Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which of the following compound haszero dipole moment? |
|
Answer» CIS - But - 2 -ene |
|
| 2. |
Which of the following statements are not correct? |
|
Answer» The ionization ENERGY of a hydrogen-like species in its ground state is equal to the magnitude of energy of the orbit having N = 1 `Delta E = R(1//n_1^2)` b) The ionization energy increases in proportion to the square of positive charge in the nucleus as is evident from the expression `R =(2pi^2 m (Ze^2 //4pi epsi_0)^2)/(h^3 c)and Delta overset(~)E_("ioniz") = R` c) The correct expression is `DELTAP Delta X GE h// 4pi` d) For a multi-electron atom, the energy of an orbital depends on both principal and azimuthal quantum numbers. The larger the value of n+ l , larger the energy . For the same value of n + l , larger the value of n , larger the energy. |
|
| 4. |
Which law is used in the process of enriching the isotope of U23s from other isotopes? |
|
Answer» BOYLE's LAW |
|
| 5. |
Why beryllium is amphoteric in nature ? |
| Answer» Solution :Beryllium hydroxide is AMPHOTERIC in NATURE as it REACTS with acid and ALKALI both. | |
| 6. |
Which of the following is not a correct match ? |
|
Answer» `Cl^(-),P^(3-),Ar""`Isoelectronics |
|
| 7. |
What happens when group 14 elements react with oxygen and water ? |
|
Answer» Solution :(a) With oxgen : When heated in air they form their respective monoxides and DIOXIDES . ex : `"" Si + O_(2) overset (Delta) to Si O_(2) (s)` `"" 2C +O_(2) overset (Delta) to 2CO(g)` `"" C+O_(2) overset (Delta) to CO_(2) (g)` (b ) With water : C, Si, GE do not react with water . However tin reacts with steam to form stannic oxideliberating hydrogen gas . `Sn + underset ("steam") (2H_(2) O) overset (Delta) to SnO_(2)+2H_(2(g))` |
|
| 8. |
Which of the following is used as food? |
|
Answer» Potash |
|
| 9. |
The specific charge of photon is 9.6xx10^7 C kg^(-1) then for an alpha-particle it will be |
|
Answer» `38.4xx10^7 " C KG"^(-1)` |
|
| 10. |
Which type of overlapping result in the formation of (i) sigma bond (ii) pi bond. |
| Answer» SOLUTION :P-P PARALLEL/ sideway/lateral OVERLAPPING. | |
| 11. |
Which of the following technique is most suitable for the purification of cyclohexanone from a mixture containing benzoic acid, isoamyl alcohol, cyclohexane, and cyclohexanone? |
|
Answer» Crystallization |
|
| 13. |
What is the electrophile in sulphonation reaction of benzene ? And how it is obtained ? |
|
Answer» Solution :`SO_(3)` is electrophilic in NATURE which is obtaied by following manner. `2H_(2)SO_(4) hArr H_(3)O^(+) + HSO_(4)^(-) + SO_(3)` |
|
| 14. |
Which of the following is most stable |
| Answer» Answer :A | |
| 15. |
The total number of lone pairs of electrons in N_(2)O_(3) is |
|
Answer» |
|
| 16. |
Two oxides of a certain metal were separately heated in hydrogen till water is produced. It was observed that 1 gm of each oxide gave 0.125gm and 0.2263 grams of water respectively. This law states that it illustrate law of |
|
Answer» Definite proportions |
|
| 17. |
Whatis polymerisation explainwith suitable example |
Answer» SOLUTION :A POLYMERIS a largemoleculeformedby thebombinationof largenumberof smallmolecules .Thisprocessis knownpolymerisation.Afewexamplesare
|
|
| 18. |
What is the oxidation number of the underlined atoms in the following ? (a)CO_(2) ,(b) SOO_(2), (c ) Cr_(2)O_(7)^(2-), (d)CrO_(4)^(2-), (e )H_(4)P_(2)O_(7) ,(f) NaH_(2)PO ,(h) H_(2)S_(2)O_(7) |
