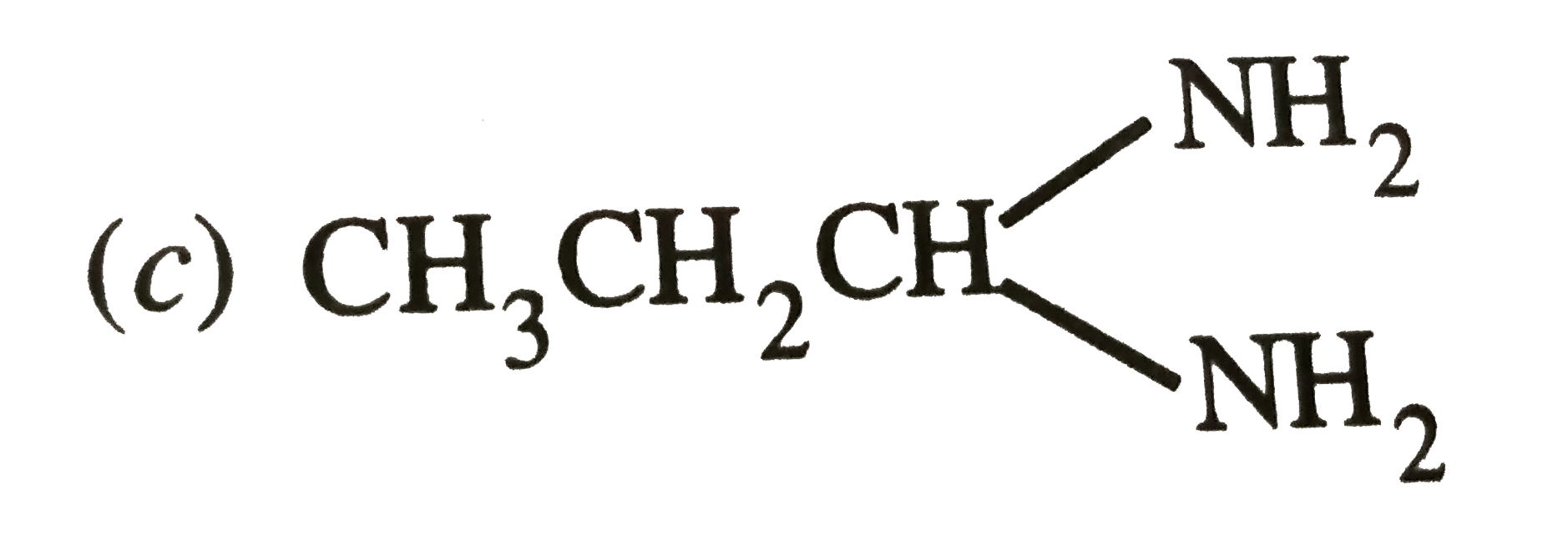Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which of the following shows correct relation between volume and temperature at constant pressure for a given amount of gas |
|
Answer»
|
|
| 2. |
Under a gives conditions, the rate of diffusion of CH_(4) gas is sqrt(2) times that of B_(2) gas. Gas B_(2) reacts with element Ato form gaseous compounds AB_(2) and AB_(3). Under a given conditon, the rate of diffusion of AB_(2) is 1.12 times that of AB_(3). the atomic mass of A (in g*mol^(-1)) - |
|
Answer» 32 |
|
| 3. |
Which of the following neutral molecule is not electrophile? |
|
Answer» `AlCl_(3)` |
|
| 4. |
What will be the correct order of vapour pressure of water, acetone and ether at 30^(@)C. Given that among these compounds, water bus maximum boiling point ? |
|
Answer» |
|
| 5. |
Why are lithium salts commonly hydrated and those of other alkail metal ions usually anhydrous? |
| Answer» Solution :`Li^+` ion being very small in size redily POLARISES water MOLECULES. THUS `H_2O`molecules are easily attaced to lithium slts (hydration) | |
| 6. |
The temperature of troposphere decreases with altitude and is minimum at about 10 km. This point of temperature inversion is known as |
|
Answer» TROPOPAUSE |
|
| 7. |
What are silicones ? |
| Answer» Solution :Simple SILICONES `(--underset|overset|Si-O--)_n` consist of chains in which alkyl or PHENYL groups OCCUPY the remaining bonding positions on each silicon. They are hydrophobic (water repellant) in NATURE. | |
| 8. |
Which of the following compounds will not give Hoffmann's hypobromite reaction ? |
|
Answer»
|
|
| 9. |
What indicate the ([A^-])/([HA]) |
| Answer» Solution :It is a ratio of concentration of conjugate BASE `A^-` and WEAK acid HA of acid in acidic buffer solution. | |
| 10. |
Which one of the following is used for laboratory preparation of D_(2)O_(2) |
|
Answer» `K_(2)S_(2)O_(8)` |
|
| 11. |
What are electrophiles and nucleophiles ? Give suitable examples for each. |
|
Answer» Solution :NUCLEOPHILES are reagents that have high affinity for electro positive centres (any two EXAMPLES) ELECTROPHILES are reagents that are attracted TOWARDS negative charge or electron rich CENTER.(any two examples) 
|
|
| 12. |
Which of the following compounds on its oxidation gives methyl ethyl ketone ? |
|
Answer» Propan-2-ol |
|
| 13. |
