Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which hybridization is found in HClO_(4) and HClO_(3)? |
| Answer» ANSWER :A | |
| 2. |
What would be the heat released when an aqueous solution containing 0.5 mol of HNO_(3) is mixed with 0.3 mol of OH^(-) (enthalpy of neutralisation is -57.1 kJ)? |
|
Answer» `28.5 kJ` `H^(+) + OH^(-) to H_(2)O`,`DELTAH = -57.1 kJ` Here `OH^(-) = 0.3 mol` . Therefore, `OH^(-)` is the limiting reactant. When 1 mol of `OH^(-)` ions are NEUTRALISED, heat released = 57.1 kJ For 0.3 mol of `oh^(-)` ions, heat released `= 57.1 XX 0.3= 17.13` kJ |
|
| 3. |
Which of the following species is/are planar ? |
|
Answer» tert-Butyl free radical |
|
| 4. |
What is the volume (lit) of oxygen required at STP to completely convert 1.5 moles of sulphur into sulphurdioxide |
|
Answer» 11.2 |
|
| 5. |
Using the equation of state pV= nRT show that at a given temperature, the density of the gas is proportion a to gas the gas pressure P. |
|
Answer» Solution :According to ideal gas equation. `PV= NRT or P=(nRT)/(V)` `N=("MASS of gas"(m))/("Molar massof gas"(M))` `P=(mRT)/(mV)` Now, density `(d)=(m)/(v) or P=(dRT)/(M)` `(d prop P)` at CONSTANT temperature. |
|
| 6. |
Two types of F X F angles are present in which of the following molecules ? |
|
Answer» `SF_(4)` 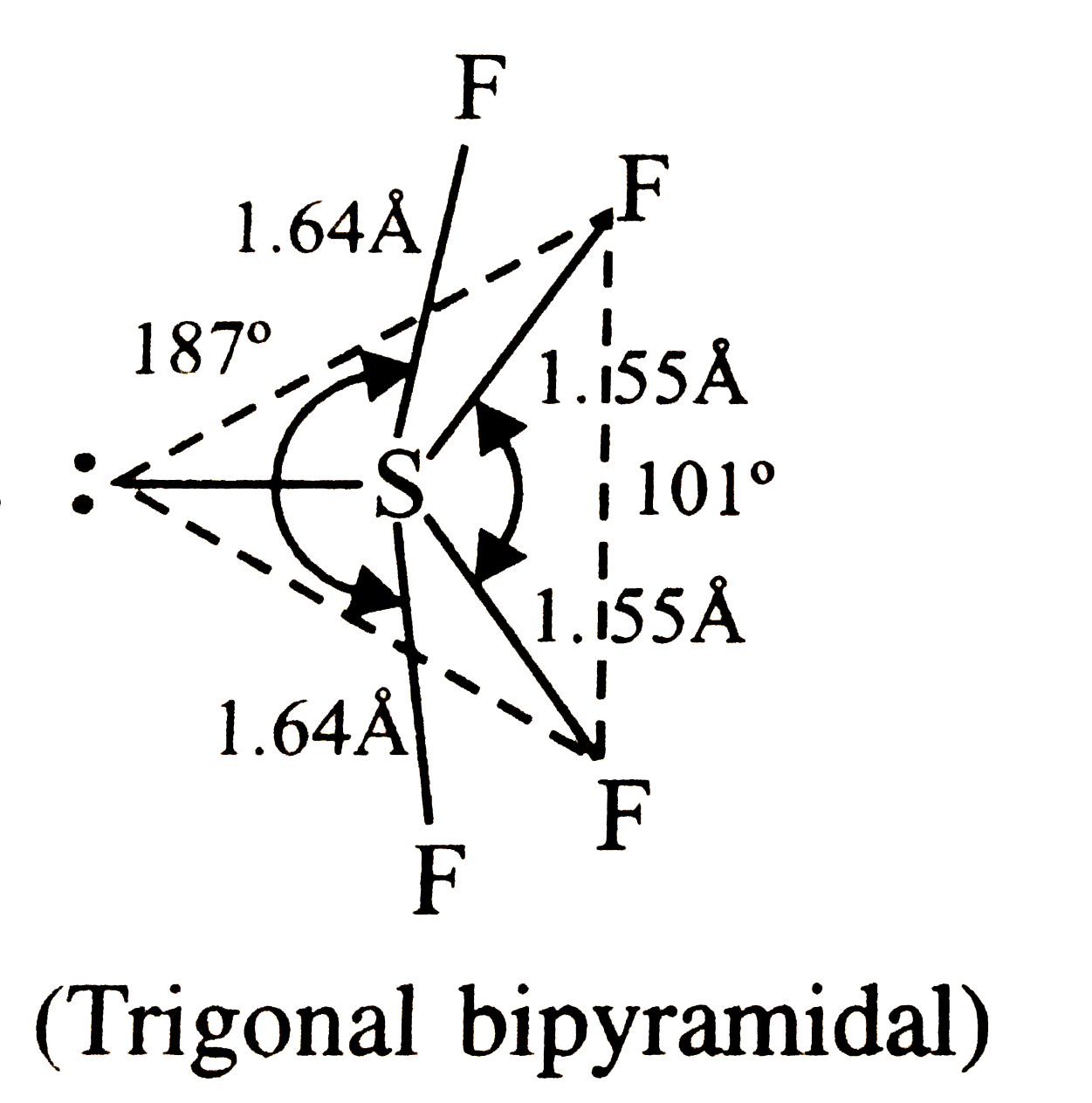
|
|
| 8. |
Which of the following is the energy of a possible excited state of hydrogen ? |
|
Answer» `+13.6 EV` |
|
| 9. |
The species Ar, K^(+) and Ca^(2+) contain the same number of electrons. In which order do their radii increase ? |
|
Answer» `Ca^(2+) LT K^(+) lt Ar` |
|
| 10. |
What is vapour of CO_(2) ? |
| Answer» SOLUTION :Temperature of `CO_(2)` gas is decreases than its critical temperature then it will convert into VAPOUR FORM then it is known as vapour of `CO_(2)`. | |
| 11. |
Which of the following bicarbonate is insoluble in water? |
|
Answer» `Mg(HCO_(3))_(2)` |
|
| 12. |
Which of the following is a redox reaction? |
|
Answer» `NaCl+KNO_(3) to NaNO_(3)+KCL` |
|
| 14. |
Which of the following possess tertiary carbon ? |
|
Answer» Propane (B) `underset("n-Butane")(overset(1^(@))(C_(3))overset(2^(@))(CH_(2))overset(2^(@))(CH_(2))overset(1^(@))(CH_(3)))` (C)`underset("2-Methyl propane")(overset(1^(@))(CH_(3))-underset(""^(1^(@))CH_(3))underset(|)overset(3^(@))(CH)-overset(1^(@))(CH_(3)))` `underset("2,2-Dimethyl propane")(overset(1^(@))(CH_(3))-underset(""^(1^(@))CH_(3))underset(|)overset(1^(@))overset(CH_(3))overset(|)(""^(4^(@))CH)-overset(1^(@))(CH_(3)))` |
