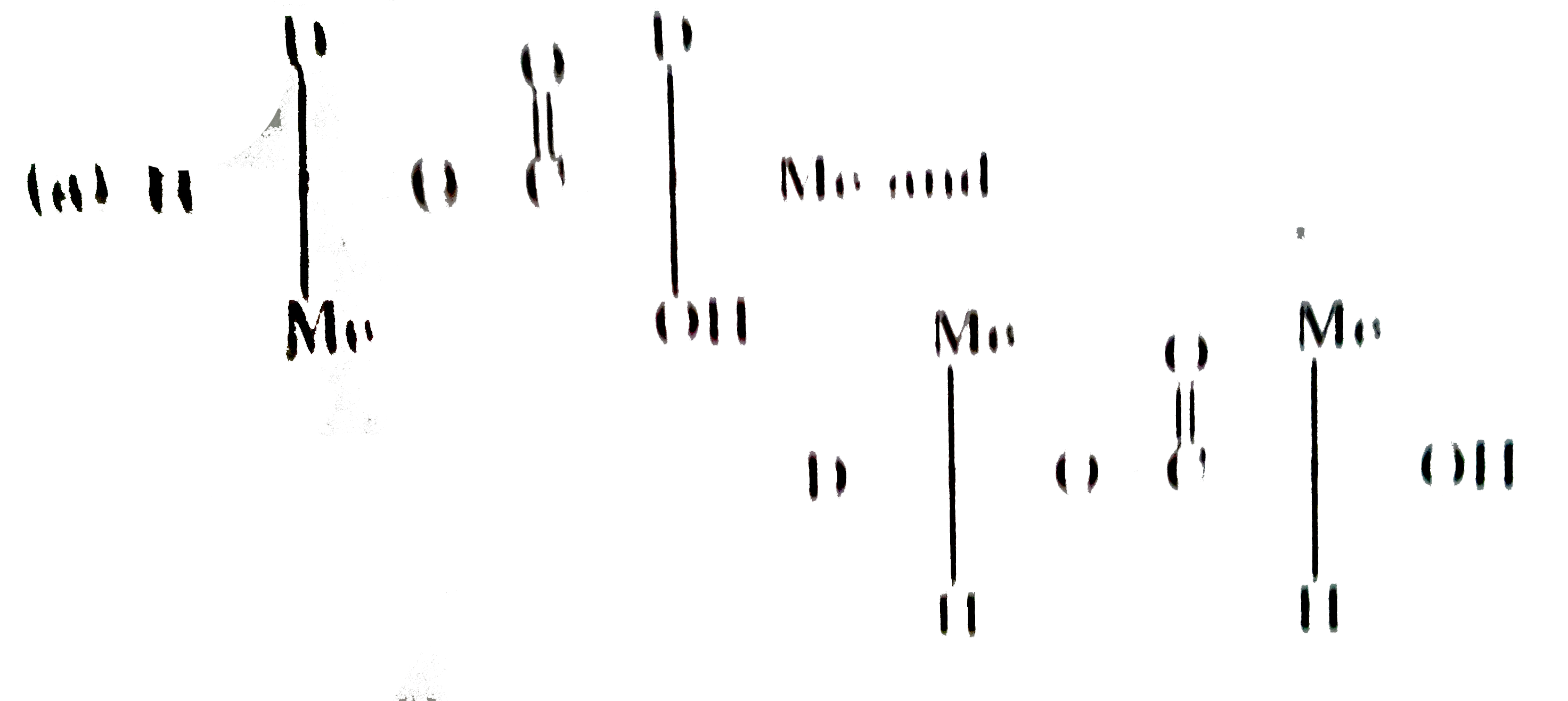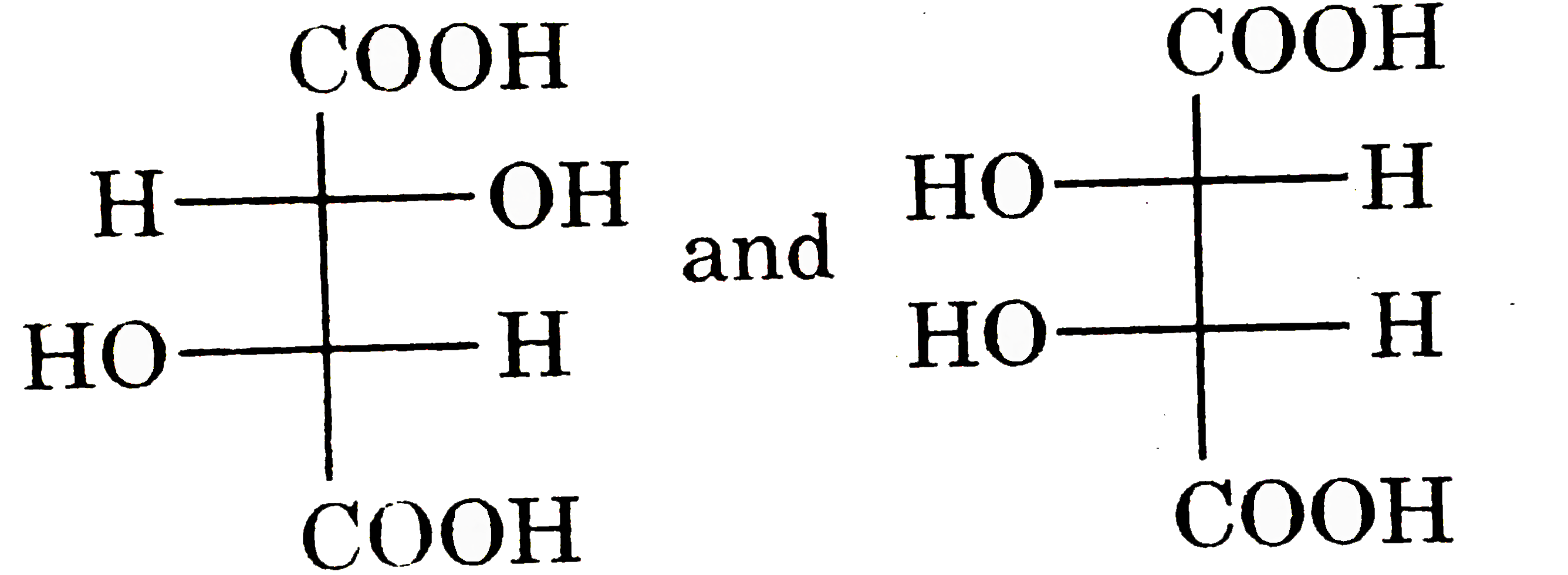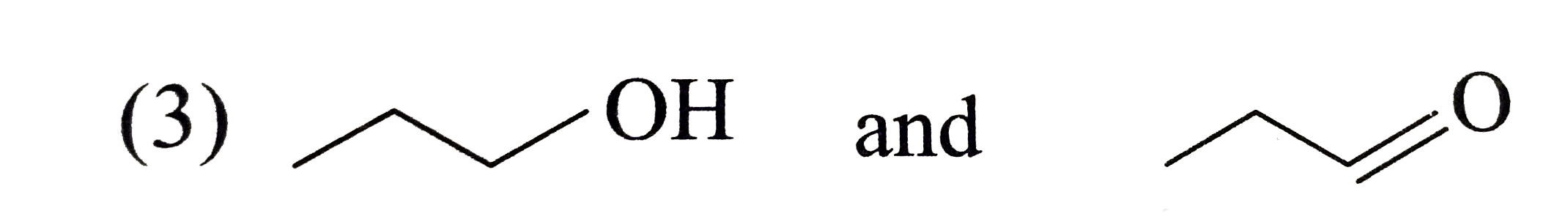Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which are isomers? |
|
Answer» ETHANOL and ethoxyethane |
|
| 2. |
What will be the product formed in the following reaction? |
|
Answer» SOLUTION :`CH_(3)-CH_(2)-CH_(2)-CH_(3)overset(kMnO_(4))(to)?` No reaction will occur as alkanes except those having tertiary HYDROGEN resist oxidation. |
|
| 3. |
what is thedegeneracy of : 1st excited state of CL=X 2nd excited state of Cl=y 1st excited state of H=z Find the value of y+z-x=t t is the degeneracy of : |
|
Answer» 2ND excitedstate of `H^(-)` y=4 z=4 t=3 |
|
| 4. |
What is the ph of 1M NaCl at 298 K. |
|
Answer» |
|
| 5. |
Which one of the following is true about the effect of increase in temperature on the distribution of molecular speeds in a gas? |
|
Answer» the area under the DISTRIBUTION CURVE remains the same as that under lower temperature The graph becomes broader as AVERAGE KE `uarr` `U_(mp) uarr` but fraction corresponding to it `DARR`. |
|
| 6. |
Which of the following metal carbonate will give of metal and oxygen on heating? |
|
Answer» `Ag_(2)CO_(3)` |
|
| 7. |
Whichof the followingis aliphatic saturated hydrocarbon |
| Answer» Answer :a | |
| 8. |
Which is a planar molecule? |
|
Answer» `CH_(2)=CH_(2)` |
|
| 9. |
Which of the following metals is obtained by electrolytic reduction process ? |
|
Answer» Fe |
|
| 11. |
