Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Write the structures of different isomeric alkyl groups corresponding to the molecular formula C_(6)H_(11). Write IUPAC names of alcohols obtained by attachment of -OH groups at different carbons of the chains. |
Answer» SOLUTION : 
|
|
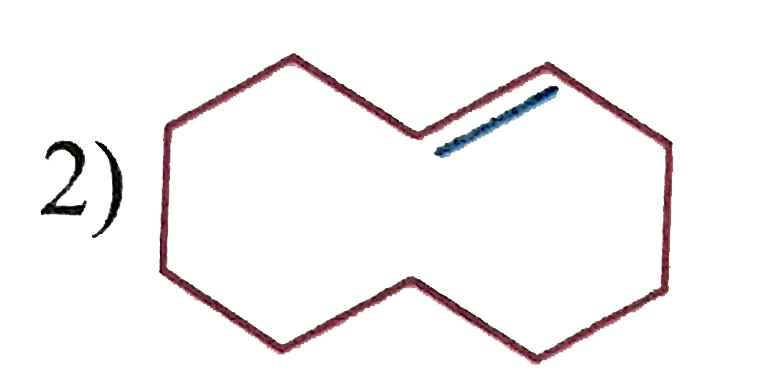
| 2. |
Which of the following show geometrical isomerism? |
|
Answer»
|
|
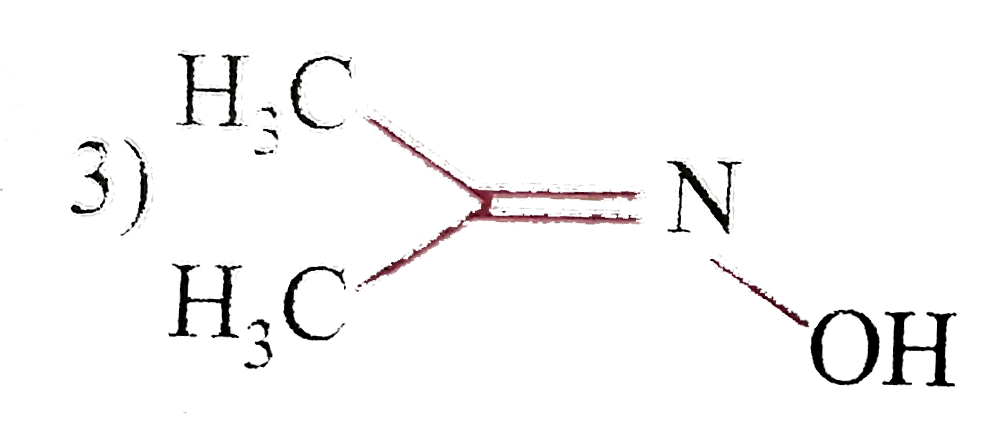
| 3. |
Which among the following molecules is not flat? |
|
Answer» `C_(6)H_(6)` |
|
| 4. |
When 10 mL of 10 M solution of H_(2)SO_(4) and 100 mL of 1 M solution of NaOH are mixed, the resulting solution will be : |
|
Answer» acidic |
|
| 5. |
Which of the following carbonate is more thermally stable |
|
Answer» `BeCO_(3)` 
|
|
| 7. |
What is the degree of hardness of a sample of water containing 24mg of MgSO_(4) (molecular mass 120) per kg of water? |
|
Answer» 10ppm 106 water will CONTAIN =24000mg of `MgSO_(4)=24` of `MgSO_(4)` So, 24g of `MgSO_(4)=(100)/(120)xx24=20g` of `CaCO_(3)` Hardness of water `=20ppm` |
|
| 8. |
What is heavy water ? Give uses of it. |
|
Answer» Solution :Heavy water is injurious to HUMAN beings, plants and animals since it SLOWS down the rates of reactions occurring in them. Thus heavy water does not support life so it is not used for drinking Heavy water `(D_2O)` is a oxide of deuterium, the isotope of Hydrogen. Heavy water is extensively used as a moderator in nuclear reactors and in EXCHANGE reactions for the study of reaction mechanisms. It can be prepared by exhaustive electrolysis of water or as a by product in some fertilizer INDUSTRIES. `CaC_2+2D_2O to C_2D_2+Ca(OD)_2` `SO_3 + D_2O to D_2SO_4` `Al_4C_3 + 12D_2O to 3CD_4+4Al(OD)_3` |
|
| 9. |
Which of the following order of energies of molecular orbitals of N_(2) is correct ? |
|
Answer» `(pi 2p_(y)) LT (sigma 2p_(Z)) lt (pi**2p_(x)) = (pi^(**) 2p_(y))` The correct increasing order of energies of MOLECULAR orbitals of `N_(2)` is given below : `sigma 1s lt sigma^(**) 1s lt sigma 2s lt sigma^(**) 2s lt (pi 2p_(x) = pi 2p_(y)) lt sigma 2p_(z) lt (pi^(**) 2p_(x) = pi^(**) 2P(y) lt (sigma^(**) 2p_(z))` |
|
| 10. |
Which of the following pair of elements shows diagonal relationship? |
|
Answer» LI and Be |
|
| 11. |
When methane is made to react with a halogen (X_(2)), halides are formed, the order of reactivity is : |
|
Answer» `F_(2) gt Cl_(2) gt Br_(2) gt I_(2)`<BR>`Cl_(2) gt F_(2) gt Br_(2) gt I_(2)` |
|
| 12. |