Zn(s)+Cu^(2+)to Zn^(2+)+Cu(s) . Is this reaction a redox reaction? IF yes, name the oxidizing agent as well as the reducing agent? |
| Answer» Solution :Yes `Cu^(2+)` is the OXIDIZING agent WHEREAS ZN is the REDUCING agent. | |
| 14. |
what is the maximum number of electrons that can be accommodated in a shell ? |
| Answer» Solution :Maximum number of ELECTRONS in any SHELL is GIVEN by the expression `2n^2`. Where n is the PRINCIPAL quantum number. | |
| 15. |
Which of the following represented the correct IUPAC names for the compounds concerned ? (a) 2, 2-Dichloropentane or 2-Dichloropentane (b) 2, 3-Dimethylpentane or 3, 4-Dimethylpentane (c) 2-Bromo-4-methylpentane or 4-Bromo-2-methylpentane (d) But-3-en-1-ol or But-4-ol-1-ene. |
|
Answer» SOLUTION :(a) 2, 2-Dichloropentane (B) 2, 3-Dimethylpentane (C) 2-Bromo-4-methylpentane (d) But-3-en-1-ol |
|
| 16. |
What is the hybridization of carbon in above compounds ? |
| Answer» Solution :`sp^(3)` in ETHANE, `sp^(2)` in ethene, sp in ETHYNE and `sp^(2)` in BENZENE. | |
| 17. |
When N_(2)(g) "and" H_(2)(g)are mixed N_(2)H_(4)(g),NH_(3)(g) or both may form, depending upon the relative amount of N_(2)and H_(2) taken. If initial mols of N_(2), H_(2)are x,y and final moles of N_(2)H_(4),NH_(3) are z,v, then the correct optionsfrom the following in order of (x,y,z,v) is/are |
|
Answer» (2,2,1,0) |
|
| 18. |
When CH_3CH_2CHCI_2, Is treated with NaOH_2, the product formed is |
|
Answer» `CH_3-CH=CH_2` |
|
| 19. |
When alkali metal dissolve in ammonia, it produces blue color solution. This color is due to ...... |
| Answer» SOLUTION :AMMONIUM ELECTRON | |
| 20. |
What are the similar properties of Li and Mg. |
| Answer» Solution :(a) Both Li and MG are hard. (b) Li(OH) and Mg`(OH)_(2)` are bases. (C ) Both Li and Mg react with ATMOSPHERE nitrogen on HEATING gives their nitrides. (d) Both Li and Mg FORMS only monoxide but not peroxides and superoxides. | |
| 21. |
What is the differencein line spectrum ofdifferentatomsand hydrogenatom ? |
|
Answer» SOLUTION :Ingasesstate all atomshave linespectrum. For all the atomlinespectrum is uniquemeansit does notresemblewith any ATOM . Hydrogenspectrum : In all regularityis observedof hydrogenissamplebut forheavyelementsor atomsits ISMORE and morecomplex. |
|
| 22. |
Write the K_(c) for the reaction CO_(2)(g) + H_(2)O(l) hArr H_((aq))^(+) + HCO_(3)^(-) |
|
Answer» SOLUTION :`H_(2)O(l)` is a PURE LIQUID ant its CONCENTRATION remains constant. So,`K_(c) = ([H^+]_(aq) xx ([HCO_3^(-)]_(aq)))/([CO_2]_g)` |