Which of the following is correct regarding stability of the following pair of species ? |
|
Answer» `CH_(2)=OVERSET(+)N=overset(-)N gt barCH_(2)-overset(+)N-=N` |
|
| 13. |
Which of the following is used in the extraction of gold |
| Answer» SOLUTION :`CO_(2)` | |
| 14. |
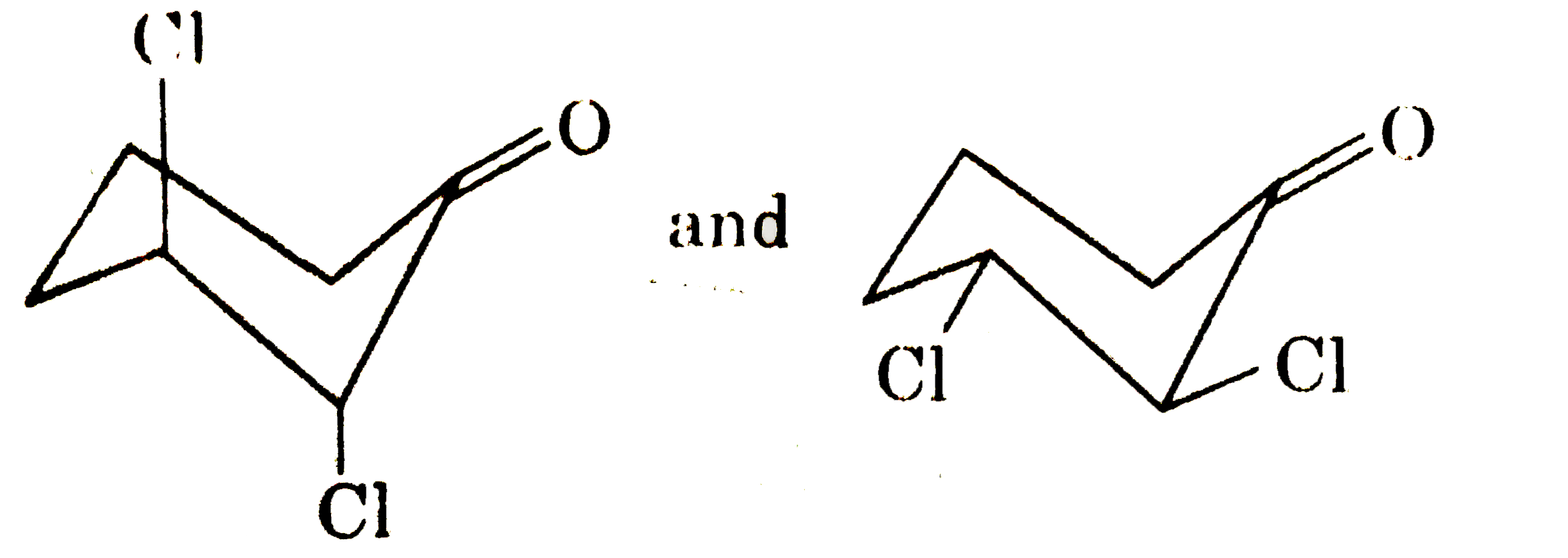
Which of the following pair of compounds can be speparted by fractional crystyallisation? |
|
Answer»
(d) They have DIFFERENT dipole MOMENT |
|
| 15. |
What is heterolytic cleavage of chemical bond? |
| Answer» Solution :The covalent bond breaks in such a FASHION that the SHARED PAIR of electrons remains with one of the fragments is CALLED heterolytic cleavage | |
| 16. |
Write the IUPAC names of the following : (i) {:(""C_(2)H_(5)""C_(2)H_(5)),("|""|"),(CH_(3)-C-CH_(2)-C-CH_(3)),("|""|"),(""NO_(2)""I):} (ii) {:(""F""Cl),("|""|"),(CH_(3)-C-CH_(2)-C-CH_(3)),("|""|"),(""C_(2)H_(5)""C_(2)H_(5)):} |
|
Answer» SOLUTION :(i) 3-lodo-3, 5-dimethyl-5-nitroheptane (II) 3-Fluoro-5-chloro-2, 5-dimethylheptane |
|
| 17. |
Which of following are correct |
|
Answer» `B_(2)H_(6)` is nonplanar |
|
| 18. |
Which subtance is used to decolourise a coloured sample of organic compound ? |
| Answer» SOLUTION :ANIMAL CHARCOAL. | |
| 19. |
Which different type of reaction mechanism is observed in addition reaction ? |
|
Answer» SOLUTION :(i) FREE radical ADDITION mechanism. (ii) ELECTROPHILIC addition reaction. |
|
| 20. |
The standard molar entropy of H_(2)O(l) is 70 JK^(-1)"mol"^(-1).Will the standard molar entropy of H_(2)O(s) be more or less than 70 J K^(-1) "mol"^(-1)? |