|
| 15. |
When burnt in air, 14.0g mixture of carbon and sulphur gives a mixture of CO_(2) and SO_(2) in the volume ratio of 2:1, volume being measured at the same conditions of temperature and pressure the number of moles of carbon in the mixture is |
|
Answer» 0.75 `(x//12)/((14-x)//32)=(2)/(1)` `therefore x=6g," MOLES of C"=(6)/(12)=0.5` |
|
| 16. |
Which of the following is used in Ion Exchange method as a lon Exchanger ? |
|
Answer» Alum |
|
| 17. |
What is the hybridisation of p inPCl_5. Why are axial P-Cl bonds longer than the equatorial bonds? |
|
Answer» <P> SOLUTION :Hybirdisation of P in `PCl_5` is `sp^3`d. SINCE the axial bond pairs suffer more repulsive INTERACTION from the EQUATORIAL bond pairs,the axial `P-Cl` bonds are slightly longer than the equatorial bonds. |
|
| 18. |
What is the action of alkali on an ammonium salt? |
|
Answer» Solution :Ammonia is realsed when ALKALI is treated with an ammonium salt. `NH_(4)CI + NAOH to NACI + H_(2)O +NH_(3)` This is because alkali is stronger base than ammonia. |
|
| 19. |
Van der waal's radius is measured for |
|
Answer» MOLECULAR substances in GASEOUS STATE only |
|
| 20. |
The two structures of benzene proposed by Kekule differ in |
|
Answer» The POSITION of CARBON nuclei |
|
| 21. |
Which of the following can show geometrical isomerism |
|
Answer» 2,3 - DIMETHYL butane  (CIS and trans) geometrical isomerism is possible in cyclic compounds (CIS and trans) geometrical isomerism is possible in cyclic compounds
|
|
| 23. |
Which ordering of compounds is according to the decreasing order of the oxidation state of nitrogen ? |
|
Answer» `HNO_(3), NO, NH_(4)Cl , N_(2)` |
|
| 24. |
What is the ratio of mass an electron to the mass of a proton |
|
Answer» `1:2` |
|
| 25. |
Which onc of the following is mainly responsible for the depletion of ozone layer |
|
Answer» Methane |
|
| 26. |
Which of the following are correct |
|
Answer» gases with EQUAL P,T and `M_1 gtM_2`, then `d_1gt d_2` b) equal P,T V `IMPLIES ` equal moles `implies` equal molecules. C) `N_1 > N_2 implies n_1 > n_2 , n alpha 1/T = T_2 > T_1` d) `n ,T` same `implies P alpha 1/V implies P_1 > P_2 implies V_1 < V_2`. |
|
| 27. |
What is the oxidation number of sulphur in the following molecules/ ions SO_4^(2-) |
|
