Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
What mass of Calcium carbonate is to be decomposed to obtain 4.4 of CO_(2) in the following reaction CaCO_(3)toCaO+CO_(2) (Molecular mass of CaCO_(3)=100) |
|
Answer» |
|
| 2. |
What 'X' in the following reaction ? C_(6)H_(5) -C-=C-H overset(Hg^(+2))underset("dil" .H_(2)SO_(4))rarr X |
|
Answer» `C_(6)H_(5) - COCH_(3)` |
|
| 3. |
Which of the following alkane can be synthesized by the wurtz reaction in good yield? |
|
Answer» `(CH_3)_2CHCH_2 CH(CH_3)_2` |
|
| 4. |
Water molecule has bent structure whereas carbon dioxide molecule is linear |
|
Answer» Solution :In `H_(2)O`, OXYGEN atom is `sp^(3)` hybridised with two lone PAIRS. The four `sp^(3) ` hybridised ORBITALS possess a tetrahedral geometry with two comers occupied by hydrogen atoms and other two by the lone pairs. The bond angle is REDUCED to `104,5^(@)` from `109.5^(@)`because of greater repulsive forces between lp - Ip and the molecule so gain V-shape or bent structure (angular structure).  In `CO_(2)` molecule, carbon atom is sp-hybridised. The two sp hybrid orbitals are ORIENTED in opposite direction forming an angle of `180^(@)`.  So `H_(2)`Omolecule has bent structure and `CO_(2)` molecule is linear. |
|
| 5. |
Which plant extract act as insecticides ? |
|
Answer» Neem |