|
Answer» |
|
| 19. |
What type of crystal defect is produced when sodium chloride is doped withMgCl_(2) ? |
| Answer» SOLUTION :It is called impuirty defect. A cation VACANCY is produced.A substitutional solid solution is formed (because `2NA^(+)` ions are replaced by one `SR^(2+)` ion in the lattice site). | |
| 20. |
Which hydride of group 15 is unstable ? |
|
Answer» `PH_3` |
|
| 21. |
Which of the following reactions do not represent the major product of given Birch reductions? |
|
Answer» (i), (III), (iv) 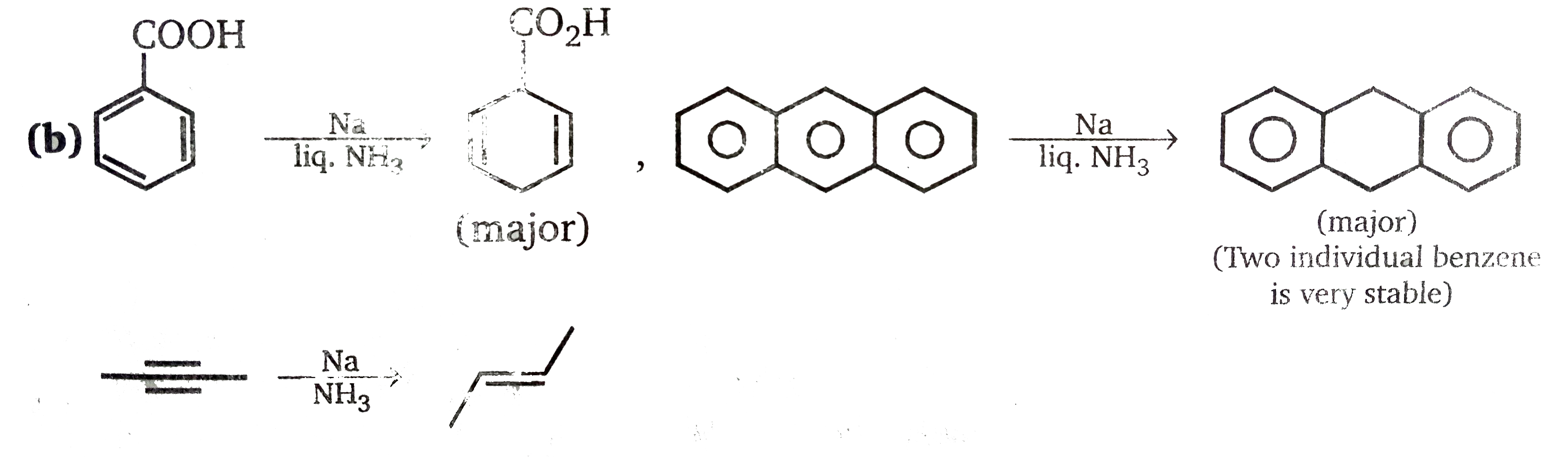
|
|
| 22. |
When CO_(2) is passed into aqueous solution of Na_(2)CO_(3) the following is formed |
| Answer» Answer :B | |
| 23. |
What shell would be the first to have a g-subshell ? How many orbitals will be possible in a g-subshell ? |
|
Answer» Solution :For g-subshell, `l = 4`. As l = 0 to N -1, hence to have `l = 4`, MINIMUM value of `n = 5`, i.e., 5th shell. For `l = 4, m = -4, -3, -2, -1, 0, +1, +2, +3, +4`, i.e., 9 values which means 9 orbitals. |
|
| 24. |
What is the common name of 2-butanone? |
|
Answer» Acetone |
|
| 25. |
Which of the following hydrocarbon cannot have cyclic type ? |
|
Answer» Alkane |
|
| 26. |
Whch element present as impurity in artifical fertilizers ? |
|
Answer» As |
|
| 27. |
Which of the following is a chemical change ? |
|
Answer» A. Souring of milk |
|
| 28. |
Which of the following sentence is incorrect? |
|
Answer» The solubiity of a solute in a liquid solvent decrease with increase in tempreature |
|
| 29. |
Name a metal found abundantly in the earth'scrust. |
|
Answer» Aluminium |
|
| 30. |
What is the change in the value of entropy when ice melts to give water. |
|
Answer» |
|
| 31. |
What will be the pH of the resulting solution if to a 100 ml of HCl solution of pH = 1.0, 900 ml of distilled water is added ? |
|
Answer» Now, `[H^(+)] = 10^(-1)//10 = 10^(-2)M :.pH = 2` |
|
| 32. |
What are viable and non-viable particulates |
| Answer» Solution :Viable particulates are small size living organisms such as bacteria, fungi, moulds, algae, etc. Non-viable particulates are formed by disintegration of large size MATERIALS or CONDENSATION of small size PARTICLES or droplets, e.g., MIST, smoke, fume and dust | |
| 33. |
Write IUPAC name and write condensed structure, dash structure line formula of problem |
Answer» SOLUTION : 
|
|
| 34. |
When I mole of pure ethyl alcohol (C_(2)H_(5)OH) is mixed with 1 mole of acetic acid at 25^(@)C with one lit volume, the equilibrium mixture contains 2//3 mole each of ester and water. CH_(2)OH_((l)) +CH_(3)COOH_((l)) harr CH_(3)COOC_(2)H_(5(l))+H_(2)O_((l)) The DeltaG^(@) for the reaction at 298 K is: |
|
Answer» 3435J  `x=2//3` `K=((2)/(3) xx (2)/(3))/((1)/(3) xx (1)/(3))=4` `DELTA G^(@)= -RT ln K IMPLIES Delta G^(@) = -3435J` |
|
| 35. |
When a sample of NO_(2) is placed in a container, the equilibrium is rapidly established. If the equilibriummixture is of a darker colour at high temperalures and at low pressures, which of the following statements about the reaction is true ?2NO_(2) (g ) - Q hArr N_(2) O_(4) (g) |
|
Answer» Formation of `N_2O_4` from `NO_2` is exothermic and `N_2O_4` is darker in COLOUR than `NO_2` |
|
| 36. |
