Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
What is electromeric effect? |
| Answer» Solution :It is defiend as the COMPLETE transfer of a shared pair of `pi` electrons to one of the atom joined by a MULTIPLE bond on the DEMAND of an ATTACKING reagent | |
| 2. |
Which of the following reaction(s) can be used for the preparation of alkyl halides? (I) CH_(3)CH_(2)OH+HCl overset("anhyd." ZnCl_(2))to (II) CH_(3)CH_(2) OH+HCl to (III) (CH_(3))_(3)COH+HCl to (IV) (CH_(3))_(2)CHOH+HCl overset("anhyd." ZnCl_(2))to |
|
Answer» (IV) only |
|
| 3. |
Which of the following is soluble in water? |
|
Answer» `C_2 H_5 OC_2 H_5` |
|
| 4. |
Which of the following statements is/are correct about the coefficient B in the virial equation of state PV_m = RT (1 = B/(V_m) + C/(V_m^2) + .....) |
|
Answer» It is INDEPENDENT of temperature |
|
| 5. |
In which of the following pairs, the two species are isostructural? |
|
Answer» `BrO_(3)^(-) ` and `XeO_(3)` |
|
| 6. |
Which of the following is a correct statement regarding D_2O |
|
Answer» 6000g. of ordinary WATER contains 1G. of `D_2O` |
|
| 7. |
Which of the following is a strongest oxidising agent ? |
|
Answer» `Br_2` |
|
| 8. |
Which of the following factor decreases across the period ? |
|
Answer» IONISATION energy |
|
| 9. |
Which one ofthe followingreactions does not correspond to thepreparation of"synthetic gasoline" during the Fischer-Tropsch process? |
|
Answer» `CO+3H_(2)rarrCH_(4)+H_(2)O_(2)` |
|
| 10. |
When 22.4 litres of H_(2(g))is mixed with 11.2 litres of Cl_(2(g)),each at STP, the moles of HCl_((g))formed is equal to - |
|
Answer» 1 MOLE of `HCl_((g))` `22.4 "lt."11.2 "lt."` 1 moleof `HCl=(1)/(2)` mole Limiting REAGENT is `Cl_(2)`. So, 1 mole HCl is formed. |
|
| 11. |
Which of the following species has tetrahedral geometry ? |
|
Answer» `BH_(4)^(-)` `BH_(4)^(-) rArr` 4 bond pairs + O lone pair `rArrsp^(3)` `rArr` hybridised to tetrahedral geometry `NII_(2)^(-) ` = V-shape ,`"" CO_(3)^(2-)` = TRIANGULAR PLANAR `H_(3)O^(+)` = pyramidal |
|
| 12. |
The three types of pesticides are named as _______ , _______and _________ |
|
Answer» |
|
| 13. |
The true statement about benzene is |
|
Answer» Because of unsaturation BENZENE easily undergoes addition reactions. |
|
| 14. |
Which of the following electronic configuration of an atom has lowest Ionisation enthalpy? |
|
Answer» `1s^2 2s^2 2p^5` |
|
| 15. |
Which is used to reduce pressure in distillation under reduced pressure? |
| Answer» SOLUTION :(i) WATER PUMP (II) VACUUM pump | |
| 16. |
What are activating and deactivating groups ? |
Answer» SOLUTION :
|
|
| 17. |