Write the electronic configurations of the elements with the following atomic numbers : 3, 8, 14, 17, 21, 38, 57 Also mention the groups of the periodic table to which they belong. |
Answer» SOLUTION : Note that here one ELECTRON ENTERS 5d orbital before filling of 4f STARTS. |
|
| 13. |
What is the function of adding NH_(4)OH in group V ? |
| Answer» SOLUTION :It converts any `NH_(4)HCO_(3) ` present into `(NH_(4))_(2)CO_(3)`. | |
| 14. |
Write the structural formula of the following: (a) 2, 3-dimethyl hexanal (b) 2-methyl-1-butene (c ) 2-aminopropanoic acid (d) 3-bromo-4-methyl heptane |
|
Answer» |
|
| 15. |
Which of the following pairs of compounds are positional isomers ? |
|
Answer» `CH_(3)-CH_(2)-CH_(2)-CHO and CH_(3)-CH_(2)-CH_(2)-OVERSET(O)overset(||)C-CH_(3)` (B) Functional isomers (C) Positional isomers (D) Not isomers |
|
| 16. |
The specific heat of a metal is 0.16. It appoximate atomic mass would be : |
|
Answer» 32 |
|
| 17. |
Which of the following statement is / are true for an electronchemical cell ? |
|
Answer» oxidation occurs at the anode only |
|
| 18. |
What is the nature of aqueous solution of NaH |
|
Answer» Acidic |
|
| 19. |
The values of critical presssure, critical volume and critical temperature for hydrogen gas are 12.97 bar, 0.065 L "mol"^(-1) and 33.2 K. Calculate the compressibility factor. |
|
Answer» SOLUTION :Compressibillity factor (z) for one MOLE of gas is GIVEN interms of critical presssure `(P_(C))`. Critical VOLUME `(V_(C))` and critical temperature `(T_(C))` as `z = (P_(c)V_(c))/(RT_(c)) = (12.97 xx 0.065)/(0.083 xx 33.2)` Compressibility factor ` = 0.306` |
|
| 20. |
What is oxidation state of K in KO_(2) ? |
| Answer» Solution :The superoxide SPECIES is represented as `O_(2)^(-)` . It has one UNIT negative CHARGE . Since the compound is neural , therefore , the oxidation state of K is `+1`. | |
| 21. |
Which of the following attain the linear structure : |
|
Answer» `BeCl_(2)` `BeCl_(2) ` (Cl - Be - Cl) and `CS_(2)` (S = C= S) both are linear. `NCO^(+)` is NON- linear . However,` ("remember that" ""^(-)NCO (""^(-)N = C = O))` is linear because it is isoelectronic with `CO_(2)`. `NO_(2)` is ANGULAR with bond angle `132^(@)` and each O - N bond length of 1.20 Å (intermediate between single and DOUBLE bond) . |
|
| 22. |
Which of the following elements shows same catenation property ? |
|
Answer» C,Si |
|
| 23. |
What should be the correct IUPAC name of diethyl chloromethane? |
|
Answer» 3-Chloro PENTANE |
|
| 24. |
Which of the following compounds possesses H-bond ? |
|
Answer» Borazine 
|
|
| 26. |