|
| 23. |
Write the IUPAC name for the following compound. (A) CH_(3)-underset(CH_3)underset(|)CH-CH_(2)-CH_(3) (B) H_(3)C-underset(CH_3)underset(|)overset(CH_3)overset(|)C-CH_(3) (ii) What are Nucleophiles and Electrophiles ? Give one example each. |
|
Answer» Solution :(A) `overset(1)CH_(3)-underset(3)underset(|)overset(2)CH-overset(3)CH_(2)-overset(4)CH_(3)-3` methyl butane (B) `H_(3)overset(1)C-overset(CH_3)overset(2|)underset(CH_3)underset(|)C-overset(3)CCH_3` -2,2- dimethyl propane (ii) Electrophiles : Electrophiles are reagents that are attrached towards negative charge or electron rich center. They are either positively CHARGED ions or electron deficient neutral meolecules. Example , `CO_(2)AlCl_(3) , BF_(3), FeCl_(3),NO,NO_(2)` etc . Nucleophiles : Nucleophiles are ragents that has high affinity for electropostive centers. They possess an ATOM has unshared pair of electrons. They are usually NEGATIVELY charged ions or electron rich neutral molecules. Example : `NH_(3),R-NH_(2)R-SH,H_(2)O, R-OH, CN, OH` etc |
|
| 24. |
Write short note on Dihydrogen. |
|
Answer» Solution :Occurrence of DIHYDROGEN : (i) Dihydrogen is the most abundant element in the universe and is the principal element in the solar atmosphere. (II) HOWEVER, due to its light nature, it is much LESS abundant (0.15 % by mass) in the earth.s atmosphere. (iii) In the combined FORM it constitutes 15.4% of the earth.s crust and the oceans. |
|
| 25. |
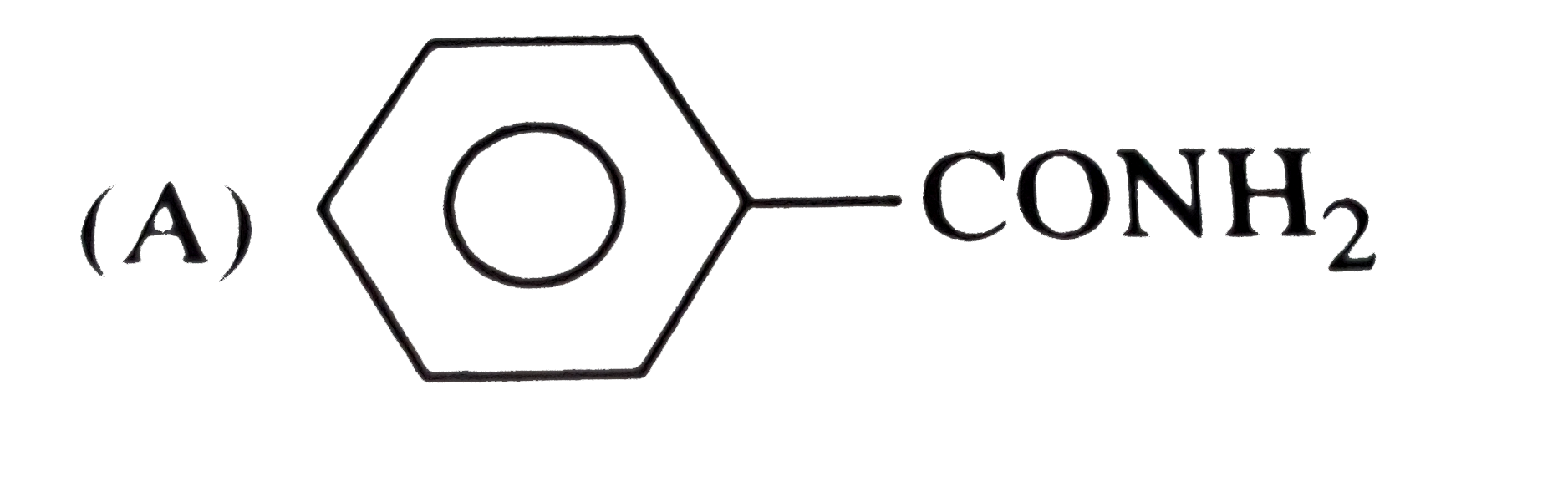
Which hybrid orbitals are used by carbon in the following molecules ? (a) CH_(3) -CH_(3) (b) CH_(3) - CH=CH_(2) (c) CH_(3) - CH_(2) - OH (d) CH_(3) - CHO(e) CH_(3) COOH |
Answer» SOLUTION : 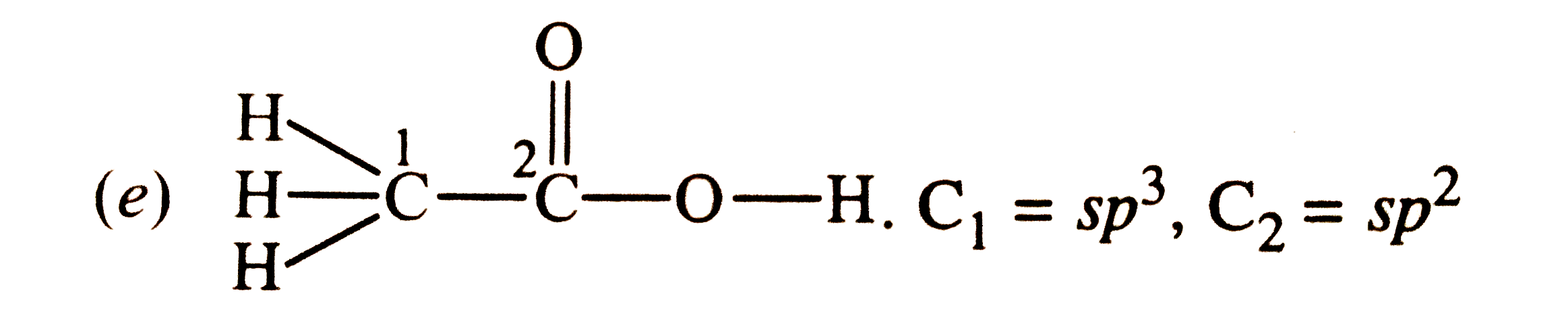
|
|
| 26. |
Whichpair ofatomicnumbersrepresents s- blockelements ? |
| Answer» SOLUTION :NA | |
| 27. |
Which is the prefix of ether in IUPAC nomenclature? |
|
Answer» a L |
|
| 28. |
Use molecular orbital theory to explain why the Be_(2) molecule does not exIst. |
|
Answer» Solution :E.C. of Be `= 1s^(2)" "2S^(2)` M.O.E.C. of `Be_(2)=sigma1s^(2)" "SIGMA2S^(2)" "SIGMA^(**)2s^(2)` Bond order `=(1)/(2)(4-4)=0` HENCE, `Be_(2)` does not exits. |
|
| 30. |
Two moles of H_(2) and three moles of I_(2) are taken in 2 dm^(3) vessel and heated. If the equilibrium mixture contains 0.8 moles of HI, calculate K_(p) and K_(c) for the reaction H_(2(g))+I_(2(g))hArr 2HI_((g)) |