Which of the following statements is not true about classical smog ? |
|
Answer» Its main components are produced by the action of sunlight on emissions of automobiles and FACTORIES |
|
| 18. |
What is the difference between atomic mass and mass number? |
|
Answer» Solution :(i) MASS number is a whole number because it is the sum of number of protons and number of neutrons. (ii) Atomic mass the fractional because it is the AVERAGE RELATIVE mass of its atom as COMPARED with mass of an atom of C - 12 ISOTOPE taken as 12. |
|
| 19. |
Write the possible isomers for the formula C_4H_9Cl with structures and names. |
|
Answer» SOLUTION :`C_4H_9Cl` (4 isomers) : (i) `CH_3-CH_2 - CH_2 - CH_2Cl`:1 - Chlorobutane (ii) `CH_3 - underset(Cl_3)underset(|)(CH) - CH_2 -CH_3`2- chlorobutane (iii) `CH_3 - underset(CH_3)underset(|)(CH) - CH_2 - CH-3`1 - Chloro-2-methylpropane (iv) `CH_3 - underset(Cl)underset(|) OVERSET(CH_3)overset(|)C - CH_3`2-Chloro-2-methylpropane |
|
| 20. |
Which of the following statements are false |
|
Answer» `Ca(OH)_(2)` is called slaked lime |
|
| 21. |
Which technique is used for separation of compound present in aqueous solution ? |
| Answer» SOLUTION :DIFFERENTIAL EXTRACTION | |
| 22. |
What happens when benzene reacts with n-propylchloride in presence of anhydrous aluminium chloride? |
Answer» Solution :The major product of the reaction is isopropyl benzene (cumcnc).  The initially formed `CH_(3)CH_(2) overset(+)(CH_(2))` gets rearranged to more stable secondary CARBOCATION `CH_(3) overset(+)(CHCH_(3))`Hence the major product is isopropyl benzene and minor product is PROPYL benzene. `CH_(3)CH_(2)CH_(2)Cl+AlCl_(3) Leftrightarrow CH_(3)CH_(2) overset(+)(CH_(2))+AlC_(4)^(-) , CH_(3) CH_(2) overset(+)(CH_(2)) overset(H^(-)" shift")to CH_(3) overset(+)(CH) CH_(3)` |
|
| 23. |
Which of the following have maximum strongly of attractive inductive effects? (a) CH_(3)CH_(3)Cl, CH_(3)CH_(2)Br, CH_(3)CH_(2)F, CH_(3)F (b) CH_(3)COOH, CH_(2)ClCOOH, CHCl_(2)COOH (c ) (CH_(3))_(3) C COOH, (CH_(3))_(2) CHCOOH, CH_(3)COOH |
| Answer» SOLUTION :(a) `CH_(3)F` (b) `CHCl_(2)COOH` (c ) `CH_(3)COOH` | |
| 24. |
Which elements contain maximum and minimum ionisation enthalpy in period ? |
| Answer» Solution :Alkali metal having LESS ionisation ENTHALPY and noble GAS having high ionisation enthalpy. | |
| 25. |
Which of the following is the strongest base ? |
|
Answer» NaOH |
|
| 26. |
Which of the following has the highest point ? |
|
Answer» o-xylene |
|
| 27. |
Which of the following ions will cause hardness in water sample ? |
|
Answer» `CA^(2+)` |
|
| 28. |
Which statement is false? |
|