Which of the following statements is/are correct about the followig reaction? Fe_(3)O_(4)overset(Delta)toFe_(2)O_(3). |
|
Answer» The EQUIVALENT weight of `Fe_(3)O_(4)` is `M_(1)(M_(1)=` molecular weight of `Fe_(2)O_(4))` `_(2)(Fe^((8)/(3)+))_(3)to3(Fe^(3+))_(2)+2E^(-)` `{:[(x=(8)/(3)xx2xx3),(x=16),("n-factor"=(2)/(2)=1),(Ew_(1)=(M_(1))/(1))]:}` `{:[(x=18),(3mol=2e^(-)),(1MOL=(2)/(3)e^(-)),("n-factor"=(3)/(2)),(Ew_(2)=(M_(2))/((2)/(3))=(3M_(2))/(2)):}]` |
|
| 27. |
What is the equation form of Langmuir isotherm under high pressure ? |
|
Answer» `(X)/(m)=(a)/(b)` `(x)/(m)=(ap)/((1+BP))` At high pressure, `bp gtgt1` Hence, `(x)/(m)=(ap)/(bp)=(a)/(b)` |
|
| 28. |
Which type of graph is obtained when we plotted pV to p of ideal gas ? |
| Answer» SOLUTION :PARALLEL to X - AXIS and STRAIGHT line. | |
| 29. |
X +AgNO_(3)rarr Y (white ppt.) Y is soluble in nitric acid an ammonia. The ppt. becomes yellowish brown upon addition of excess reagent owing to the formation of Z. If aqueous suspension of Y is boiled then it gives same product Z. Here Z is: |
|
Answer» `Ag_(2)CO_(3)` |
|
| 30. |
X +AgNO_(3)rarr Y (white ppt.) Y is soluble in nitric acid an ammonia. The ppt. becomes yellowish brown upon addition of excess reagent owing to the formation of Z. If aqueous suspension of Y is boiled then it gives same product Z. Her, X and Y are: |
|
Answer» `CO_(3)^(2-)` and `Ag_(2)O` |
|
| 31. |
What is reducing agent ? |
| Answer» Solution :During reaction, which substance DONATE hydrogen or accept OXYGEN such substance is KNOWN as redusing agent. | |
| 32. |
What are factors which influence the electron gain enthalpy ? |
|
Answer» Solution :Factors influencing ELECTRON gain enthalpy : (i)SIZE of the atom : Electron affinity is inverselyproportional to the size of the atom .As the size of atom increases , the effective nuclear charge decreases or the nuclear attraction for ADDING electron decreases. (ii)Nuclear charge : Electron affinity is directly proportional to effective nuclear charge Greater the nuclear charge more will be the TENDENCY toaccept electrons so Electron affinity is more . (iii)Electronic configuration : An atom with stable electronic configuration has no tendency to gain an electron . Such atoms have zero or almost zero electron gain enthalpy. (iv)Shielding effect :Electron affinity is inversely proportional to shielding effect . Electronic energy STATE , lying between nucleus and outermost state hinder the nuclear attraction for incoming electron . Therefore , greater the number of inner lying state , less will be the electron affinity. |
|
| 33. |