| Answer» SOLUTION :Less than `70 JK^(-1)"mol"^(-1)`, because ice is more ordered than `H_(2)O(l)`. | |
| 21. |
When does Frenkel defect arise ? Give reason. |
| Answer» Solution :When some ions (usually cations) are missing from the LATTICE sites and they occupy the interstitial sites so that electrical neutrality as WELL as stoichiometry is MAINTAINED , it is called Frenkel defect | |
| 22. |
True statement (s) about the following compound is/are : |
|
Answer» It shows geometrical isomerism  Optical active one chiral carbon Is present 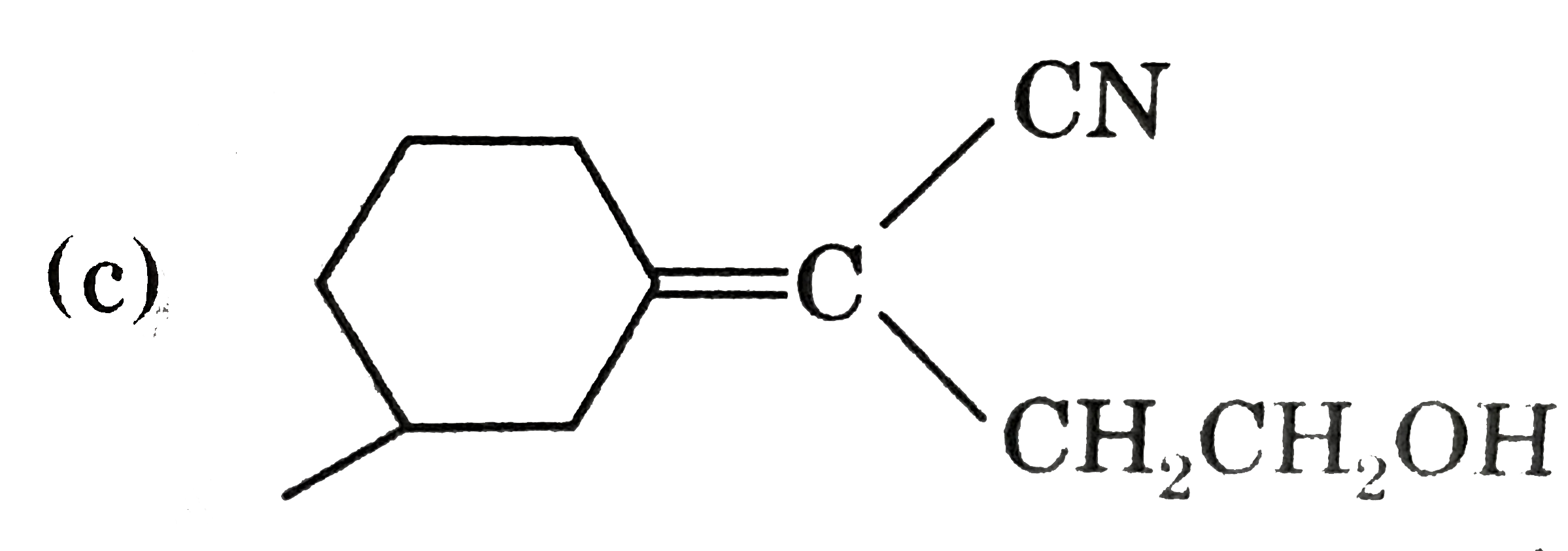 Senior groups are on opposite side 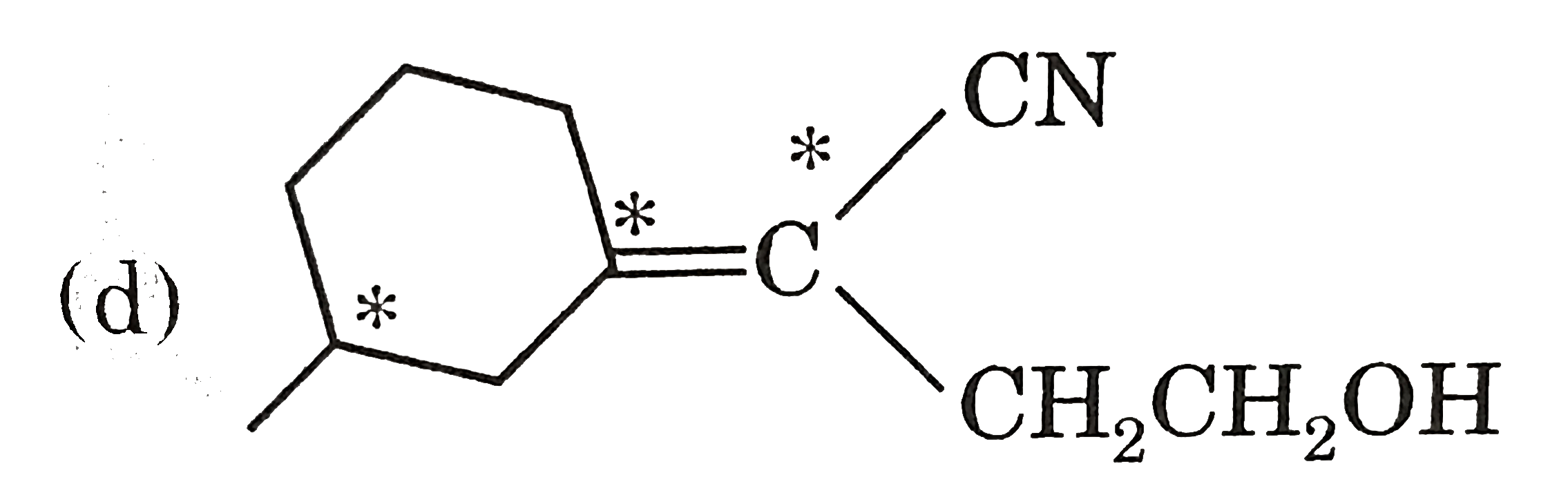 Three STEREOCENTRE Three STEREOCENTRE
|
|
| 23. |
Which of the following are the correct reasons for anomalous behaviour of lithium? |
|
Answer» EXCEPTIONALLY SMALL size of its atom |
|
| 24. |
Which compound in each of the following pairs will react faster in S_(N)2 reaction with OH ion ? (i) CH_(3) Br or CH_(3) I (ii) (CH_(3))_(3) C Cl or CH_(3) Cl |
|
Answer» Solution :SINCE 1 ion a better leaving group than Br ion , therefore `CH_(3) I` will react faster than `CH_3 Br` in `S_(N) 2` reaction with `OH^(-)` ion . (ii) On steric grounds `I^(@)` alkyl halides are more reactive than tert alkyl halide in `S_(N) 2` reactions . Therefore , `CH_(3) CL` will react at a faster RATE than `(CH_(3) )_(3) C Cl` in a `S_(N) 2` reaction with OH ion . |
|
| 25. |