When one mole of gaseous methane s burnt in excess of gaseous oxygen, 890 kJ of heat is evolved in addition to the formation of gaseous CO_(2) and liquid water. (a) Write the thermochemical equation for this reaction. (b) Calculate the amount of heat evolved when 10 grams of methane is burnt. |
|
Answer» SOLUTION :(a) `CH_(4)(g) + 2O_(2)(g) rarr CO_(2)(g) + 2H_(2)O(l)Delta H = -890 KJ` (b) From the equation `1 " mole of "CH_(4) = 16g = 890 kJ " heat"` `16g CH_(4)` RELEASES `= 890 kJ` heat `10 g CH_(4)` releases `= (890 kJ xx 10 g)/(16g) = 556 kJ` Amount of heat liberated for the combustion of 10 g `CH_(4) = 556 kJ`. |
|
| 37. |
Which one of the following molecule has complete octet? |
| Answer» Solution :`C Cl_(4)` | |
| 38. |
Which one of the following molecules contains both ionic and covalent bonds? |
|
Answer» `CH_2Cl_2` |
|
| 39. |
What is p-block element ? Discuss gases element and metallic properties of p-block elements. |
|
Answer» Solution :General electronic CONFIGURATION of p-block element is `ns^(2)sp^(1) " to " ns^(2) np^(6)`. NOBLE Gases : The p-block elements comprise those belonging to group 13 to 18 and these together with the s-block elements are called the representative elements or main group elements. The outermost electronic configuration varies from `ns^(2)np^(1) " to " ns^(2)np^(6)` in each period. Halogens Elements : The noble gases thus exhibit very low chemical reactivity. Preceding the noble gas family are two chemically important groups of non-metals. They are the halogens (Group-17) and the chalcogens (Group-16). Chalcogens Elements : `16^(th)` group of elements are known as chalcogens group. Electron gain enthalpy of these group is very high. Elements of group 16 accept 2 electrons and form octate state LIKE noble gas. O, S, Se, Te elements present in 16. group. Metalic and non-metalic Properties : When we go left to RIGHT in p-block non-metalic property increases. When we go for top to BOTTOM metalic property increases. |
|
| 40. |
Why carbon differs from rest of the members of its group ? Explain. |
|
Answer» Solution :Like first member of other groups, carbon also DIFFERS from rest of the members of its group. It is due to its smaller size, higher electro negativity, higher ionisation enthalpy and unavailability of d-orbitals. In carbon, only s and p-orbitals are available for bonding and therefore, it can accommodate only four pairs of electrons around it. This would limit the MAXIMUM covalence to four whereas other members can expand their covalence due to the presence of d-orbitals. Carbon also has unique ability to form pt-pt multiple bonds with itself and with other atoms of small size and high electronegativity. Few examples of multiple bonding are:C=C, C`-=`C , C=O , C=S , C`-=`N. Heavier ELEMENTS do not form pn-pn bonds because their atomic orbitals are too large and diffuse to have effective overlapping. Carbon atoms have the tendency to link with ONE another through covalent bonds to form chains and rings. This property is called catenation. This is because C-C bonds are very strong. Down the group the size increases and electro negativity decreases and there by, tendency to show catenation decreases. This can be clearly seen from bond enthalpies values. The order of catenation is C `GT gt` Si `gt` Ge `approx` Sn. Lead does not show catenation. Due to property of catenation and `ppi-ppi`bond formation, carbon is able to show allotropic forms. 
