Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
What is the product formed in the following reaction. |
|
Answer»
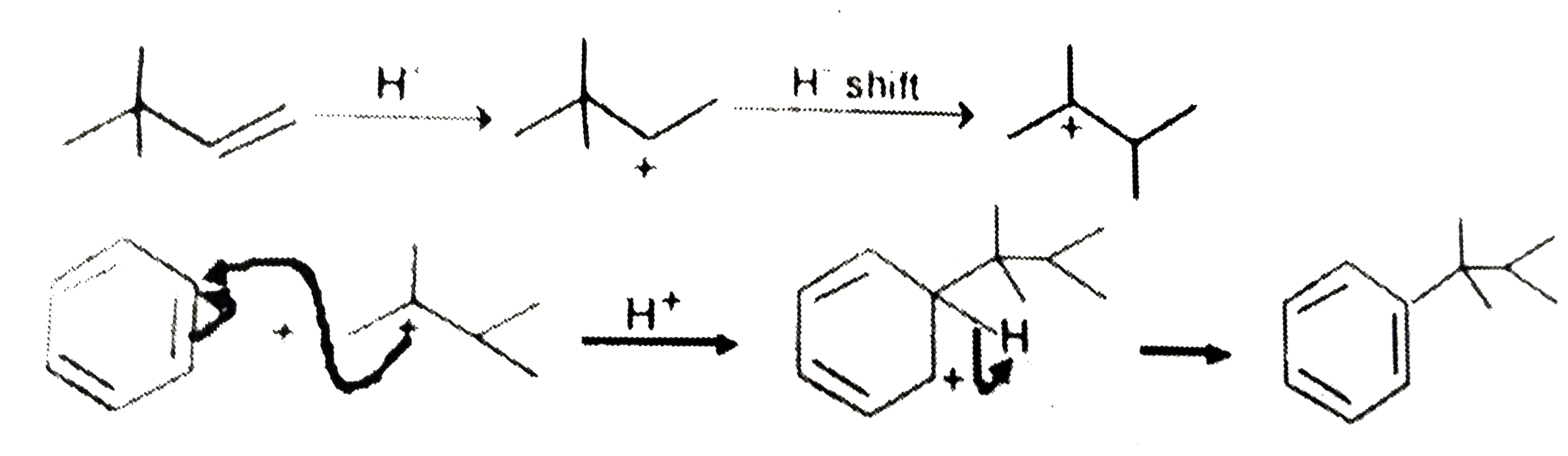
|
|
| 2. |
Which of the following statements about polar stratosphere clouds (PSCs) is not correct? |
|
Answer» `PSCs` do not react with CHLORINE nitrate and `HCl` |
|
| 3. |
Two solutions of 0.1M cr_(2) O_(7)^(2-)(aq.) and 0.1M MnI_(4)^(-) (aq.) are to be used to titatre (titrating solution) be required for a given solution of fe^(2+)(aq.) (b) If a given titration requires 24.50 mL of 0.100M Cr_(2)O_(7)^(2-) (aq.),how many mL of 0.100M MnO_(4)^(-) (aq.)would have been required if ithad been used instead? |
|
Answer» |
|
| 4. |
Theunpairedelectronsin AIand sI arepresentin 3porbital. Whichelectron willexperiencemoreeffectivenuclearchargefrom thenucleus ? |
|
Answer» SOLUTION :Theelectronof Si has moreeffectivenuclear CHARGE. Electroncnfigurationof AI(z=13) Ne `3s^(2) 3p^(1)` as bothhas samevalenceshell(z=14) `Ne^(10) 3s^(2) 3p^(2)` so nuclearchargeis more.sothere ismoreeffectivenuclear chargean si. |
|
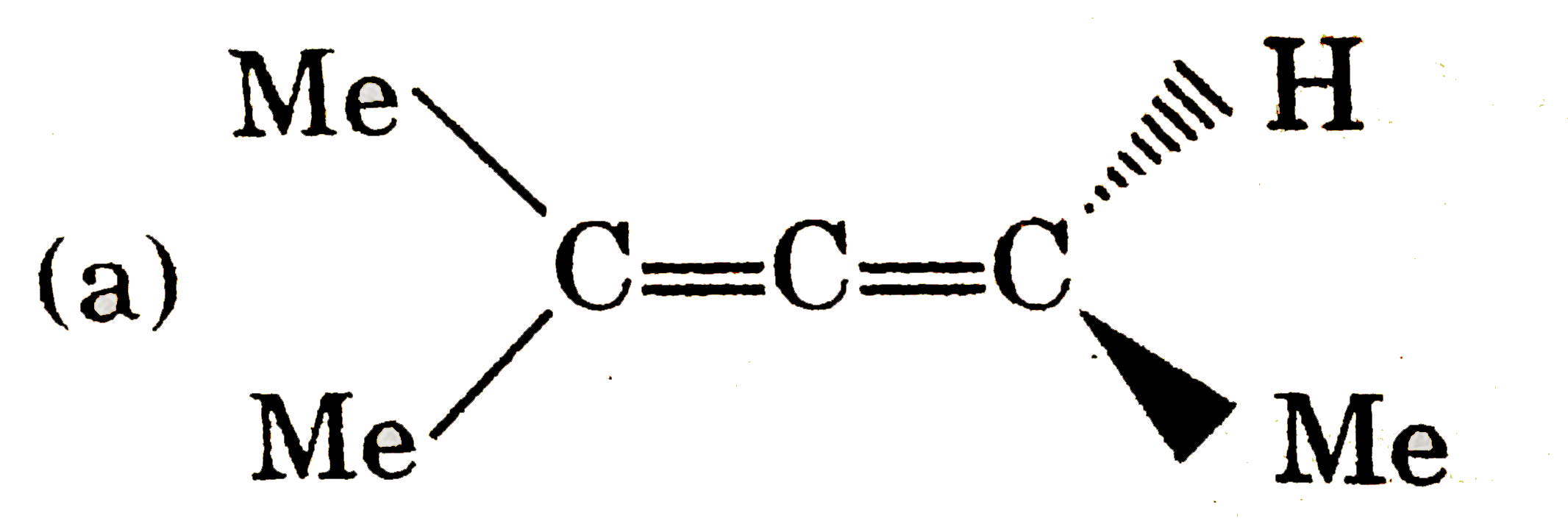
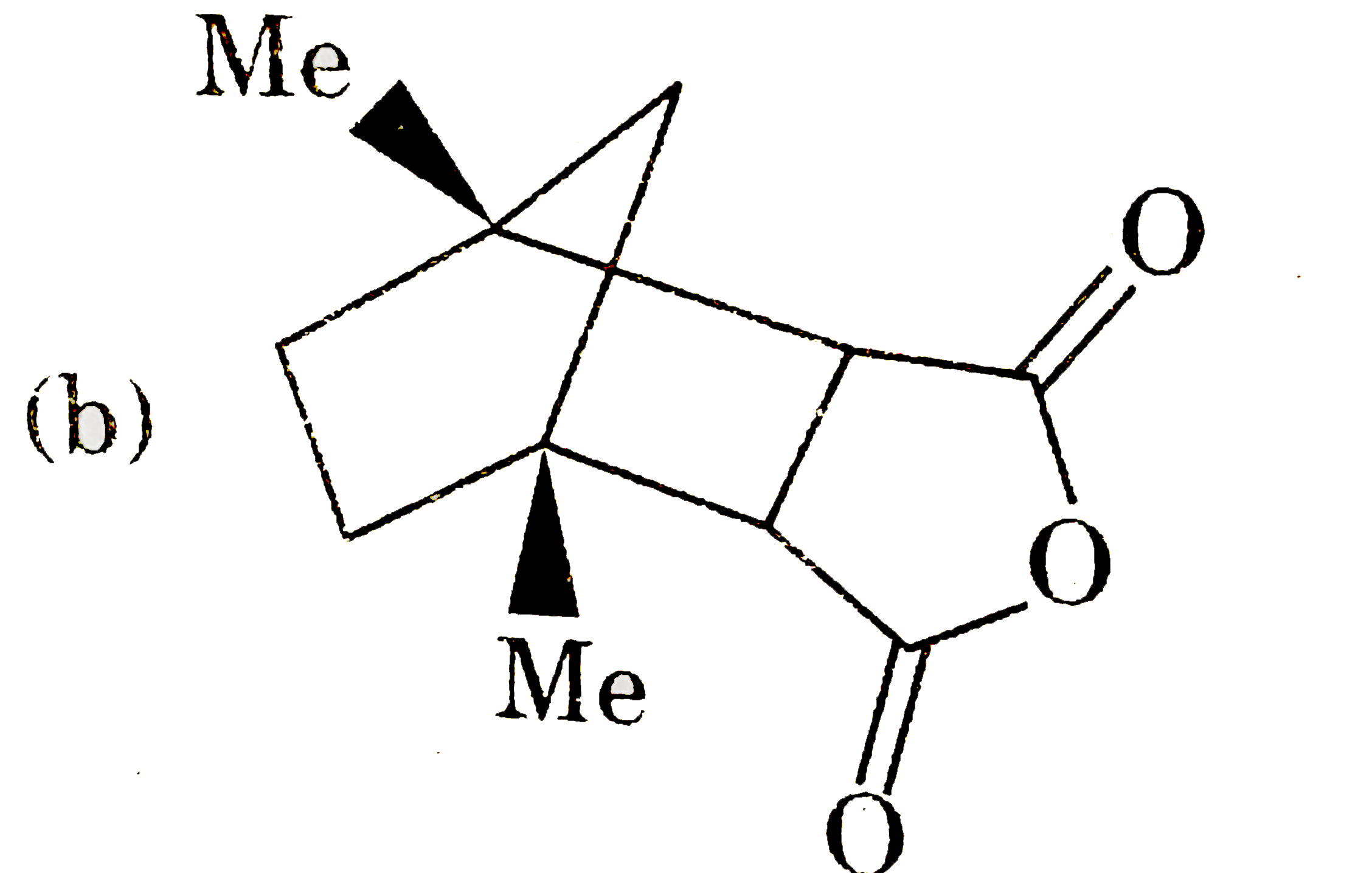
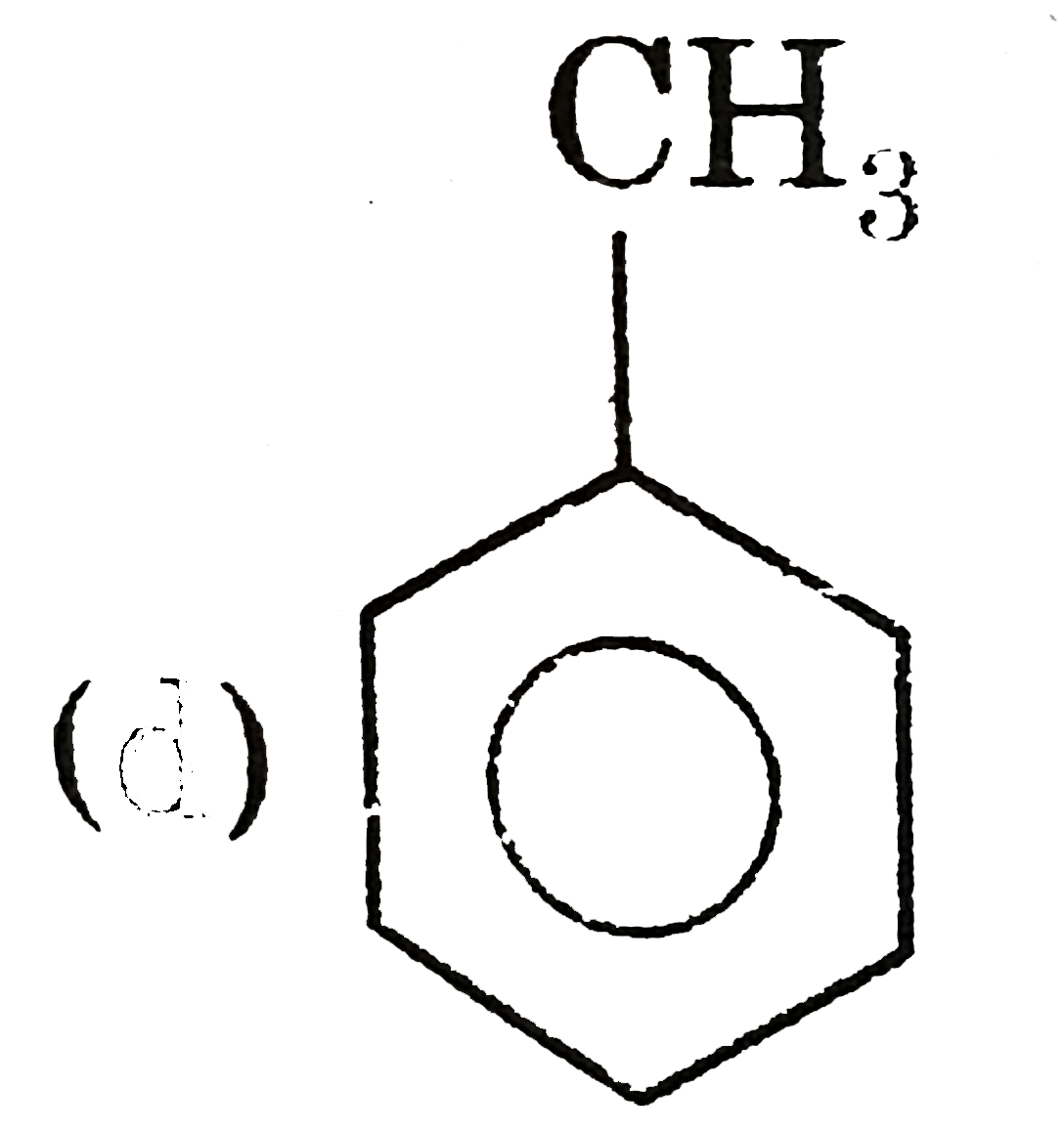
| 5. |
Which of the following pair of compounds exhibit tautomerisim? |
|
Answer» ETHANAL and vinyl alcohol Ethanal has `alpha`-hydrogen to CARBONYL group and it has tautomer vinyl alcohol. (B) , (C ), (D) do not have `alpha`-hydrogen to carbonyl group and their enol from |
|
| 6. |
underset("Sucrose")(C_(12)H_(22)O_(11))+H_(2)Ooverset(dil." "H_(2)SO_(4))tounderset("Fructose")(C_(6)H_(12)O_(6)(aq))+underset("Glucose")(C_(6)H_(12)O_(6)(aq)) in this reaction, dilute H_(2)SO_(4) is called. |