Van der Waal's constant 'a' has the dimensions of |
|
Answer» mol `lit^(-1)` |
|
| 26. |
''The total mass of reactants is always equal to the total mass of products in a chemical reaction.'' This statement is known as |
|
Answer» LAW of CONSERVATION of mass |
|
| 27. |
Which of the following is not a non-viable particulates |
|
Answer» Smoke DUE to combustion of organic MATTER |
|
| 28. |
The standard enthalpy of formation (Delta H^(0)) at 298K for methane, CH_(4(g)) is 74.8 kJ mol^(1). The additional information required to determine the average energy for C-H bond formation would be |
|
Answer» The dissociationeneergy of hydrogen molecule, `H_(2)` |
|
| 29. |
Which of the following sets of quantum numbers are correct ? |
|
Answer» `{:(n,L,m_(1)),(1,1,+2):}` If `n=2, l =0, 1`. For `l = 1, m = -1, 0 +1`. Hence (b) is correct If `n=3, l =0, 1, 2`. For `l =2, m=-2, -1, =, +1, 2`. Hence (c) is correct If `n=3, l != 4`. Hence, (d) is wrong |
|
| 30. |
what is the nature of the reaction for the following ? DeltaG=0 |
| Answer» SOLUTION :`DeltaG=0` : The PROCESS is in EQUILIBRIUM . | |
| 31. |
When a real gas is converted from its initial to final state by adiabatic expansion, it is not pssible to calculate its volume using Boyle's law. Why ? |
| Answer» SOLUTION :BOYLE's LAW is applicable only at the codition of CONSTANT temperature. Since is adiabatic expansion, temperature is lowered Boyle's law cannot be applied. | |
| 32. |
The volume strength of solution formed by mixing 1 lit 0.5 M H_2O_2with 2 lit 0.5 M H_2O_2 |