|
| 6. |
What is order of first second and third ionisation enthalpy? Why? |
|
Answer» Solution :First ionisation ENTHALPY of element is less than second ionisation enthalpy of element. Second ionisation enthalpy of element is less than third ionisation enthalpy of element. Order : `3^(rd) gt 2^(nd) gt 1^(st)` ( ionisation enthalpy) Because, positive charge INCREASES attraction increases, so more ENERGY is required to lose electron. |
|
| 7. |
Which one of the following forms micelles in aqueous solution, above certain concentration. |
|
Answer» GLUCOSE |
|
| 8. |
Write the conjugate acids for the following Bronsted bases : NH_2^(-) , NH_3 and HCO_3^(-) . |
| Answer» SOLUTION :`NH_3 , NH_4^(+)` and `H_2CO_3` | |
| 9. |
Which of the following is a secondary haloalkane'? |
|
Answer» BROMOETHANE |
|
| 10. |
Which of the following orders orders is true regarding the acidic nature of phenol ? |
|
Answer» PHENOL `GT` o-cresol `LT` nitrophenol |
|
| 11. |
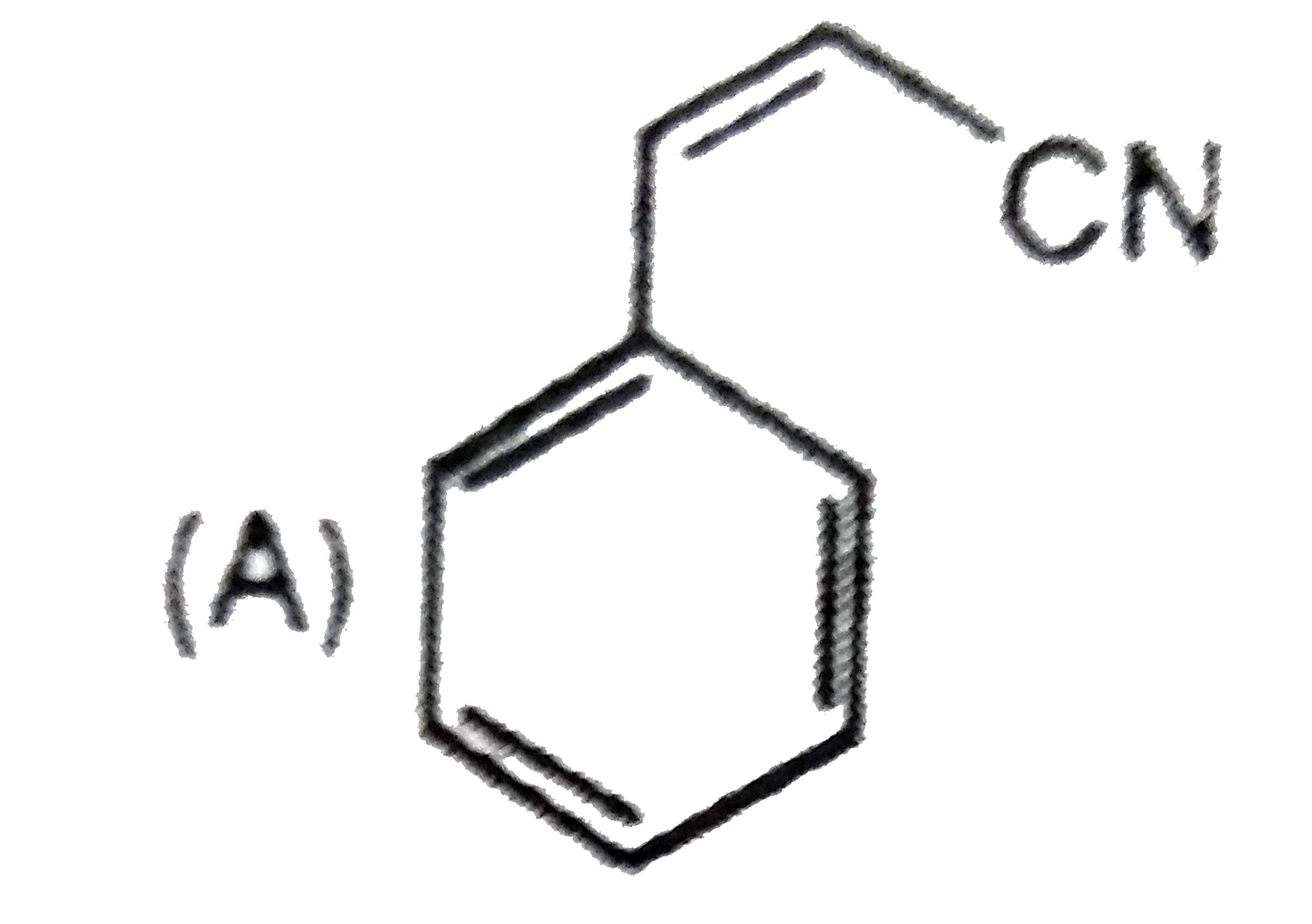
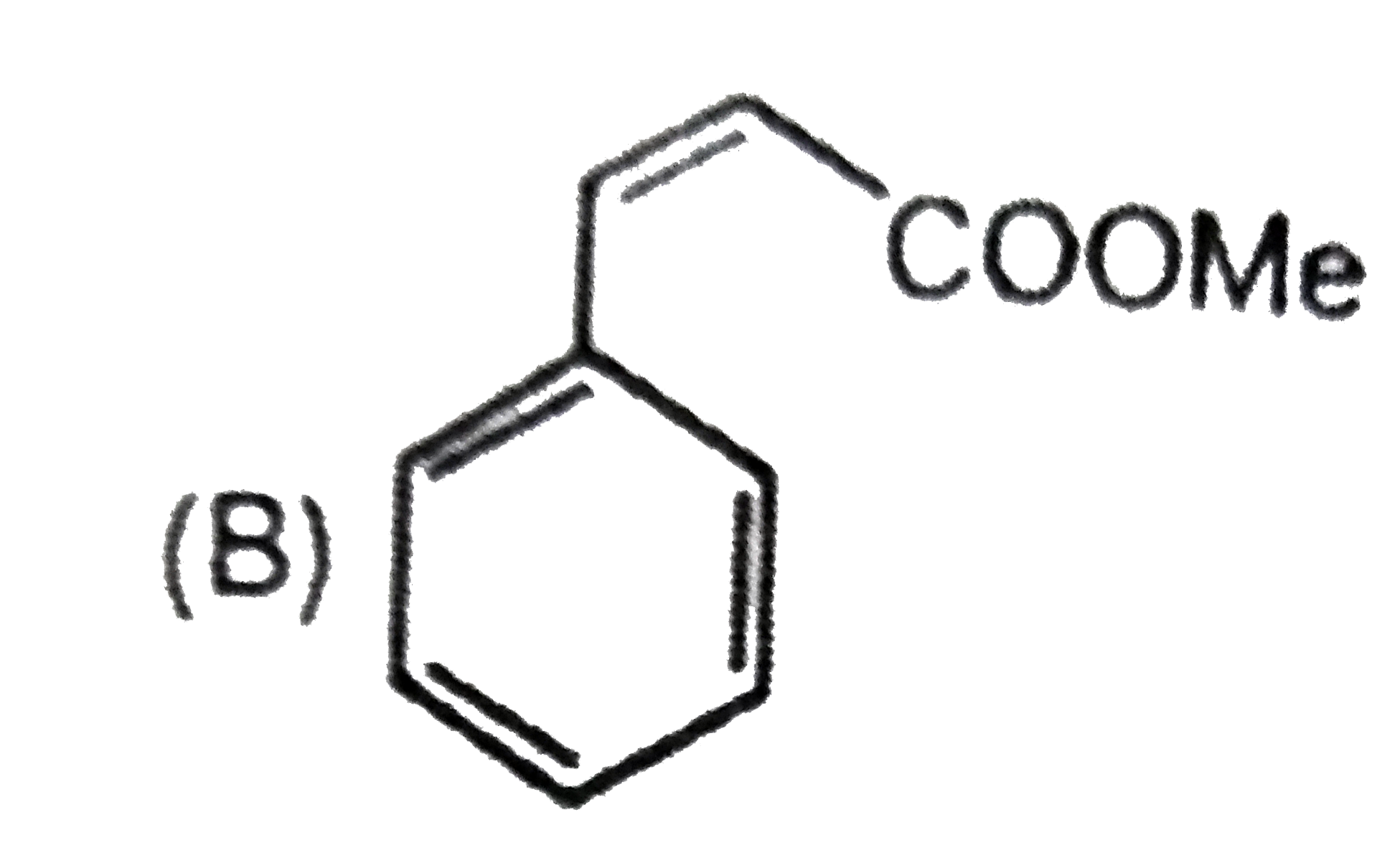
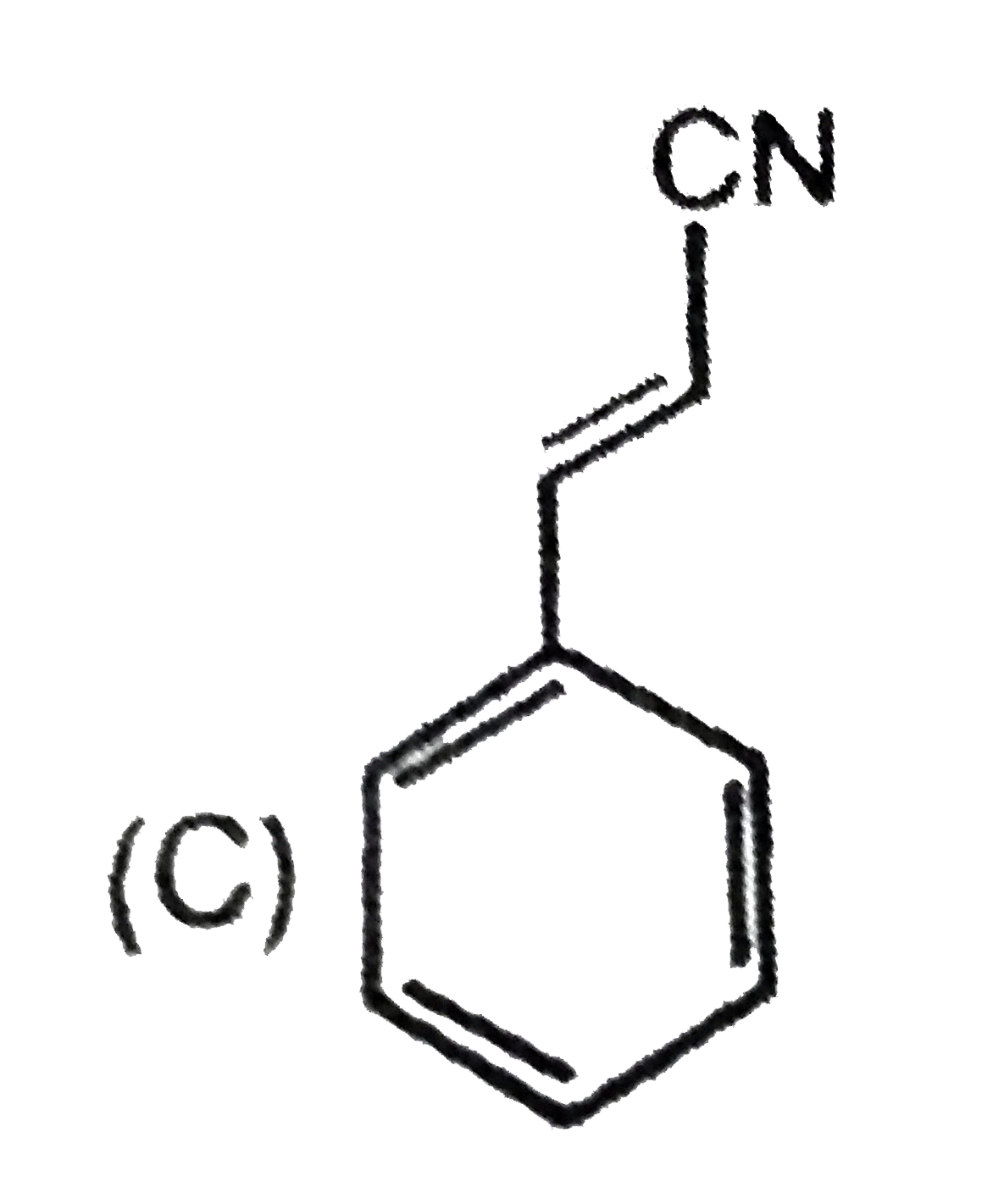
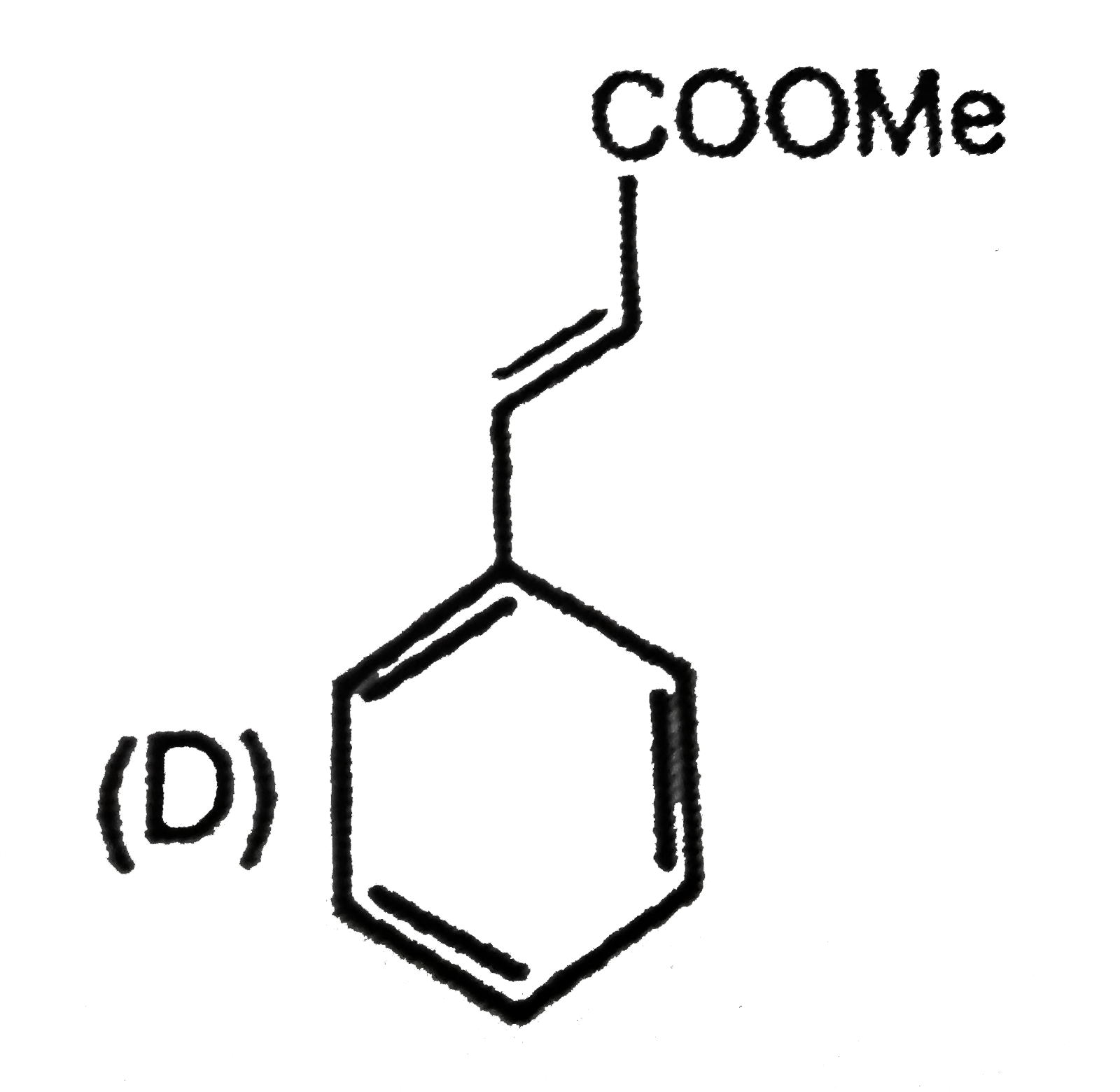
Which compound show tautomerism: |
|
Answer»

|
|
| 13. |
Which of the following is not an ore of Mg? |
|
Answer» Gypsum |
|
| 14. |