|
Answer» Homogenous catalysis |
|
| 7. |
The standard enthalpy of formation of hypothetical MgCl is -125kJ mol^(-1) and for MgCl_(2) is -642 kJ mol^(-1). What is the enthalpy of the disproportionation of MgCl ? |
|
Answer» `-492 KJ mol^(-1)` (ii) `Mg_((s)) + 1//2Cl_(2(g)) rarr MgCl_((s)) , DELTA H_(2) = - 125` (iii) `2MgCl rarr MgCl_(2) + Mg, Delta H`= ? `Delta H = Delta H_(1)- Delta H_(2) = - 392 kJ mol^(-1)` |
|
| 8. |
Which reducing agents of the following can be used for the following transformation ? |
|
Answer» `CH_(3) - CH = CH.COOH rarr CH_(3)CH = CH.CH_(2)OH` |
|
| 9. |
What will be de-Broglie wavelength of an electron moving with a velocity of 1.2xx10^5 "ms"^(-1) |
|
Answer» `6.071xx10^(-9)` `lambda=h/"mv"=(6.62xx10^(-34))/(9.1xx10^(-31)xx1.2xx10^5)` `lambda=6.071xx10^(-9)`m |
|
| 10. |
Write the expected formulas for the hydrides of 4^(th) period elements.What is the trend in the formulas? In what way the first two numbers of the series different from the others ? |
|
Answer» Solution :The expected formulas for the hydrides of 4th PERIOD elements MH. (electron PRECISE). M,H (electron deficient) and `MH_(3)` (electron rich). The trend in formula is (i) Electron precise hydrides `-CH_(4)`,`C_(2)H_(6)`,`SiH_(4)`, `GėH_(4)`, (ii) Electron deficient hydrides-`B_(2)H_(6)` (iii) Electron rich hydrides -`NH_(3)`,`H_(2)O`. The first two members of the series KH, Cal, are ionic hydrides WHEREAS the other members of the series `CH_(4)`, `C_(2)H_(6)`, `SiH_(4)`, `B_(2)H_(6)`, `NH_(3)` are covalent hydrides. |
|
| 11. |