Which statement is false for elimination reaction . |
|
Answer» `beta`-elimination is more common than `alpha` & `gamma` elimination |
|
| 34. |
X overset("Ni)underset(H_2)rarrY,Yoverset(450^@C)rarrQ Here X,Y and Q are gaseous hydrocarbons, then X, Y and Q respectively are |
|
Answer» `{:(X,Y,Q),(C_2H_4,C_2H_6,C_2H_4):}` |
|
| 35. |
Which of the following is unstable and can be kept only in the atmosphere of CO_(2) ? |
|
Answer» `MgCO_(3)` |
|
| 36. |
Which of the following pairsof elements wouldhavemorenegativeelectron gainenthalpy ? Explain (i) Nor O(ii)s or O(iii) C or Si |
|
Answer» SOLUTION :(i) Theelectron gainenthalpy ofO ishighlynegativewhilethatof N isslightly positive. Reason . The electronicconfigurationof N isquitestable`(1s^(2) 2s^(2) 2p^(1)_(x) 2p_(x)^(1) 2p_(z)^(1))` sinceit hasexactly half-filled2p-orbitaland hencehas notendencyto acceptan EXTRA electron. Inotherwordsenergyhas tobe suppliedto addan extra electron .Thuselectron- gainenthalpy of N isslightly positive . Incontrast the electronic CONFIGURATION of O `(1s^(2) 2s^(2) 2p_(x)^(2) 2p_(x)^(1) 2p_(z)^(1))` is not sostablebut ithas highernuclearchargeand loweratomicsizethan Nandhenceit hasa hightendency to acceptan extra electron . Inotherwordselectron gainenthalpyof Ois highly negative . (ii) S hasmorenegativeelectron gainenthalpy than O Reason . The sizeof O ismuchsmallerthanthatof S. Asa resultthe electron- electron replsionsin thesmaller 2p- subshell of O arecomparatively largerthan thosepresent in thebigger 3p- subshellof s. Therefore, Shashighertendency to acceptan additionalelectron than O (iii) C hasmorenegativeelectron gainenthalpy than Si Reason. this is because C- atom hassmallersizeSi- atom (NOTE that theelectron - electron repulsions in these ATOMS are notverylargebecausetheycontainonly 4 electrons in theoutermostshell) |
|
| 37. |
When charcoal burns in air signs of DeltaH and DeltaS "are " 2C_((s)) + O_(2(g)) rarr 2CO_((g)) |
|
Answer» `{:(Delta H,Delta S),(+,-):}` `Delta S gt 0 :. Delta n_(G) gt 0` |
|
| 38. |
What isthe differencein groundstateand exicited stateof hydrogen ? |
|
Answer» SOLUTION :In groundstateof hydrogenelectronpresentin 1S ORBITAL . ITIS the moststablestatewithleastenergy in exicited stateelectronsare in any orbitalsexcept1sorbitals2s,3p,3s,3p thesestateare UNSTABLE. |
|
| 39. |
What happens when limited amount of CO2 gas is passed into milk of lime? Give equation. |
| Answer» Solution :Lime water turns milky or WHITE `CA(OH)_(2)toCaCO_(3)+H_(2)O` | |
| 40. |
What is dead burnt plaster ? |