Answer» Elements of VB group are transition elements. |
|
| 29. |
Which of the following is incorrect |
|
Answer» `H_(2)O_(2)` is a weak acid `H_(2)O_(2) IFF H^(+)+HO_(2)^(-)` (hydroperoxide ion) `H_(2)O_(2) iff 2H^(+)+O_(2)^(2-)` (Peroxideion) `H_(2)O_(2)` behaves as oxidising as well as reducing agent in ACIDIC and BASIC medium. In acidic medime, `H_(2)O_(2)+2H^(+)2e^(-) RARR 2H_(2)O` in basic medime, `H_(2)O_(2)+OH^(-1)+2e rarr 3OH^(-)` As reducing agent, in acidic medium, `H_(2)O_(2) rarr 2H^(+)+O_(2)+2e^(-)` In alkaline medium, `H_(2)O_(2)+2OH^(-) rarr 2H_(2)O+O_(2)+2e^(-)` |
|
| 30. |
Which one of the following element mostly present in the sun and the stars? |
|
Answer» Hydrogen |
|
| 31. |
Which of the following exhibits chain isomerism? |
|
Answer» `CH_3CH_2NH_2` `CH -=C-CH_2-CH_2-CH_3 and CH-=C-UNDERSET(CH_3)underset(|)CH-CH_3` |
|
| 32. |
Whenever a hydrogen atom emits a quantum in the visible region of Balmer series : |
|
Answer» it may emit another quantum in Balmer series again `1/(lambda) = R [(1)/(n_1^2) - 1/(n_2^2) ] = 109677 [1/(1^2) - 1/(2^2)]` `lambda = 122 xx 10^(-7) cm = 122 nm` After coming to `2^(nd)` orbit then electron can go `1^(st)` orbit. So lyman series is possible . |
|
| 34. |
The standard enthalpy of formation of Fe_(2)O_(3)(s) is - 824.2 kJ mol^(-1) . Calculate the enthalpychange for the reaction :4 Fe (s) + 3O_(2) (g) rarr 2Fe_(2) O_(3) (s) |
|
Answer» `= [2(-824.2) ] -[0+0]` `= - 1648.4kJ` |
|
| 35. |
Which of the following options will be correct for the stage of half completion of the reaction A hArr B. |
|
Answer» `DELTAG^(Θ)=0` At the stage of half completion of the reaction A `hArr` B, [A]=[B]. HENCE, K = 1 . Therefore, `Delta G^(@)=0`. |
|
| 36. |
Define Graham's law of diffusion. |
| Answer» SOLUTION :Graham's DIFFUSION Law : The rate of diffusion or EFFUSION is INVERSELY PROPORTIONAL to the square root of molar mass. The statement is called Graham's law of diffusion/effusion. | |
| 37. |
Which pollutant can be used as fertiliser |
|
Answer» VEGETABLE WASTE |
|
| 38. |
When cyclohexanone and + X The no . Of hybrid orbitals of phosphorus involved formation X. |
|
Answer» |
|
| 39. |
Which type of bond is not present in HNO_(2) molecule ? . |
|
Answer» COVALENT 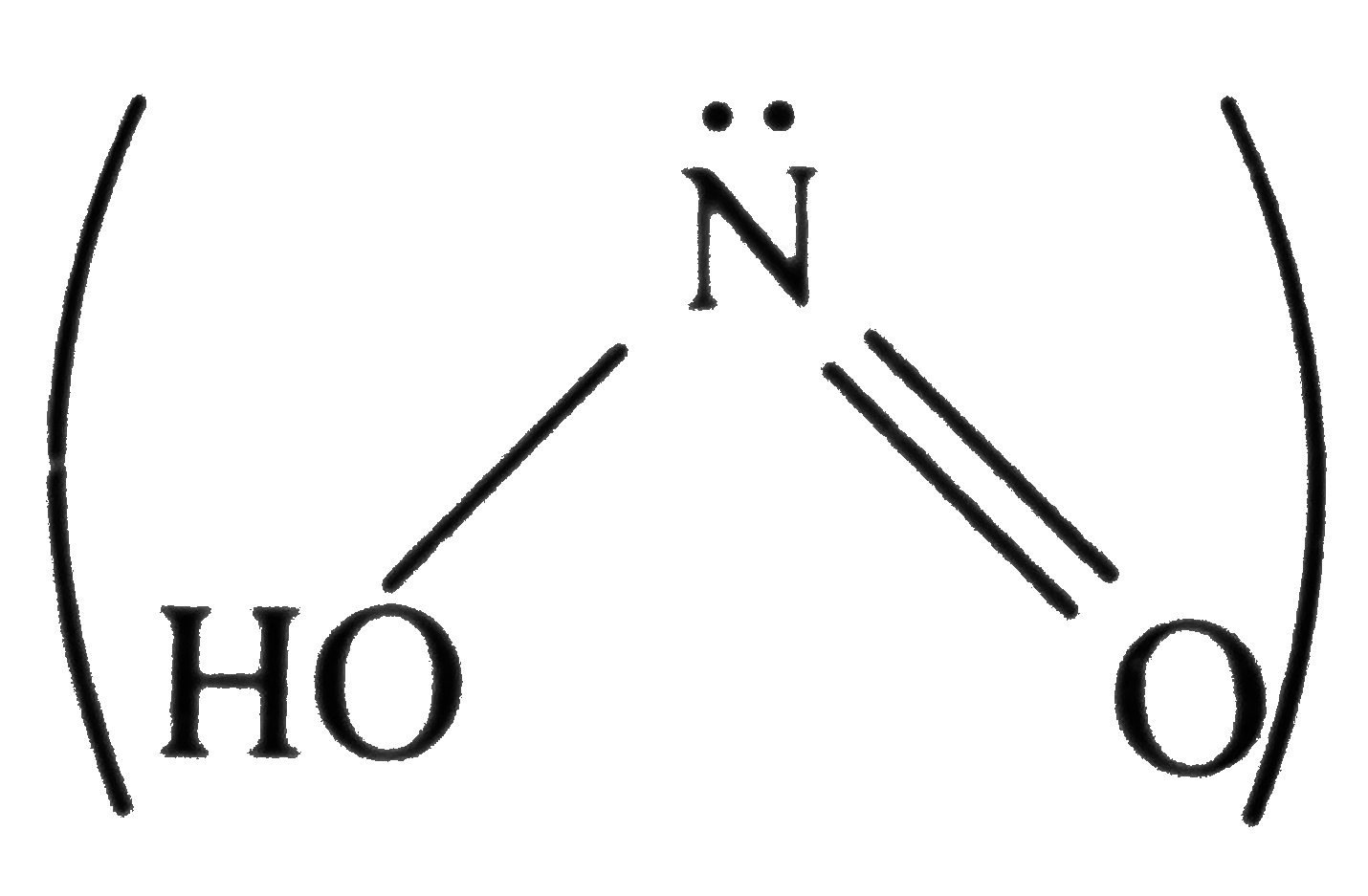
|
|
| 40. |
Which of the following is not an emulsion |
|
Answer» Butter |
|
| 41. |
The standard reduction potential for silver electrode is +0.80 V. It is connected to a standard hydrogen electrode to make a galvanic cell. State whether silver electrode will act as anode or cathode. |
| Answer» SOLUTION :CATHODE | |
| 42. |