The Rubin number with was proposed by Ostwald as an alternative to the Gold number in order to measure the protective efficiency of a lyophillic colloid may be difined as the |
|
Answer» mass in milligrams of a colloid per 100 c.c. of solution which just prevent the colour change of STANDARD sol of dye Congo-Rubin FORM red to violet when 0.16 G eq. KCl is added to it. |
|
| 12. |
What is standard boiling of the liquid. |
| Answer» Solution :Temperature at which vapour PRESSURE of a liquid becomes EQUAL to one bar `(10^(-5)` PA) is CALLED standard biling point of liquid. | |
| 13. |
Which of the following is used catalyst in preparation of PVC ? |
| Answer» Answer :B | |
| 14. |
The reducing power of a metal depends on various factors. Suggest the factor which makes Li, the strongest reducing agent in aqueous solution. |
|
Answer» SUBLIMATION enthalpy |
|
| 15. |
x millimole of KlO_(3) react completely with y millimole of KI to give I_(2) quantitatively. It z millimole of hypo are required for complete titration against the I_(2) product, then |
|
Answer» `Z=6x` m eq `KIO_(3)` = m eq KI = m eq `I_(2)` `x xx5=yxx1=nxx(5)/(3)` `I_(2)+Na_(2)S_(2)O_(3)rarrNa_(2)S_(4)O_(6)+NaI` m eq `I_(2)` = m eq `Na_(2)S_(2)O_(3)` `x xx2=zxx1` `implies x=(z)/(2)implies5x=y=(5z)/(6)`. |
|
| 16. |
When metal 'M' is treated with NaOH, a whitegelatinousprecipitate 'X' is obtainedwhich issolublein excess of NaOH. Compound 'X' whenheatedstronglygives an oxide which is usedin chromatography as an adsorbent.Then metal 'M' |
|
Answer» Zn `Al(OH)_(3) OVERSET(NaOH)rarr NaAl(OH)_(4)` `2Al(OH)_(3) overset(Delta)rarr Al_(2)O_(3) + 3H_(2)O` `Al_(2)O_(3)` is used as an adsorbent in chromatography. |