Which one of the two, CIO_(2)6(-) or CIO_(4)^(-) shows disproportionation reaction and why ? |
|
Answer» Solution :The oxidation state of CI in `CIO_(2)^(-)` is `+3`. So, CHLORINE can get oxidised as well as reduced and can ACT as reductant and oxidant. The disproportionation reaction of `CIO_(2)^(-)` is `overset(+1)(3CI)O_(2)^(-)to CI^(-)+overset(+5)CIO_(3)^(-)` In `CIO_(4)^(-),CI` is in its highest oxidation state, So it can only be an oxidant. |
|
| 15. |
Which of the following compounds is odd from the remaining |
|
Answer» `NaNO_(3)` |
|
| 16. |
What is isonisation enthalpy? |
|
Answer» Solution :Energy REQUIRED to lose 1 mole ELECTRON is KNOWN as ionisation enthalpy. "Energy requird to lose one electron from .mole neutral atom is known as Ionisation enthalpy". `X_((G)) + Delta_(i)H = X_((g))^(+) + e^(-)` Where , `Delta_(i) H ` = FIRST Ionisation Enthalpy. |
|
| 17. |
Which one of the following has zero dipole moment? |
| Answer» Solution :NO | |
| 18. |
Wurtz reaction involves the comdensation of two molecules of alkyl halides in the presence of sodium and dry ether R-X+2Na+R-Xoverset("dry ether")toR-R+2NaX In this reaction small amount of alkene is also formed as by product. CH_(3)CH_(2)Br+CH_(3)CH_(2)Broverset("Na/dry ether")toCH_(3)-CH_(2)-CH_(2)-CH_(3)+underset("By-product")ubrace(CH_(2)=CH_(2)+CH_(3)-CH_(3)) Tertiary