
|
Answer» `SbCl_(2)` |
|
| 45. |
What is the major product that will be formed in the following reaction ? CH_3-undersetunderset(CH_3)(|)oversetoverset(CH_3)(|)C-CH=CH_2+HCl to |
|
Answer» `CH_3-undersetunderset(CH_3)(|)oversetoverset(CH_3)(|)C-undersetunderset(CH_3)(|)CH-CH_3` |
|
| 46. |
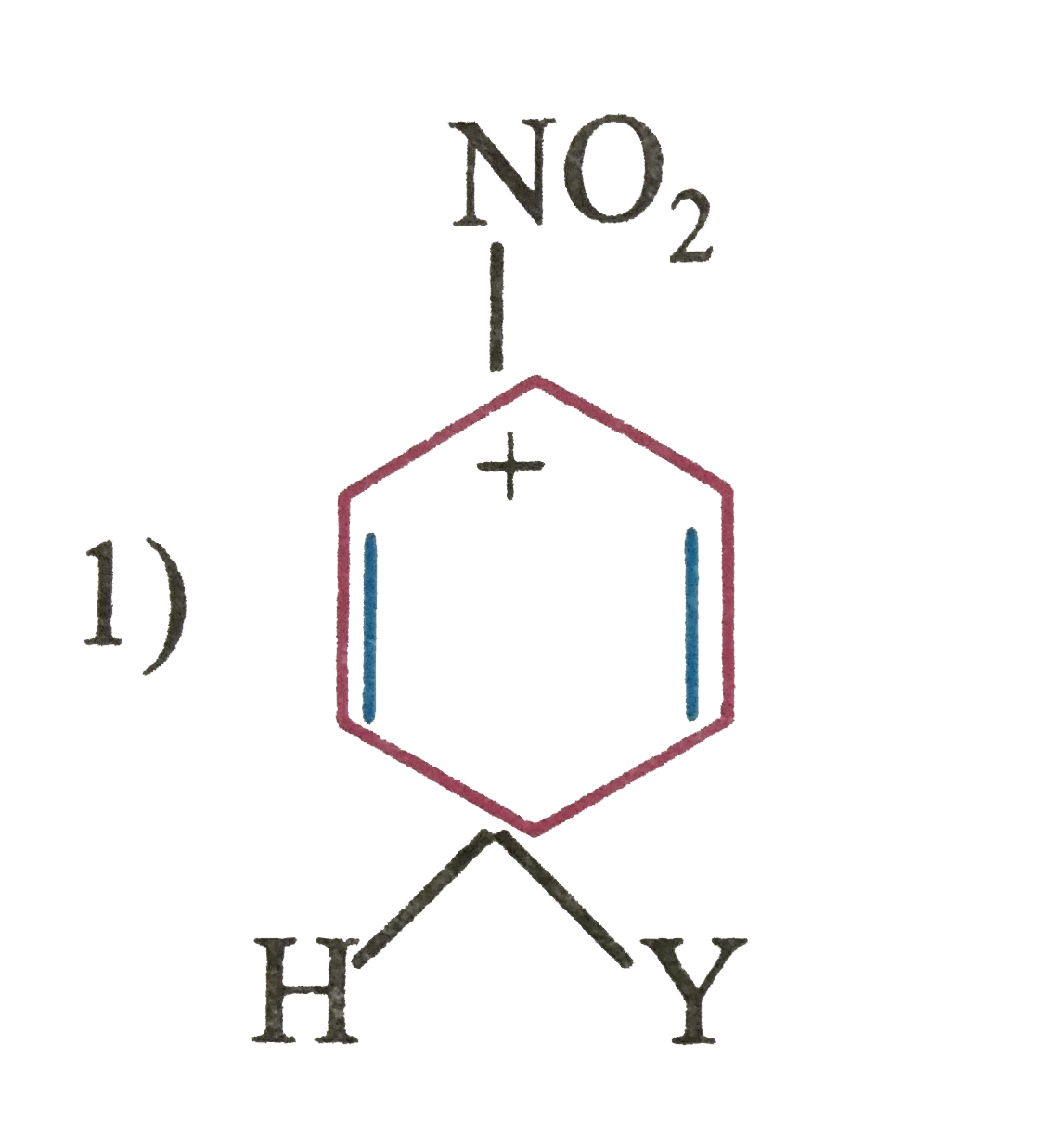
Which of the following carbocation is expected to be most stable |
|
Answer»
|
|
| 47. |
Which of the following reaction is endothermic reaction ? |
|
Answer» `CaCO_(3) to CaO+ CO_(2)` |
|
| 48. |
Thermite is a mixture of Fe_(2)O_(3) and |
|
Answer» Zn powder |
|
| 49. |
The type of H-bonding and intra orthonitro phenol and -nitro phenol are ........... Respectively. |
|
Answer» INTER MOLECULAR H-bonding and INTRA molecular H-bonding |
|