|
| 17. |
When hydrogen peroxide is shaken with an acidified solution of potassium dichromate in presence of ether, the ethereal layer turns blue due to the formation of |
|
Answer» `Cr_(2)O_(3)` |
|
| 20. |
To which orbit the electron in the hydrogen atom will jump after absorbing 1.94 xx 10^(-18)J of energy ? |
|
Answer» Solution :Energy of electron in the nth orbit of H-atom is GIVEN by: `E_(n) = - (21.8 xx 10^(-19))/(n^(2)) J:. E_(1) = - 21.8 xx 10^(-19) J` (`:' n = 1` for ground STATE of H-atoms) After absorbing `1.94 xx 10^(-8) J` of energy, the energy of the electron will be `= -21.8 xx 10^(-19) + 1.94 xx 10^(-18) J = - 2.4 xx 10^(-19) J` Thus, `- (21.8 xx 10^(-19))/(n^(2)) = - 2.4 xx 10^(-19) or n^(2) = (21.8)/(2.4) = 9 or n = 3` Thus, the electron will jump to the third orbit. |
|
| 22. |
What is the maximum no. of emission lines obtained when the excited electrons of a hydrogen atom in n=5 drop to a ground state |
|
Answer» 10 `therefore ((5-1)(5-1+1))/2 =20/2` =10 lines |
|
| 23. |
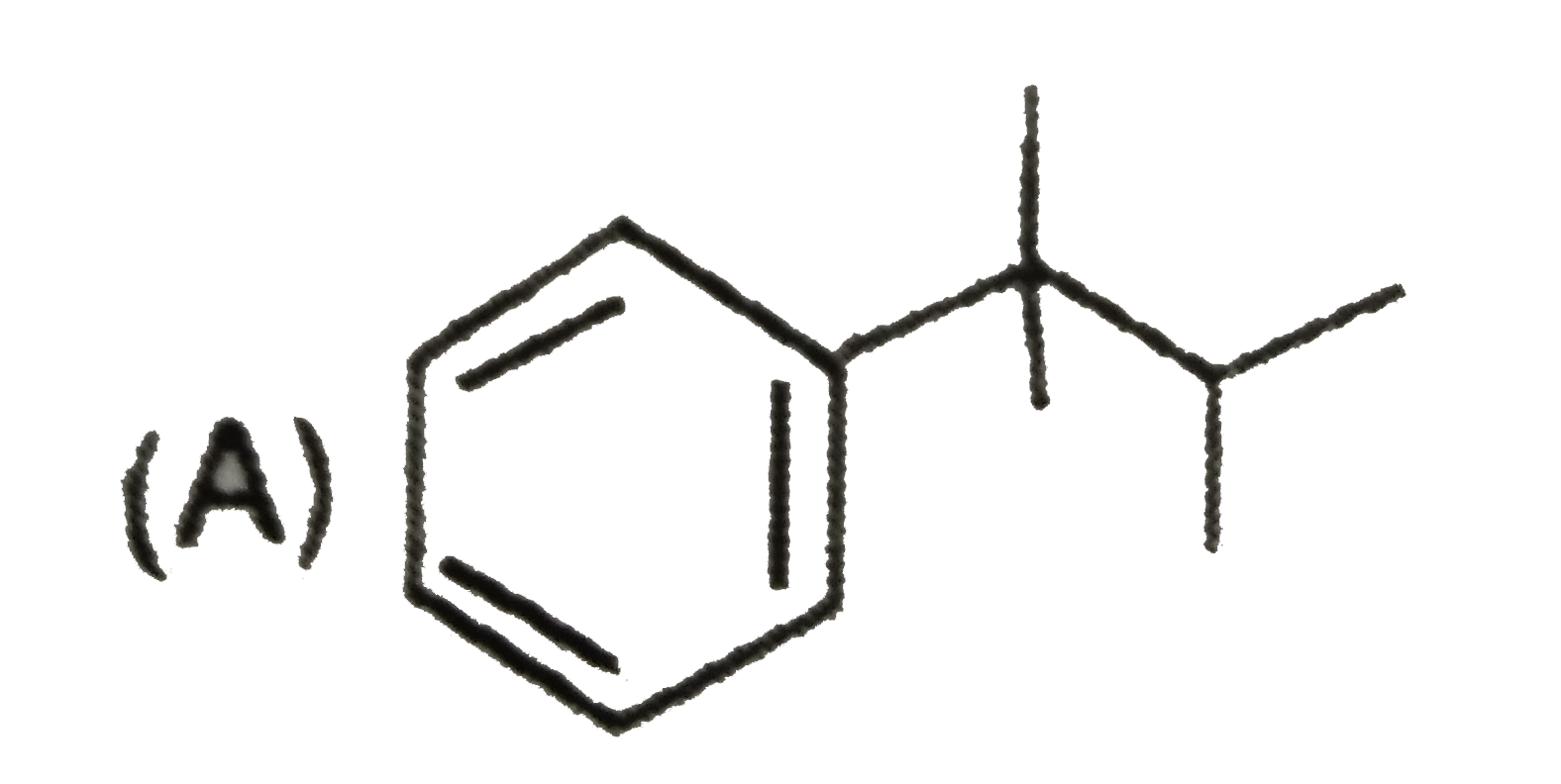
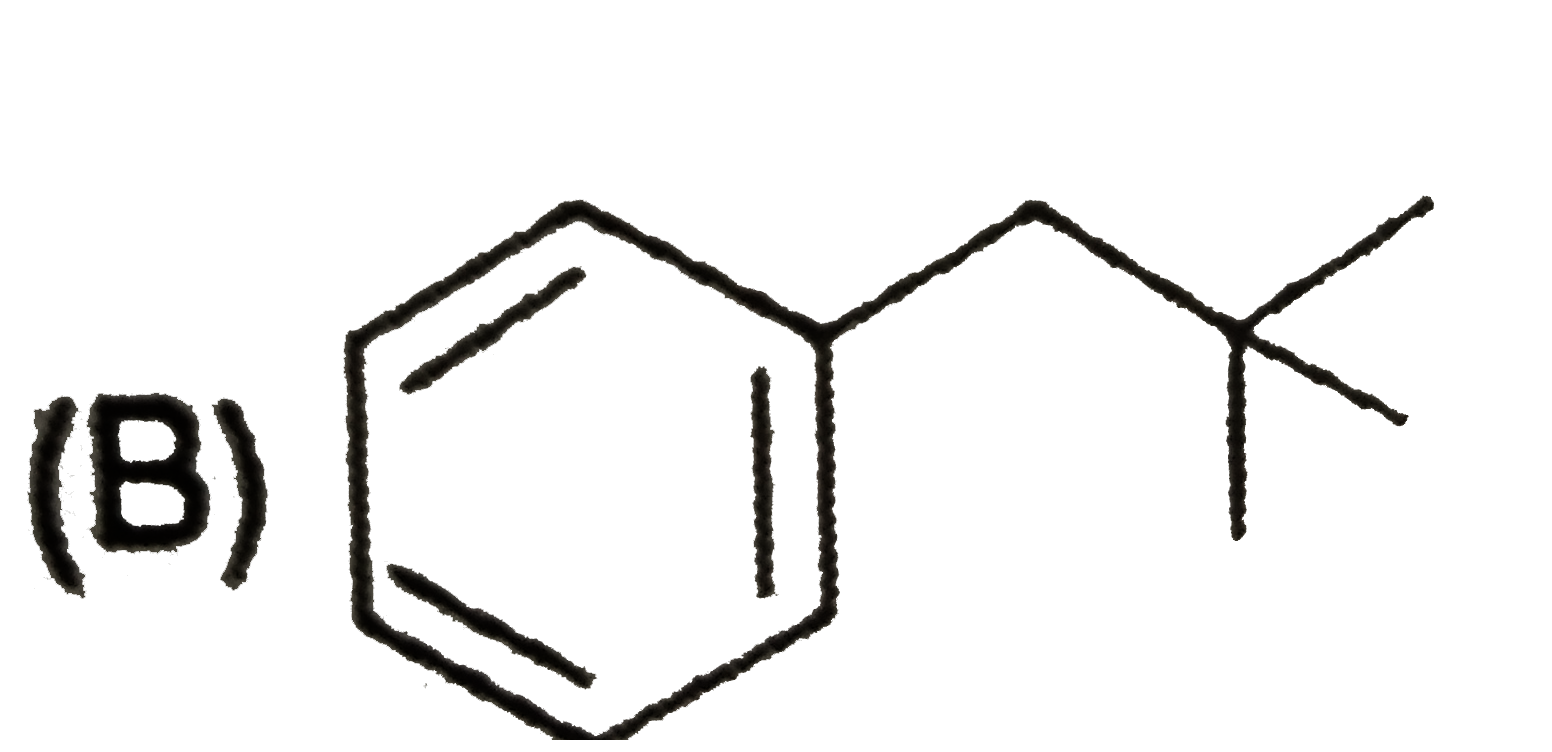
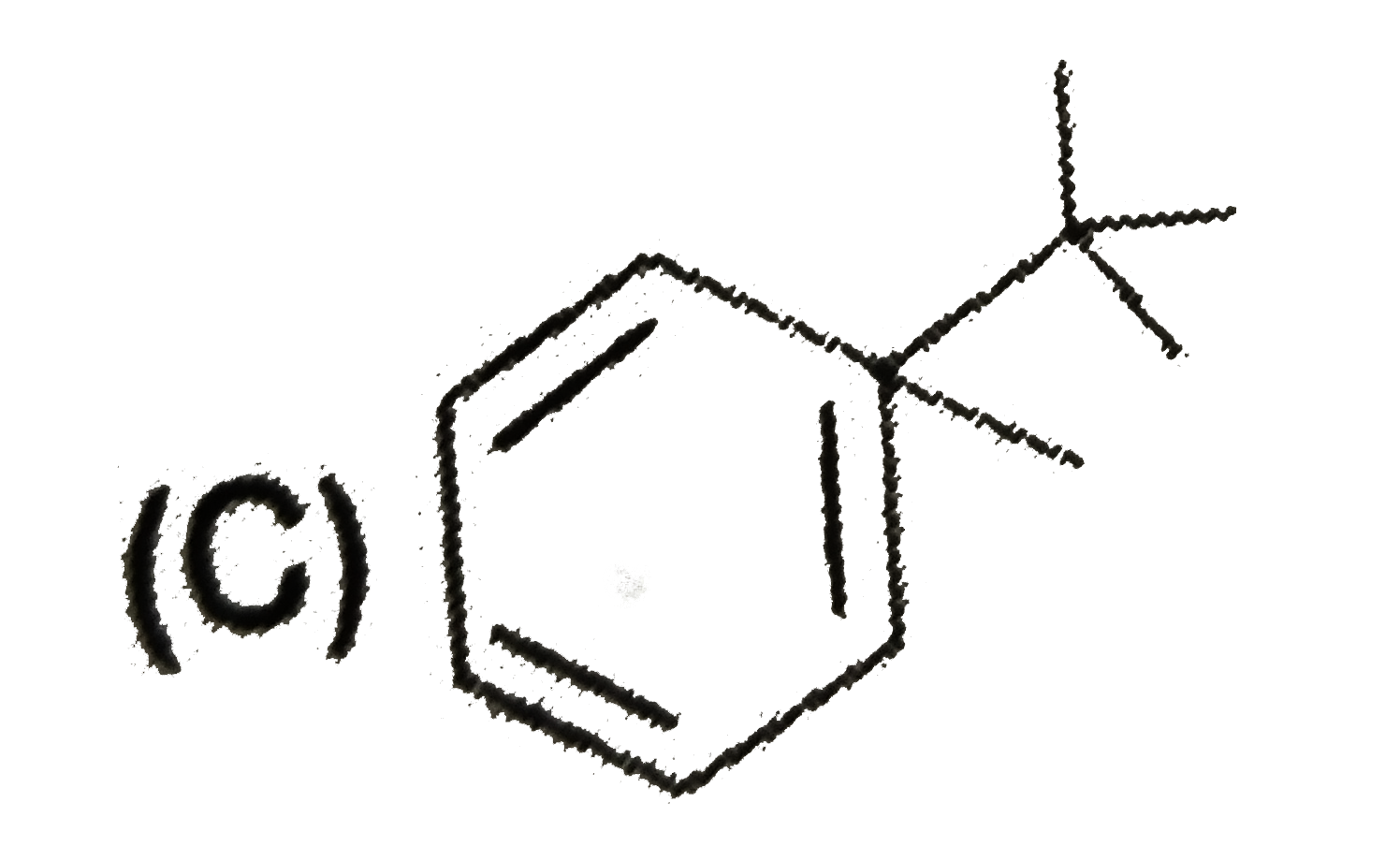
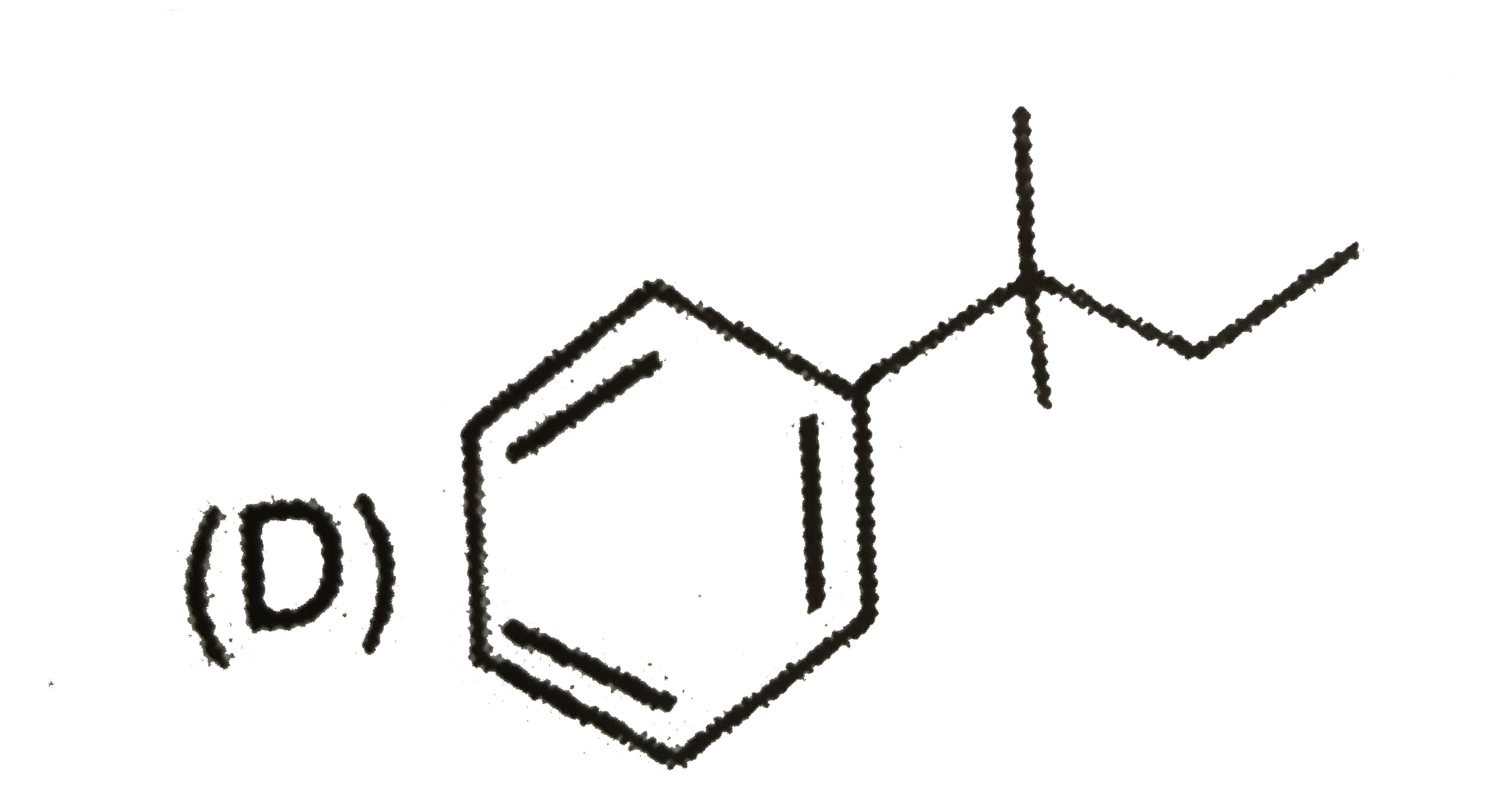
Which of the following compund is achiral |
|
Answer»
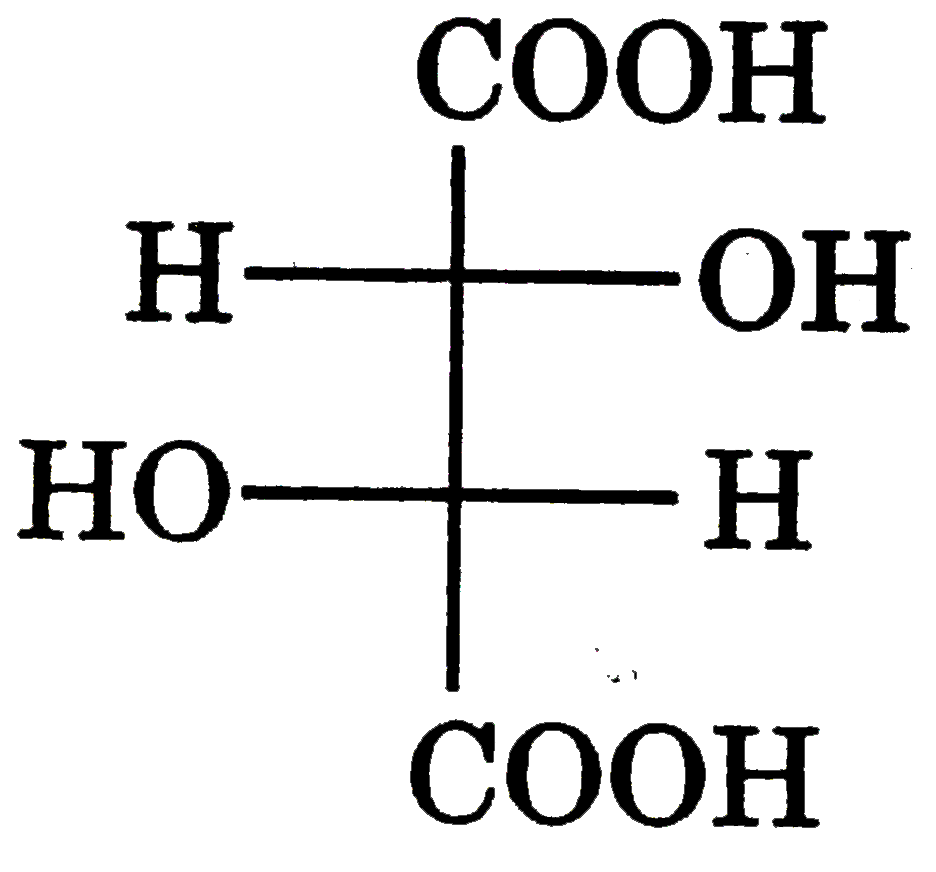 No plane of symmetry. No plane of symmetry.
|
|
| 24. |
Which reactant is used to prepare trans alkene from alkyne ? |
| Answer» SOLUTION :SODIUM METAL in LIQUID AMMONIA. | |
| 25. |
What type of defect can arise when a solid is heated ?Which physical property is affected by it and in what way ? |