|
Answer» |
|
| 31. |
Two gases A and B having the same volume diffuse through a porous partition in 20 and 10 seconds respectively, the molecular mass of 'A' is 49 amu. Molecular mass of 'b' will be: |
|
Answer» 25 amu `(V//20)/(V//10) = sqrt((m_(2))/(49))` `(1)/(2) = sqrt((m_(2))/(49))` `m_(2) = 12.25 am u` |
|
| 32. |
What is the percent by mass of titanium in rutile, a mineral that contains Ti and oxygen if structure can be descirbed as a a closest packed array of oxide ions with titanium ions in one- half the octahedral holes ? What is the oxidation number of Ti ( Atomic mass of Ti= 48) |
|
Answer» Solution : If no .of oxide ions = n , then octahedral holes = n and hence ` O^(2-)" ions" = n/2` ` Ti =O = n/2 : n = 1/2 : 1 = 1: 2 ` Hence, FORMULA of the oxide is ` TiO_(2)` . Molar mass of ` TiO _(2) = 48 +32 = 80 G MOL^(-1)` % of Ti ` = 48/80 xx 100 = 60% ` Oxidation state of Ti in `TiO_(2) = +4` |
|
| 33. |
Which of the following statement is true |
|
Answer» The pressure of a fixed amount of an ideal gas is propotional to its TEMPERATURE on |
|
| 34. |
What is triad system? Give example. |
|
Answer» Solution :In this system hydrogen atom oscillates between THREE polyvalent atoms. It involves 1, 3-migration of hydrogen atom from one polyvalent atom to other with in the molecule (ii) The most IMPORTANT type of triad system is keto-ENOL tautomerism and the two groups of tautomers are keto form and enol form 
|
|
| 36. |
Which of the following is an n-type semiconductor ? |
|
Answer» SI doped with Ga |
|
| 37. |
Which of the following molecular orbital has the lowest energy for O_(2) molecule? |
|
Answer» `sigma_(2pz)` |
|
| 38. |
Which factor affecting on ionisation enthalpy? |
| Answer» SOLUTION :(1) Atomic VOLUME (2) Nuclear charge (3) CORC orbitals and effective nuclear charge. (4) Effect of electron of s, p, d, F. (5) electronic configuration. | |
| 39. |
Write the general electronic configuration of sp-d-, and f-block elements? |
|
Answer» Solution :(i) s-block elements: `NS^(1-2)` where n=2-7. (ii) p-block elements, `ns^(2)np^(1-6)` where n-2-6 (III) d-block elements: `(n-1)d^(1-10)ns^(0-2)` where n=4-7. (IV) F-block elements: `(n-2) f^(0-14)(n-1)d^0-1)ns^(2)` where n=6-7. |
|
| 40. |
Which of the following has the lowest Lattice energy ? |
|
Answer» LIF |
|
| 41. |
Which of thefollowingatom has the highestfirstionizationenergy ? |
|
Answer» Rb |
|
| 42. |