The wavelength of H_(alpha) line of Balmer series is 6500 Å. What is the wavelength of H_(beta) line of Balmer series? |
|
Answer» Solution :`H_(ALPHA)` line of Balmer series is obtained when `n_(1) = 2, n_(2) = 3` `H_(beta)` line of Balmer series is obtained when `n_(1) = 2, n_(2) = 4` Thus, `bar(v) H_(alpha) = (1)/(.^(lamda)H_(alpha)) = R_(H) ((1)/(2^(2)) - (1)/(3^(2))) = R_(H) ((1)/(4) - (1)/(9)) = R_(H) xx (5)/(36)` ...(i) `bar(v) H_(beta) = (1)/(.^(lamda)H_(beta)) = R_(H) ((1)/(2^(2)) - (1)/(4^(2))) = R_(H) ((1)/(4) - (1)/(16)) = R_(H) xx (3)/(16)`..(ii) Dividing eqn. (i) by eqn. (iii), we get `(.^(lamda)H_(beta))/(.^(lamda)H_(alpha)) = (5)/(36) xx (16)/(3) = (20)/(27) :. .^(lamda)H_(beta) = (20)/(27) xx .^(lamda)H_(beta) = (20)/(27) xx 6500 Å = 4814.8 Å` |
|
| 43. |
(vii) S_(2)O_(3)^(2-)+I_(2)to S_(2)O_(6)^(2-)+I^(-). |
Answer» SOLUTION : . . Eqalise the increase/ DECREASE in O.N by multiplying the S species by 1 and I species by 3. `S_(2)O_(3)^(-)+3I_(2)to S_(2)O_(6)^(2-)+3I^(-)`. Balance all other atoms EXCEPT O and H `S_(2)O_(3)^(2-)+3I_(2)to S_(2)O_(6)^(2-)+6I^(-)` Balance O ATOM by adding `H_2O` on the side falling short of Oxygen. `S_(2)O_(3)^(2-)+3I_(2)+3H_(2)Oto S_(2)O_(6)^(2-)+6I^(-)` Balance H atom by adding `H+` ion on the side falling short of hydrogen. `S_(2)O_(3)^(2-)+3I_(2)+3H_(2)Oto S_(2)O_(6)^(2-)+6I^(-)+6H^(+)`. Add EQUAL number of `OH^(-)` ion on the both side since hte medium is alkaline. `S_(2)O_(3)^(2-)+3I_(2)+3H_(2)O+6OH^(-)to S_(2)O_(6)^(2-)+6I^(-)+6H^(+)+60H^(-)`. `S_(2)O_(3)^(2-)+3I_(2)+3H_(2)O+6OH^(-)to S_(2)O_(6)^(2-)+6I^(-)+6H_(2)O`. |
|
| 44. |
Write the coordination numbers of cations and anions in the following ionic compounds : (a)Zinc blende (b)Fluorite |
|
Answer» SOLUTION :ZINC blende=ZnS, `Zn^(2+)=4 , S^(2-)`=4 FLUORITE=`CaF_2, Ce^(2+)`=8 , `F^-` = 4 |
|
| 45. |
Weakest acid among the following is |
|
Answer» Acetic acid `HC -= CH LT H_(2)O lt` Phenol `lt CH_(3)COOH` |
|
| 46. |
What happens to the ionic product of water if some acid is added into water ? |
| Answer» SOLUTION :If REMAINS UNCHANGED. | |
| 47. |
What happens when quick lime reacts with (i) H_(2)O and (ii) CO_(2) |
|
Answer» Solution :`CaO + H_(2)O to Ca(OH)_(2)`(calcium hydroxide) (ii) `CaO + CO_(2) to CaCO_(3)`(calcium CARBONATE) |
|
| 48. |
Which of the group has highest +I effect ? |
|
Answer» `CH_(3)^(-)` |
|
| 49. |
The rule that explain the reason for chromium to have [Ar]3d^(5)4s^(1) configuration instead of [Ar]3d^(4),4s^(2) is |
|
Answer» Pauli's exclusion PRINCIPLE |
|
| 50. |
Which biological pump is important for biochemical reaction in human body? |
|
Answer» Ca-Mg |
|