| Answer» SOLUTION :When a solid is heated, vacancy defect is produced in the crystal. This is because on heating , some ATOMS or ions LEAVE the alttice site completely, i.e, some lattice sites become vacant. As a RESULT of this defect, the DENSITY of the substance because some atoms/ions leave the crystal completely . | |
| 26. |
What happens whensodium peroxide dissolves in water? |
|
Answer» Solution :When sodium peroxide is DISSOLVED in water, it is READILY hydrolysed to form sodium hydroxide and water. The chemical EQUATION INVOLVED in the reaction is : `Na_(2)O_(2(s)) +2H_(2)O_((l)) to 2NaOH_((AQ)) +H_(2)O_(2(aq))` |
|
| 27. |
Which of the following is the correct orderr of acidity of carboxylic acids? (i) Cl_(3) C C OOH gt Cl_(2)CHCOOH gt ClCH_(2)COOH (ii) CH_(3)CH_(2)COOH gt (CH_(3))_(2)CHCOOH gt (CH_(3))_(3)C COOH (iii) F_(2)CHCOOH gt FCH_(2)COOH gt ClCH_(2)COOH |
|
Answer» (i) and (II) |
|
| 28. |
Which information give following figure ? |
|
Answer» Solution :Information-1: A and B are reactants. Its concentration is DECREASE with time and after it constant A + B `to`product. Information-2: C and D are PRODUCTS. Initially its concentration is zero. The amount of C and D increase with time and after some time it become constant. Reactions `to`(C + D) (products) Information-3: After some time RATE of FORWARD reaction become constant and concentration of A, B, C, D remain constant A + B `hArr` C+ D. |
|
| 29. |
When organic substance undergoes degradation through bacteria , the hydrocarbon formed is |
|
Answer» `C_2H_4` |
|
| 30. |
Which of the following statement(s) is/are true? |
|