What can we do to reduce the rate of global warming ? |
|
Answer» Solution :Greenhouse gases increases in the atmosphere because buring of fossil FUELS, cutting down forest and TREES. Due to these the global warming take place. Some of the solution for prevention are as follows: (i) Control on USE of chemical and other things that are responsible for global warming, which reduce the atmospheric temprature. (II) Minimise the use of automobile. e.g., one can use bicycle, public transport system or GO for carpool. (iii) We should plant more trees to increase the green cover. Avoid burning of dry leaves, wood etc. (iv) Do not smoke in public palces and work places. (v) Many people do not understand the greenhouse effect and the global warming. So aware them by sharing this information. |
|
| 43. |
V_(1) mL of xN NaOH and V_(2) mL of yN Ba(OH)_(2) were together sufficient to neutralise 100mL of 0.1N HCl. The ratio of V_(1) : V_(2) is 1:4 and x:y is 4:1 . What is the fraction of acid neutralised by barium hydroxide solution? |
|
Answer» Solution :Number of m. eq of HCl =M.eq ofNaOH +m.eq. of `BA(OH)_(2)``=100xx0.1=10=V_(1)x+V_(2)Y` By dividing with `V_(2)Y`. We GET `(10)/(V_(2)y)=(V_(1)x)/(V_(2)y)+1=(1)/(4)xx(4)/(1)+1=2` `V_(2)y=5` number of m.eq.of `Ba(OH)_(2))=5` FRACTION of acid neutralised by `Ba(OH)_(2)=(5)/(10)=(1)/(2)=0.5` |
|
| 44. |
Which one of the following is a path function? |
|
Answer» PRESSURE |
|
| 45. |
Which one of the following is calledInorganic graphite ? |
| Answer» SOLUTION :BN is calledinorganic graphitebecause its structureresembles with that ofgraphite. | |
| 46. |
To measure the pressure exerted by a trapped gas A, an open ended mercury manometer is used. After sparking the gas A, it dissociates as, A(g) to B(g) + 3C(g). If the pressure of A decreases to 0.9 atm, then (assume temperature to be constant, 300K) |
|
Answer» total pressure INCREASED to 1.3 atm |
|
| 47. |
Write electronic configuration of Z = 117 and Z = 120 . |
|
Answer» Solution :`Z = 117 : [Rn]^(86) 5f^(14)6d^(10) 7s^(2) 7p^(5)` `Z = 120 : "[Uuo]"^(118) 8S^(2)` |
|
| 48. |
What is meant by internal pressure of an ideal gas? |
|
Answer» <P> Solution :The pressure for one MOLE of a REAL GAS is given as: `P + a/(V^2)`. The term `a/(V^2)`is a measure of the attractive force of the molecules. It is called INTERNAL pressure or cohension pressure. It is of importance in connetion with inter-molecular forces. |
|