|
|
| 41. |
The thermodynamic property that measures the extent of molecular disorder is called entropy. The direction of a spontaneous process for which the energy is constant is always the one that increases the molecular disorder. Entropy change of phase transformation can be calculated using Trouton's formula (DeltaS= (DeltaH)/(T)). In the reversible adiabatic process, however, DeltaS will be zero. The rise in temperature in isobaric and isochoric conditions is found to increase the randomness or entropy of the system. DeltaS= 2.303 C log (T_(1)//T_(2)), (C = C_(P) or C_(V)) If water in an insulated vessel at -10^(@)C, suddenly freezes, the entropy change of the system will be |
|
Answer» zero |
|
| 42. |
Which one of the alkaline earth metal carbonates is thermally the most stable ? (a) MgCO_(3) (b) CaCO_(3) (c) SrCO_(3) (d) BaCO_(3) |
|
Answer» Solution :THERMAL stability increases with the increase in the size of the cation present in the carbonate. The INCREASING order of the cationic size of the GIVEN alkaline earth metals is `Mg lt Ca lt Sr lt Ba`. HENCE, the increasing order of the thermal stability of the given alkaline earth METAL carbonates is, `MgCO_(3) lt CaCO_(3) lt SrCO_(3) lt BaCO_(3)` |
|
| 44. |
Write reactions to justify amphotericnature ofaluminium. |
|
Answer» Solution :It DISSOLVE both in ACIDS and alkalines evolvingdihydrogen. `2Al(s) + 3H_(2)SO_(4)(aq) rarr Al_(2)(SO_(4))_(3) (aq) + 3H_(2)(G)` `2Al(s) + 2NaOH (aq) + 6H_(2)O (L) rarr underset(" Sod. Tetrahydroxoaluminate (III) ")(2 Na^(+)[Al(OH)_(4)]^(-)(aq))+ 3H_(2) (g)` |
|
| 45. |
What [H_(3)O^(+)] must be maintained in a saturated H_(2)S solution to precipitatePb^(2+) but not Zn^(2+) from [K_(sp)H_(2)S=1.1xx10^(-22) and K_(sp)ZnS=1.0xx0^(-21)] |
|
Answer» Solution : `K_(sp) ` for `ZnA = 10^(-21) and [Zn^(2+)] = 0.01 M = 10^(-2) M` But `K_(sp) [ZNS) = [ Zn^(2+) ] [S^(-2)], i.e., 10^(-21) = [10^(-2)] [S^(2-)]or [S^(2-) ] = 10^(-19) M` Thus, to prevent precipitation of `Zn^(2+)` ions, `[S^(2-)]` MUST be less than `10^(19)` M. Futher, `H_(2)S OVERSET(2H_(2)O)RARR 2 H_(3)O^(+) + S^(2-)` `:.K_(sp) (H_(2)S) = [ 2 H_(3)O^(+)]^(2) [S^(2-)]` `1.1xx10^(-22)=[2H_(3)O^(+)]^(2)xx10^(-19) or [2 H_(3)O^(+)]^(2)=1.1xx10^(-3) = 11xx10^(-4)` or `[2H_(3)O^(+)] = 3.32xx10^(-2) M or [H_(3)O^(+) ] = 1.66xx10^(-2) M` |
|
| 46. |
Write the product obtainedon action of oxygen on diborane ? |
| Answer» | |
| 47. |
What volume gain by gas at STP ? |
| Answer» SOLUTION :At STP there are no attraction FORCES present between MOLECULES of GASES, so they expand and resists space then volume gain of container. | |
| 48. |
What is standard boiling point of H_(2)O ? |
| Answer» Answer :A | |
| 49. |
Whatwouldbe theatomicnumberof the next(i)alkali metal (ii)alkalineearthmetal (ii)halogenand (iv)inertgas Ifdiscoveredin future ? |
|
Answer» Solution :(i) Thenextalkali metalifdiscovered willhave tobeplacedin theeighth period and henceitsouterelectronicconfigurationwillbe `8s^(1)` . Thereforeitsatomicnumberwillbe(118 +1)= 119. (ii)Similarly , the nextalkalineearthmetalifdiscovered willhave `8s^(2)` as itsouterelectronicconfigurationand henceitsatomicnumberwill be(118 + 2) =120. (III) thenexthalogenif discoveredwill have`7s^(2) 7p^(5)` as itsouterelectronicconfiguration.Sincethefillingof 7p-orbitals willbeginafterfillingof 6d- orbitals in accordancewithaufbauprinciplewe canaccommodate only 112elements thereforethe atomicnumbero the nexthalogenif discoveredwill be(112 + 5) =117 . (iv) Ina similarwaywe caneasilyexplainthat the nextinertgas , if discoveredwill have`7s^(2) 7p^(6) ` asouter electronicconfigurationand itsatomicnumberwill be(112 +6) =118. |
|