Answer» SOLUTION :In `SO_4^(2-)` LET x be oxidation number of S, Oxidation number of Oxygen is -2. `S O_4^(2-)` Let oxidation number be x -2 `THEREFORE (1 times x)+ (4 times -2)=-2 implies x-8=-2 implies x=-2+8 implies x=6` Oxidation number of S in `SO_4^(2-)` is +6. |
|
| 28. |
What weight of AgCl would be precipitated if 10 mL of HCl gas measured at 12^@C and 750 mm pressure were passed into the excess of a solution of silver nitrate ? |
| Answer» SOLUTION :0.0605 G | |
| 29. |
Wavelength of an electron is 5Å Velocity of the electron is |
|
Answer» `1.45 XX 10^(8)` cm/s |
|
| 30. |
Which of the following practices will not come under Green chemistry? |
|
Answer» Use of `CO_2` as SOLVENT INSTEAD of `CI_2` |
|
| 31. |
Which one of the following chlorides will not fume in air ? |
| Answer» Solution :`C Cl_(4)` will not fume in moist air. | |
| 32. |
What do you understand by exothermic reaction and endothermic reaction? Give one example of each type. |
|
Answer» Solution :Exothermic reactions : Reactions which evolves HEAT are known to be as exothermic reactions. Evolution of heat PRODUCED can be in the form of `.+. sign or DeltaH` in - sign along with product. `C_((s))+O_(2(g))to CO_(2(g))+393.5 kJ` `H_(2(g))+(1)/(2)O_(2(g)) to H_(2)O_((L)),` `DeltaH=-285.8 kJ"mol"^(-1)` Endothermic reactions : Reactions which absorbs heat are called endothermic reactions. Absorbed heat can be is shown as - sign or in terms of `DeltaH` with a+ singn E., `C_((s))+H_(2)O_((g)) to CO_((g))+H_(2(g))-131.4 kJ` `N_(2(g))+3H_(2(g)) to 2NH_(3(g)), DeltaH=+92.4 kJ mol^(-1)` |
|
| 33. |
Which of the following is not the correct order of stability? |
|
Answer» `CH_(3)-OVERSET(oplus)(CH_(2)) gt CH_(2)=overset(oplus)(CH)` `R - underset( * * )overset(* *)O- CH_2 harr R - overset(oplus)(O) = CH_2` |
|
| 34. |
To neutralise completely 20 mL of 0.1 M aqueous solution of phosphorus acid (H_(3)PO_(3)), the volume of 0.1 M aqueous KOH solution required is : |
|
Answer» 10 mL `(M_(1)V_(1))/(n_(1))=(M_(2)V_(2))/(n_(2))` `(0.1xx20)/(1)=(0.1xx V_(2))/(2)` `V_(2)= 40 mL` |
|
| 35. |
Which of the following is easily formed? |
|
Answer» CALCIUM chloride |
|
| 36. |
Water is often treated with chlorine to |
|
Answer» INCREASE OXYGEN content |
|
| 37. |
What do you understand by 'continuity of state' in gases and liquids ? |
| Answer» Solution :By SUITABLY ADJUSTING the conditions of temperature and pressure,it is possible to change a gas into a liquid or a liquid into a gas such that there is ALWAYS a single phase PRESENT. This is called CONTINUITY of liquid state. | |
| 38. |