|
Answer» 11.2 |
|
| 33. |
What are electron deficient compounds ? Are BCl_3 and SiCl_4 electron deficient species ? Explain. |
|
Answer» Solution :In an electron-deficient compound, the octet of electrons is not complete, i.e., the central metal atom has an incomplete octet. Therefore, it needs electrons to complete its octet. (i)`BCl_3` : `BCl_3`is an appropriate example of an electron-deficient compound. B has 3 valence electrons. After forming three covalent BONDS with chlorine, the number of electrons around it INCREASES to 6. HOWEVER, it is STILL short of two electrons to complete its octet. (ii) `SiCl_4` : The electronic configuration of silicon is `ns^2np^2`. This indicates that it has four valence electrons. After it forms four covalent bonds with four chlorine atoms, its electron count increases to eight. Thus, `SiCl_4` is not an electron-deficient compound. |
|
| 34. |
What is the correct reactivity order of the following electrophites? Toluene, p-H_3C-C_6H_4 - CH_3,p-CH_3- C_6H_4-NO-2 ,p-O_2N-C_6H_4-NO_2 |
Answer» SOLUTION : Methyl group is an electron releasing group. It increases RECTIVITY of benzene ring with an electrophile. On the other hand nitro is an electron with drawing group. It deactivates the benzene ring towards reacting with an electrophile. On the basis of this, we can CONCLUDE that greater the number of methyl groups attached to the ring greater is its reactivity with an electrophile, and greater the number of nitro groups on the thrring, smaller is its reactivity with an electrophile. The activating ability order of various groups is : `NH_2 > - NHR > - NR_2 > - OH > - OR > - X`. |