Answer» The ratio of rms speed to AVERAGE speed is independent of TEMPERATURE |
|
| 31. |
The relationship between the values of osmotic pressure of 0.1Msolutions ofKNO_3 (P_1)andCH_3 COOH(P_2)is |
|
Answer» ` P_1 GT P_2` |
|
| 32. |
Which ofthe following does not represent a type of crystal system |
|
Answer» Triclinic |
|
| 33. |
When a metal rod M is dipped into an aqueous colourless concentrated solution of compound N, the solution turns light blue. Addition of aqueous NaCI to the blue solution gives a white precipitate O. Addition of aqueous NH_(3) dissolves O and gives an intense blue solution. The metal rod M is: |
| Answer» Answer :B | |
| 34. |
When a metal rod M is dipped into an aqueous colourless concentrated solution of compound N, the solution turns light blue. Addition of aqueous NaCI to the blue solution gives a white precipitate O. Addition of aqueous NH_(3) dissolves O and gives an intense blue solution. The final solution contains: |
|
Answer» `[Pb(NH_(3))_(4)]^(2+)` and `[CoCI_(4)]^(2-)` |
|
| 35. |
When a metal rod M is dipped into an aqueous colourless concentrated solution of compound N, the solution turns light blue. Addition of aqueous NaCI to the blue solution gives a white precipitate O. Addition of aqueous NH_(3) dissolves O and gives an intense blue solution. The compound N is: |
|
Answer» `AgNO_(3)` |
|
| 36. |
Which gases are responsible for greenhouse effect? List some of them. |
| Answer» Solution :`CO_2` is mainly RESPONSIBLE for greenhouse effect. Other greenhouse gases are methane, NITROUS oxide, water VAPOURS, CFCS and ozone. | |
| 37. |