alkyl halides do not give wurtz reaction. Frankland reaction is similar but has certain advantages over wurtz reaction. it is useful in the synthesis of symmetrical alkanes. frankland reaction is shown by primary, Secondary as well as tertiary alkyl halide. Q. Among all isomers of hexane which of the following isomer cannot be obtained in a good yield by wurtz reaction (from symmetrical alkyl halides)? 1. n-hexane 2. iso hexane 3. 2,3-dimethyl butane 4. neo hexane |
|
Answer» 1,4 |
|
| 19. |
Which of the following reactions is used to bring about the following conversion ? |
|
Answer» Hofmann reaction |
|
| 20. |
Which of the following quantum numbers are not possible ? |
|
Answer» n = 2,L = 1, m = -1, s = -1/2 |
|
| 21. |
Write the steps and energy change of formation of ionic bond. |
|
Answer» SOLUTION :To form positive ION : Ionization enthalpy is always endothermic. To from NEGATIVE ion : ELECTRON gain enthalpy is always exothermic. To form SOLID : Lattice enthalpy is always more exothermic reaction |
|
| 22. |
What is E_(O_3)in the following reaction, 2O_3 rarr 3O_2 |
|
Answer» 16 2moles of `O_3 = 3xx 4 ` eq of `O_2 (E_(O_2) =32/4)` 1 MOLE of `O_3 = ` 6eq of `O_2` 1 eq. of `O_2 = (48)/6 GM O_3` 1 eq of `O_ 2 = (48)/6 gm O_3` 1 eq of `O_2 =8g ` of `O_3` |
|
| 23. |