| Answer» Solution :When gypsum is heated above 393 K, no water of CRYSTALLISATION is left and anhydrous calcium SULPHATE, `CaSO_(4)` is FORMED.is formed. This is KNOWN as .dead burnt plaster. | |
| 41. |
Which main product is obtained when 3-methyl-pent-2-ene react with HBr in presence of peroxides ? |
|
Answer» 3-bromo-3-methylpetane `underset("2-Bromo-3-methylpentane(Main)")(CH_(3)-underset(Br)underset(|)(CH)-underset(CH_(3))underset(|)(CH)-CH_(2))-underset("3-Bromo-3-methylpentane(MINOR)")(CH_(3)+CH_(3)CH_(2)-underset(CH_(3))underset(|)overset(Br)overset(|)(C)-CH_(2)-CH_(3))` |
|
| 42. |
Which of the following are not state functions ? (i) q+w (ii) q (ii) w (iv) H- TS |
|
Answer» (i) and (iv) |
|
| 43. |
What is the geometrical shape of [Pt(Cl)_(4)]^(2-)? |
|
Answer» PLANAR triangular |
|
| 44. |
Which of the following has larger dipole moment ? Explain . 1-Butyne or 1-Butene |
Answer» Solution :Their strcutures are : `H_(3) underset("1-Butyne")(C-CH_(2) - C-=C-H)`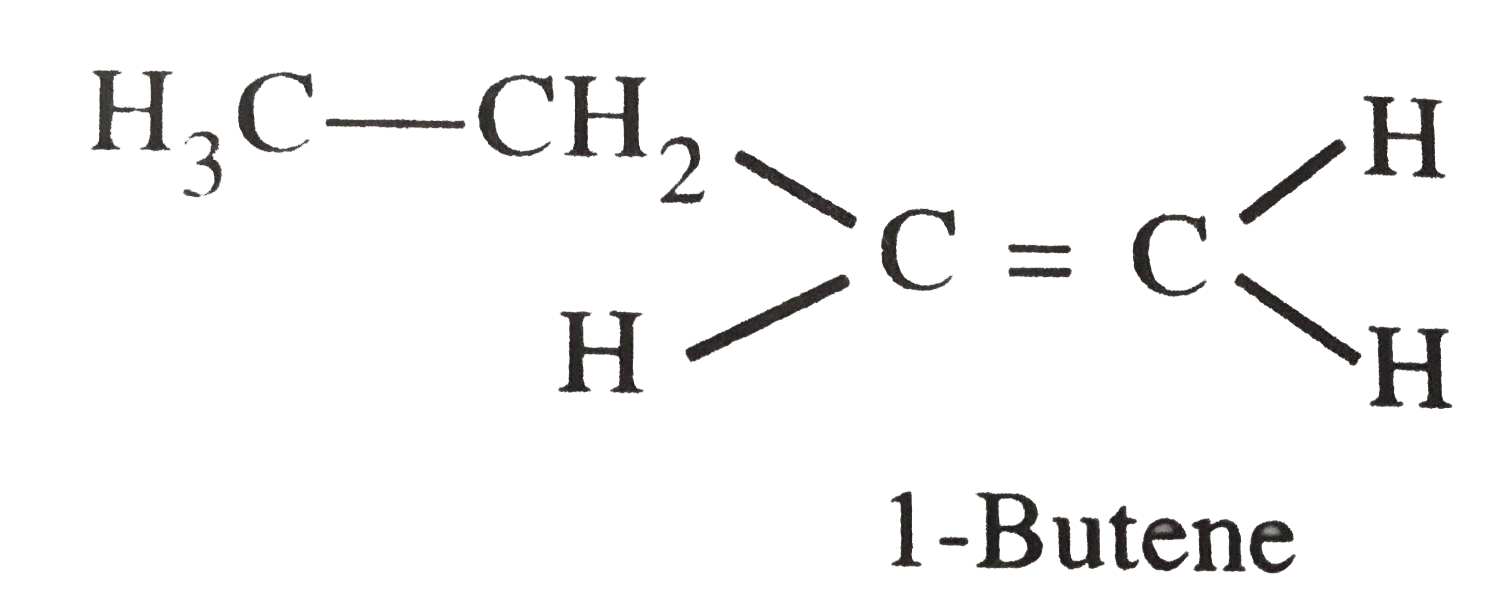 In 1-Butyne , each triply bondedC-atom is SP -hybridized whereas in 1-Butene, each doubly bonded C-atom is `sp^(2)` -hybridized . Hence 1-Butyne has LARGER dipole MOMENT because the electronegativity of sp-C is more than that of `sp^(2) - C` |
|
| 45. |
Which is used as Friedel -Craft catalyst? |
|
Answer» `AlCl_(3)` |
|
| 46. |
The value of resonance energy of nitromethane is represent by which equation? |
| Answer» SOLUTION :`(("RESONANCE "),("energy of"),("nitromethane"))= (("STABILIZATION energy of maximum"),("stable resonance"),("STRUCTURE of nitromethene"))- (("Stabilization energy of hybrid"),("structure of nitromethane"))` | |
| 48. |
Which of the following metal ions will have maximum number of unpaired electrons |
|
Answer» `Fe^(+2)` 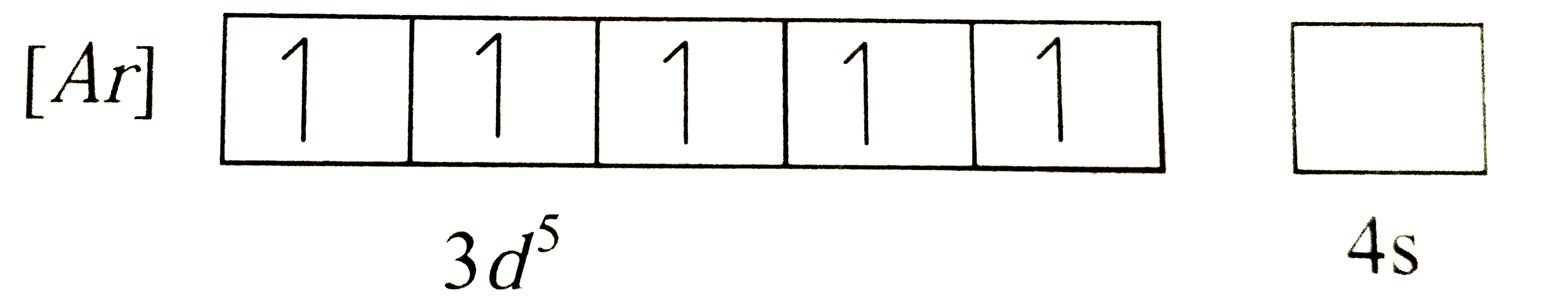
|
|
| 49. |
Which of the followingelementshas themostnegativeelectrongainenthalpy ? Givereasons (i) [Ne] 3s^(2) 3p^(2)"" (ii) [Ne] 3s^(2) 3p^(4)"" (iii) [Ne] 3s^(2)3p^(5) |
| Answer» Solution :The element(chlorine) correspodingto theelectronic cofiguration(III) will have thethe mostnegativeelectrongainenthalpy. Thisis due tothe REASONTHAT itcontainsoneelectrons lessthat thestablenoblegasconfigurationi.e.,[Ne] `3p^(2) 3p^(6)` and hencehas astrongtendency to acceptone electron to acquire thestablenoblegasconfiguration | |
| 50. |
The standard enthalpy of combustion at 25^(@)C of hydrogen,cyclohexene ( C_(6)H_(10)) and cyclohexane (C_(6)H_(12)) are - 241, -3800 and -3920 kJ // mol respectively. Calculate the heat of hydrogenation of cyclohexene. |
|
Answer» |
|