Use molecular orbitals theory to explaiin why the Be_(2) molecular does not exist . |
|
Answer» Solution :E.C. of `._(4) Be = 1s^(2) 2s^(2)` M.O.E.C. of ` Be_(2) = SIGMA _(1s)^(2) sigma_(1s)^(2) sigma_(2s)^(2) sigma_(2s)^(2)` `THEREFORE ` Bond ORDER = `(1)/(2) (4-4) = 0 `. Hence , `Be_(2)` does not exist . |
|
| 38. |
What is oxidation number of N in HO_(4) ? |
|
Answer» Solution :By conventinal method O.N of N is : +1+4(-2)+x=0 or x =+7 this is impossible because N cannot have an O.N of more than + SINCE it has only 5 electrons in valence shell Therefore O.N of N in `HNO_(4)` must be CALCULATED by chemical bonding method the structure of `HNO_(4)`is since it contins a peroxide linkage therefore each of the two oxygen atmos of the peroxide bonds has an O.N of -1 ON of -2 thus if x is the o.n of N then +1-1-1+x-2-2=0 or x=+5 |
|
| 39. |
Which one of the following statement(s) is/are incorrect ? |
|
Answer» `KHCO_(3)` is less soluble than `NaHCO_(3)` and is thus prepared easily by FOLLOWING reaction |
|
| 40. |
Value of electron gain enthalpy of 16, 17 groups are positive or negative which change is observed ? |
| Answer» SOLUTION :VALUE of electron GAIN enthalpy of (16, 17 GROUPS are negative) | |
| 41. |
Which bond possess more bond length between C=C and C-=C ? |
| Answer» SOLUTION :BOND LENGTH of C=C `GT` Bond length of `C-=C`. | |
| 42. |
The relationship between K_c and K_p is K_p = K_c (RT)^(Deltan) ,What would be the value of Deltan for the reaction ? NH_4Cl_((s)) hArr NH_(3(g)) + HI_((g)) |
|
Answer» 1 `K_p=K_c(RT)^(Deltan)` Where, `Deltan` = (number of MOLES of gaseous products - (number of moles of gaseous REACTANTS) For the reaction,`NH_4Cl_((s)) HARR NH_(3(g)) + HCl_((g))` `Deltan`=2-0=2 |