What will be the pressure of the gaseous mixture when 0.5 L of H_(2) at 0.8 bar and 2.0 L of dioxygen at 0.7 bar are introduced in a 1 L vessel at 27^(@)C ? |
|
Answer» Solution :Dihydrogen Gas `(H_(2))` Volume `V_(H_(2))=0.5 L` PRESSURE `p_(H_(2))=0.8` bar Dihydrogen Gas `(O_(2))` Volume `V_(O_(2))=2` Lit Pressure `p_(O_(2))=0.7` bar Temperature `= 27^(@)C=300 K` Which is constant Volume of mixture `= V_("total")=1` Lit. Calculation of Partial Presure of dil hydrogen gas `pH_(2)` of 1 Lit. Volume : `p_(1)V_(1)=p_(2)V_(2)` `therefore ("0.8 bar")(0.5 L) = p_(2)xx1L` `therefore p_(2)=(("0.8 bar")(0.5 L))/(1L)` `therefore p_(2)=0.4` bar `= p_(H_(2))` where, At gas `H_(2)` `p_(1)=0.8` bar `V_(1)=0.5 L` `V_(2)=1L` `p_(2)=(?)` Caslculation of Partial Pressure of di Oxygen gas `(PO_(2))` of 1 lit volume : `p_(1)V_(1)=p_(2)V_(2)` `therefore ("0.7 bar")(2.0 L)=(p_(2))(1L)` `therefore p_(2)=(0.7xx2.0)/(1)` bar `therefore p_(2)=1.4` bar `= p_(O_(2))` where, At gas `O_(2)` `p_(1)=0.7` bar `V_(1)=2.0L` `V_(2)=1.0L` `p_(2)=(?)` `p_("total")=p_(H_(2))+p_(O_(2))` = 0.4 bar + 1.4 bar = 1.8 bar |
|
| 24. |
Which of the following act both as oxidising as well as reducing agents? |
|
Answer» `HNO_2` |
|
| 25. |
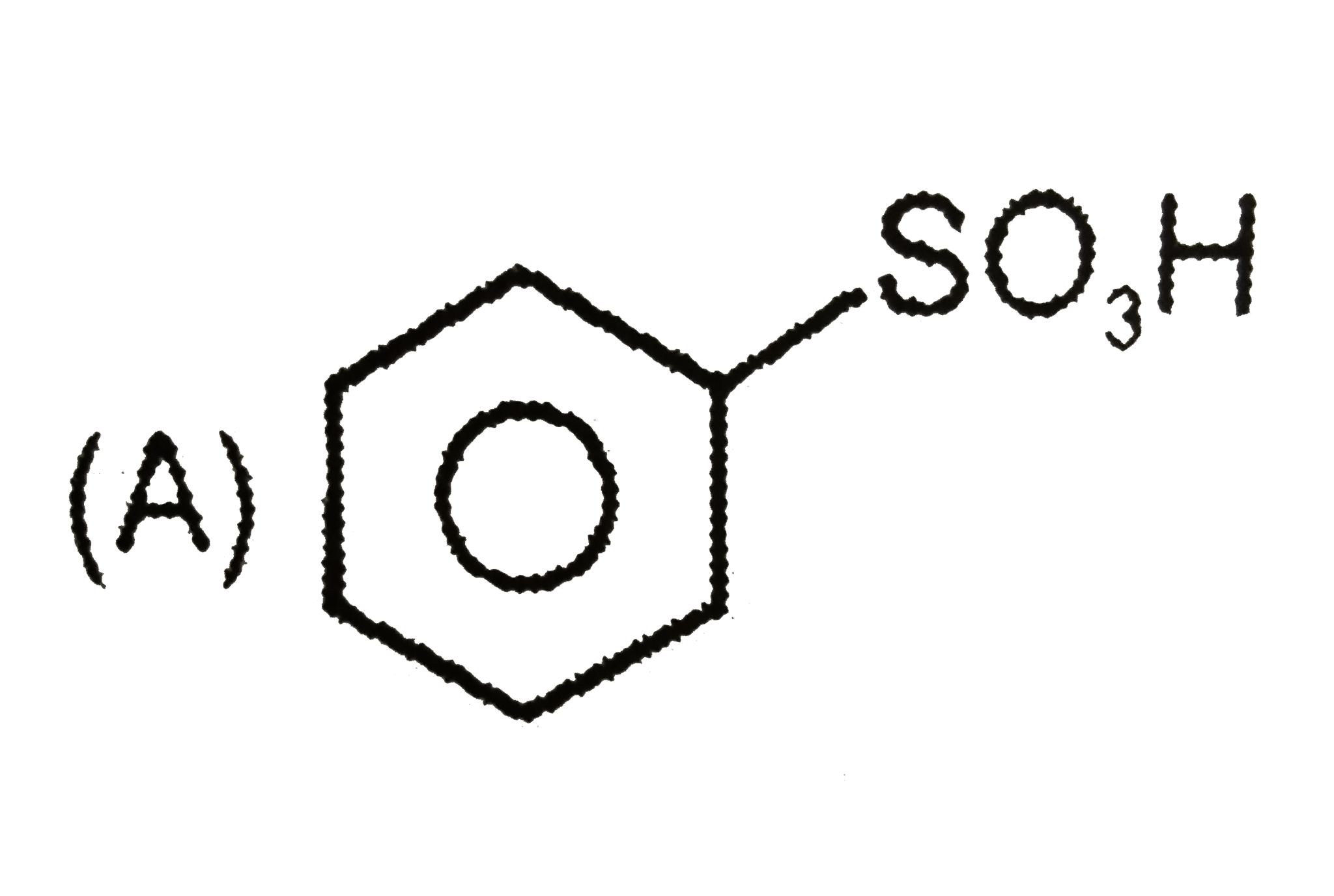
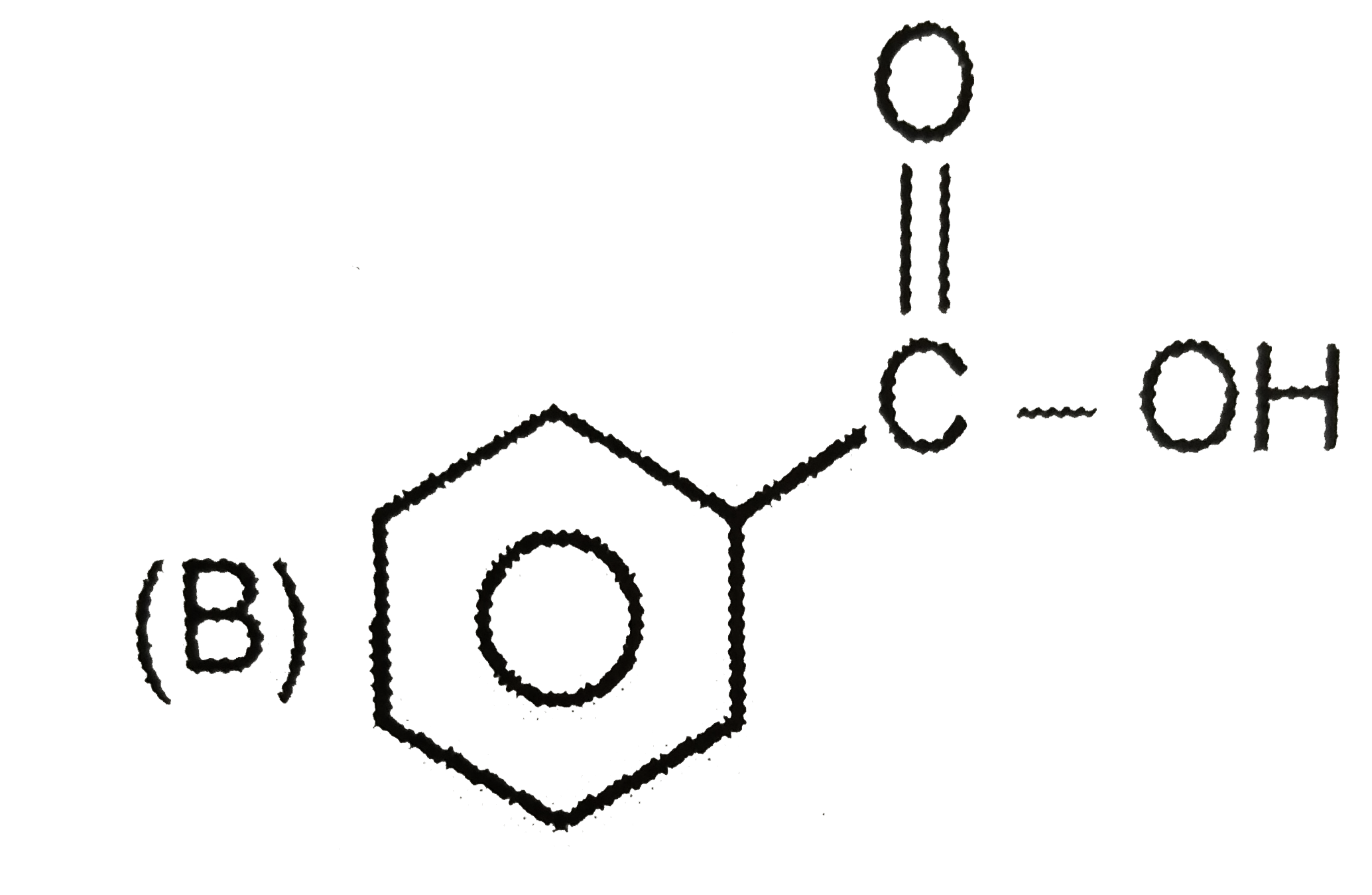
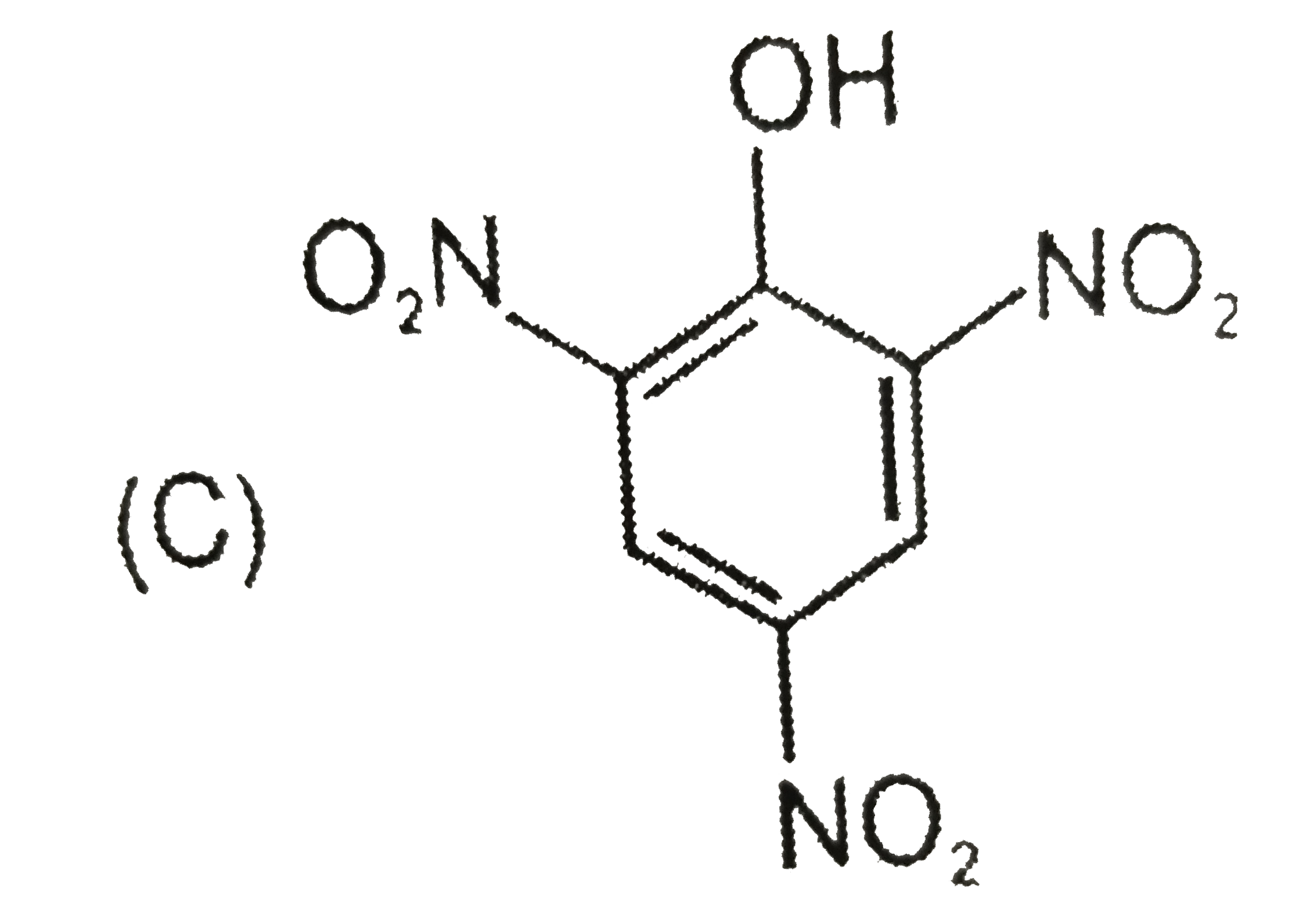