Why can Solvay process not be used for the manufacture of potassium carbonate? |
| Answer» SOLUTION :`KHCO_3`is FAIRLY SOLUBLE in water EVEN in the PRESENCE of `CO_2`. | |
| 39. |
Which of the followingprocess must bespontaneous ? |
|
Answer» Isobaric and isoentropic PROCESS with negativeenthalpy charge. |
|
| 40. |
Which of the following has lowest melting point ? |
|
Answer» B |
|
| 41. |
Which of the following is the first member of monosaccharides ? |
|
Answer» `CH_(2)OH-OVERSET(O)overset(||)(C)-CH_(2)OH` |
|
| 42. |
Van der Waals constant 'b' for helium is 2.93 xx 10^(-5) m^(3) mol^(-1). Calculate actual volume of helium atom. What is the radius of helium atom? |
|
Answer» SOLUTION :Excluded volume = `b = 4 xx (V_a xx N_0)` where `V_a` is the actual volume of helium atom. `V_a = (b)/(4N_0) = (2.93 xx 10^(-5))/(4 xx 6.022 xx 10^(23))` `= 1.02 xx 10^(-29) m^3` Volume of helium atom = `4/3 PI R^3` Radius of helium atom, `r = 1.3 xx 10^(-10) m`. |
|
| 43. |
Which among the following statement are correct with respect to adsorption of gases on a solid ? (1) The extent of adsorption is equal to kp^(n) according to Freundlich isotherm (2) The extent of adsorption is equal to kp^(1//n) according to Freundlich isotherm (3) The extent of adsorption is equal ((a+bp))/(ap) according to Langmuir isotherm The extent of adsorption is equal (ap)/((1+bp)) according to Langmuir isotherm (5) Freundlich adsorption fails at a low pressure |
|
Answer» 1 and 3 `(x)/(m)=kp^(1//n)` According to Langmuir isotherm, `(x)/(m)=(ap)((1+bp))` |
|
| 44. |
Why boron and aluminium tend to for, covalent compounds ? |
| Answer» Solution :Both boron and aluminium have give high sum total of first THREE ionisation enthalpies `(IE_(1)+IE_(2)+IE_(3))`.Hence, they cannot LOSE electrons easily to form `B^(3+)` ion or `Al^(3+)` ion. Hence, they form covalent compounds. | |
| 45. |
What is stoichiometry ? |
|
Answer» |
|
| 46. |
Which of the following cannot be used as a test for H_(2)O_(2)? |
|
Answer» A paper dipped in PBS (BLACK) true white when brought in contact with `H_(2)O_(2)`. |
|
| 47. |
Whichorbitals are thenin hydrogenatom ? |
| Answer» SOLUTION :In HYDROGENATOM in GROUND state1s ORBITAL is there in exicited stateall the orbitals2s, 3s,3p3d,4s,4p ,4d,4f | |
| 48. |
Which one is rapidly degradable pollutants ? |
|
Answer» Agriculture waste |
|
| 49. |
Which property is same for both normal hydrogen and deuterium |
|
Answer» BOILING point |
|