|
| 43. |
Whichof thefollowingion hasthe highestnumber of theupairedelectrons ? |
|
Answer» `FE^(3+)` |
|
| 44. |
When 3.2 g sulphur is vaporised at 450^(@)C and 723 mm Hg pressure, the vapours occupy a volume of 780 mL. What is the molecular formula of S vapours ? |
|
Answer» `S_(2)` `THEREFORE``M=(3.2xx0.0821xx760xx1000xx723)/(723xx780)=256` `because` 32 g of SULPHUR has 1 atom of S. `therefore` Molecular formula=`S_(8)` |
|
| 45. |
Write the generalelectronicconfigurationof s-,p-, d-, andf-block element : |
|
Answer» SOLUTION :(i) s- BLOCKELEMENT`: ns^(1-2) ` where n=2-7(II)p-Blockelements `: ns^(2)np^(1-6)` where n= 2-7 (iii)d- Blockelements `: (n-1) d^(1-10)ns^(0-2) ` where n=4-7 (iv) F- Blockelements `: (n-2) f^(0-14) `(n-1) `d^(0-2)ns^(2)` where n= 6-7 |
|
| 46. |
What is the mass percentage of Carbon in Methane (CH_(4)) (Molecular mass of CH_(4)=16) |
| Answer» SOLUTION :`12/16xx100=75%` or 75% | |
| 47. |
What is the oxidation state of sulphur in (NH_(4))_(2)S_(2)O_(8) ? |
|
Answer» 5 (ii) `S_(2)O_(8)^(2-)` has one peroxy - O - O - BOND in which two oxygen atoms are in (-1) state, Remained six .O. atoms have -2 oxidation number. `therefore2S+2(O)+6(O)=-2` `therefore2x+2(-1)+6(-2)=-2` `therefore2x-2-12=-2` `therefore2x=+12` `thereforex=+6`  The two .O. atoms have (-1) oxidation number |
|
| 48. |
What is relation between vapour pressure and temperature ? What is vapour pressure at 373 K temperature ? |
| Answer» SOLUTION :Vapour PRESSURE `prop` Temperature, At 373 K or `100^(@)C` vapour pressure BECOMES 1 atm. | |