Which of the following reactions will produce anisole? |
|
Answer» Phenol and METHYL chloride 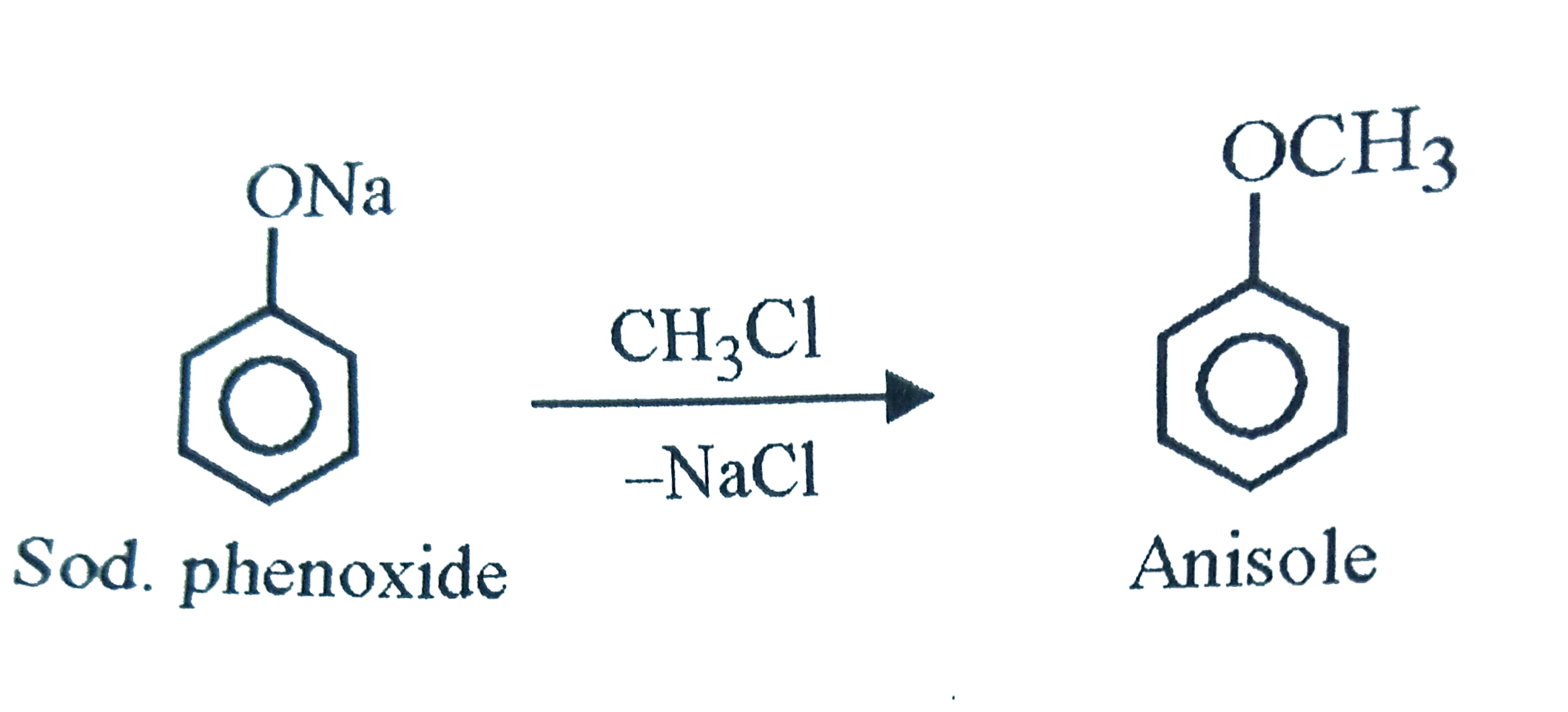
|
|
| 26. |
The solubility product of SrF_(2)in water is 8xx10^(-10) Calculate its solubility in 0.1 M of aqueous NaF solution. If its solubility is expressed as y xx 10^(-8)then what is the value of 'y' ? |
|
Answer» ` [F^(-) ] " from " 0.1 M NAF " is "0.1 M` TOTAL ` [F^(-) ] =0.1 +2S = (0.1 )^(2) ` ` K_(sp)= S( 0.1) ^(2) rArr(8XX 10 ^(-10) )/( 10^(_2)) =SrArr S = 8 xx 10 ^(-8) ` |
|
| 27. |
What would be the IUPAC name and symbol for the element with atomic number 120 ? |
| Answer» SOLUTION :Unbinlium, UBN | |
| 28. |
Toluene undergoes nitration easily than Benzene why? |
|
Answer» Solution :`(i)` Toluene has a methyl group on the benzene ring which is electron releasig group and hence activate the benzene ring by pushing the electrons on the benzene ring by pushing the electrons on the benzene ring. `(ii)` `CH_(3)` group is ortho-paro director and ring ACTIVATOR. There fore in toluene, ortho and para positions are the most reactive towards an ELECTROPHILIC, THUS promoting electrophilic substiution reaction. `(iii)` The methyl group hence makes it around `25` times more reactive than benzene. `:.` it UNDERGOES nitration than benzene. 
|
|
| 29. |
Which of the following is powerful reducing agent that used widely in organic chemistry ? |
|
Answer» `KalH_(4)` |
|
| 30. |
What is the electrophile when RCl + AlCl_3 are used in Friedel-Crafts reaction ? |
|
Answer» `CL^+` |
|
| 31. |
Which one of the following expressions represent the electron probability function (D) |
|
Answer» `4pir" DR "PSI^(2)` |
|
| 32. |
Write the IUPAC names of the following compounds. (i) CH_(3)-underset(CHO)underset(|)(CH)-CH_(2)-CH_(2)-CH_(3) (ii) CH_(3)-CH_(2)-CH_(2)-underset(COOH)underset(|)(CH_(2))=CH-CH_(2)-CH_(3) (iii) CH_(3)-CH_(2)-underset(CH_(3))underset(|)(CH)-CH_(2)-CH_(2)CN |
|
Answer» SOLUTION :(i) PENTAN-2-al (II) Pentan-(2-ene-2-propyl)-1-oic ACID (III) 4-methyl-1-cyanohexane |
|
| 33. |
Which one of the following statement is incorrect? |
|
Answer» `H_(2)` reacts with `Cl_(2)` to form HCl, an electron pair SHARED between H and Cl |
|
| 34. |
Which out of NH_(3) and NF_(3) has higher dipole moment and why ? |
|
Answer» SOLUTION :The dipole moment of `NH_(3) ` is more than `NH_(3)`. Dipole moment of `NH_(3) = 1.47 D = 4.9 xx 10^(-30)` C m Dipole moment of `NF_(3) = 0.23 D = 0.8 xx 10^(-30)` C m `mu (NH_(3)) gt mu (NF_(3)) : ` Both the molecules have pyramida shape with a lone pair of electrons on nitrogen atom.  Electronegativity: H (2.1), N (3.0) and F (4.0) os, partially N NEGATIVE in `NH_(3)` but positive in `NF_(3)`. In case of `NF_(3)` the orbital dipole due to lone pair is in the same direction as the RESULTANT dipole moment of the N - H bonds where as in `NF_(3)` the orbital dipole is in the direction opposite to the resultant dipole moment of the three N - F bonds. The orbital dipole because of lone pair decreases the effect of the resultant N - F bond moments. Which results i the low dipole moment of `NF_(3)` as represented below. 
|
|
| 35. |
Which of the following respresents 2, 2, 3-trimethyl hexane? |
|
Answer» `CH_3 - C(CH_3)_2 - CH_2 - CH_2 - CH (CH_3)_2` |
|
| 36. |
Viscosity of liquids decreases with increase of temprature. Why? |
| Answer» Solution :VIscosity of liquids decreases as the temperature RISES because at high temperature MOLECULES have high kinetic ENERGY and can overcome the intermolecular forces to SLIP PAST one another between the layers. | |
| 37. |
What is the type of hybridisation of each carbon in the following compounds? (a) CH_(3)Cl (b) (CH_(3))_(2)CO (c ) CH_(3)CN (d) HCONH_(2) (e ) CH_(3)CH = CHCN |
Answer» SOLUTION : `sp^(3)` carbon has FOUR single bond `sp^(2)` carbon has one double bond sp carbon has one TRIPLE bond or two double bond |
|
| 38. |
The resonance effect of groups is electron removing (+R) or electron attracting (-R)? |
|
Answer» Solution :`+R` effect containing : In (a), `-OH` group, the MOLECULE push the ELECTRON away from it. (-R) effect containing group: In (b) `-NO_(2)`, (C ) `-CHO` (d) -`CHO` (e ) `OVERSET(+)(C )H_(2)` and (F) `overset(+)(C )H_(2)`. These molecule attract the `pi` electron towards it |
|
| 39. |
What is theuncertaintyDelta v . Delta xfor 1 mgobject? Whatin indicate ? |
|
Answer» SOLUTION :`Delta v . Delta X= (h)/( 4PI m) = (6.626 xx 10^(34) j s)/(4XX 3.1416xx 10^(6)kg )` Thevalue of`Delta v. Delta x`obtainedextremely smalland isinsignificant. |
|
| 40. |
Which among the following has zero dipole moment BF_(3),mH_(3),H_(2)O? |
| Answer» SOLUTION :`BF_(3)` | |
| 41. |
Which reaction is not occuring in Solvay process ? |
|
Answer» `(NH_(4))_(2)CO_(3)+H_(2)O +CO_(2) to 2NH_(4)HCO_(3)` |
|
| 42. |
Which of the compounds with molecular formula C_5H_10 yields aceton on ozonolysis ? |
|
Answer» 2-Methyl-1-butene |
|
| 43. |
Which elements accept2e^(-) " or" 1e^(-)and from stable structure like noble gas ? |
| Answer» Solution :Elements of group-16 accept 2e and form stable STRUCTURE and elements of group-17 accept `1E^(-)`and form stable structure because their electron NEGATIVITY ENTHALPY is more. | |
| 44. |
What are the conventions adopted in writing thermochemical equation? |
|
Answer» Solution :A thermochemical equation is a balanced stoichiometric chemical equation that includes the enthalpy change `(DeltaH)`. Conventions adopted in thermochemical EQUATIONS: (i) The coefficients in a balanced thermochemical equation REFER to number of moles of reacts and products involved in the reaction. (ii) The enthalpy change of the reaction `DeltaH` has unit kJ. (iii)When the chemical reaction is reversed, the value of All is reversed in sign with the magnitude (iv)Physical states (GAS, liquid, aqueous and solid) of all species is important and must be SPECIFIED in a thermochemical reaction since `DeltaH` depends on the phases of reactants and products (v) If the thermochemical equation is MULTIPLIED throughout by a number, the enthalpy change is also be multiplied by the same number value. (vi) The negative sign of `DeltsH_(r)^(@)` indicates the reaction to be an exothermic and the positive sien of `DeltsH_(r)^(@)`indicates an endothermic type of reaction. |
|
| 45. |
The group 1 metals of the periodic table of elements are collectively called alkali metals. Alkali metals are never found free in nature. Give reason. |
| Answer» Solution :They are HIGHLY REACTIVE and hence do not OCCUR FREE. | |
| 46. |
Which combination cannot be used for the preparation of hydrogen gas in the laboratory ? I. Zinc/conc. H_(2)SO_(4) II. Zinc/dil. HNO_(3) III. Pure zinc/dil. H_(2)SO_(4) |
|
Answer» I and II |
|
| 47. |
Which of the following compounds will show tautomerism ? |
|
Answer» 2,2-Dimethylpropanal 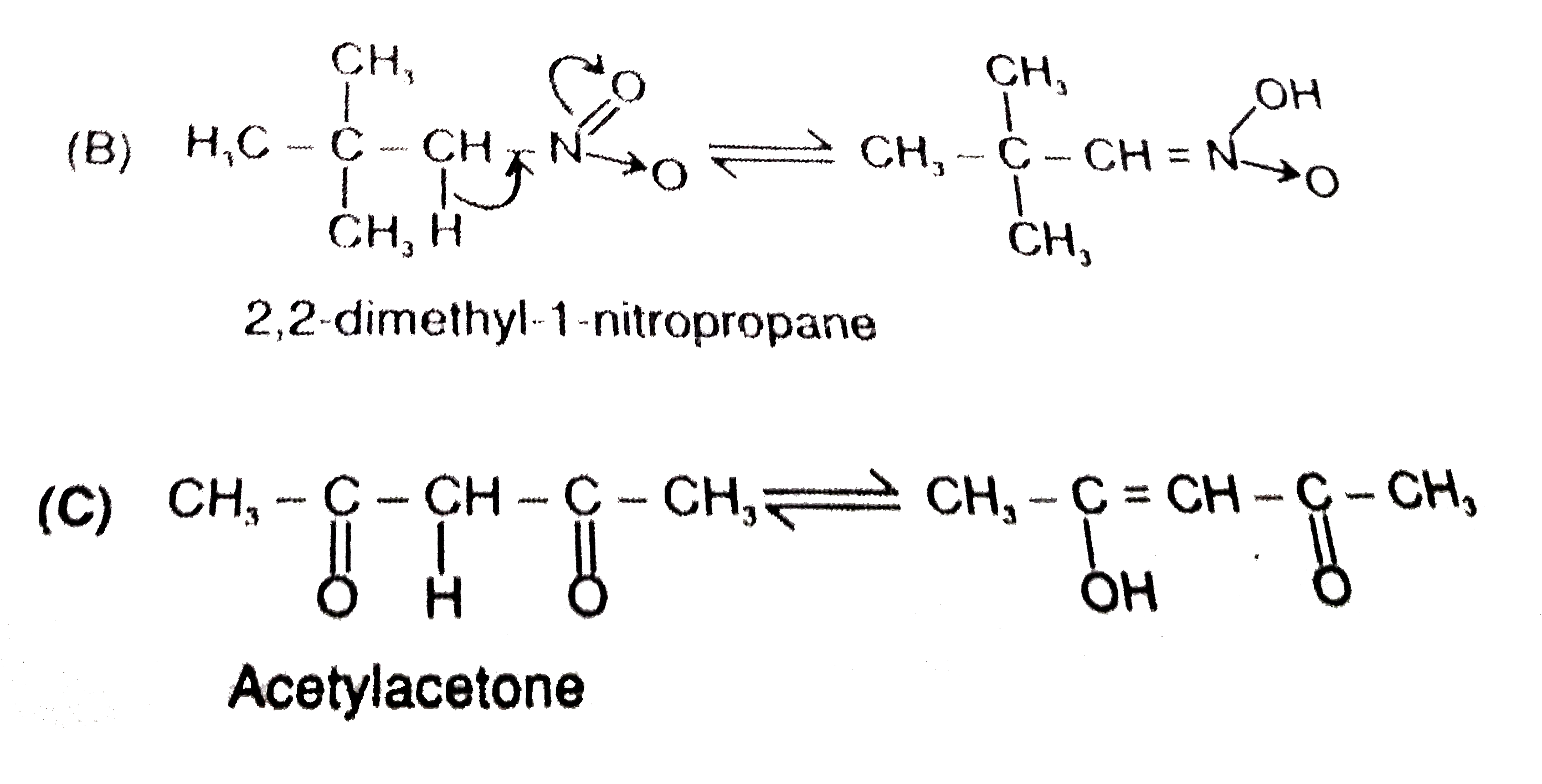
|
|
| 48. |
Which of the following would produce effervescence with sodium bicarbonate ? |
|
Answer»
|
|
| 49. |
What is nascent hydrogen? |
| Answer» SOLUTION :Nascent HYDROGEN or 'just born hydrogen' is the hydrogen at the MOMENT of its GENERATION. | |
| 50. |
What is the significance of the word average in the definition of atomic mass ? |
| Answer» Solution :The term AVERAGE in the definition of ATOMIC MASS signifies that the atomic mass of the GIVEN element is the average of atomic masses of all the known isotopes of that element in the RATIO of their occurrence in nature. | |