|
| 35. |
Which of the following types of metals from the most efficient catalysts |
|
Answer» Alkali metals |
|
| 36. |
Vapour density of a gas, relative to air is 1.528. what is the mass of 2L of the gas at 27^(@)C temperature and 750 mm Hg pressure? [vapour density of air, relative to hydrogen=14.4] |
|
Answer» Solution :Vapour density of a gas`=("mass of certain volume of a gas")/("mass of same volume "H_(2)" gas ")` [at certain temperature and pressure] `therefore`Vapour density of the gas `=("mass of V "cm^(3)" of gas")/("mass of V "cm^(3)" of air")xx("mass of V "cm^(3)" of air")/("mass of V "cm^(3)" off "H_(2)" gas")` [at the same temperature and pressure] `=1.528xx14.4=22` `therefore`Molecular mass of the gas=`2xx`vapour density=44 Again, volume of 1 gram-mole of any gas at STP=22.4 L So, mass of 22.4L of given gas at STP=44g Now, let the volume of the gas be V L at STP. i.e., `P_(1)=750mm,P_(2)=760mm,V_(1)=2L,V_(2)=VL,` `T_(1)=27^(@)C=(27+273)=300K,T_(2)=273K` `therefore (760xx2)/(300)=(760xxV)/(273) or, V=(750xx2xx273)/(300xx760)=1.796L` Now, mass of 22.4 L of the gas at STP=44g. `therefore`Mass of 1.796g of the gas at STP`=(44xx1.796)/(22.4)=3.528g` HENCE, the mass of the gas =3.528g. |
|
| 37. |
Which of the following ores is best concentrated by froth floatation method |
|
Answer» Malachite |
|
| 38. |
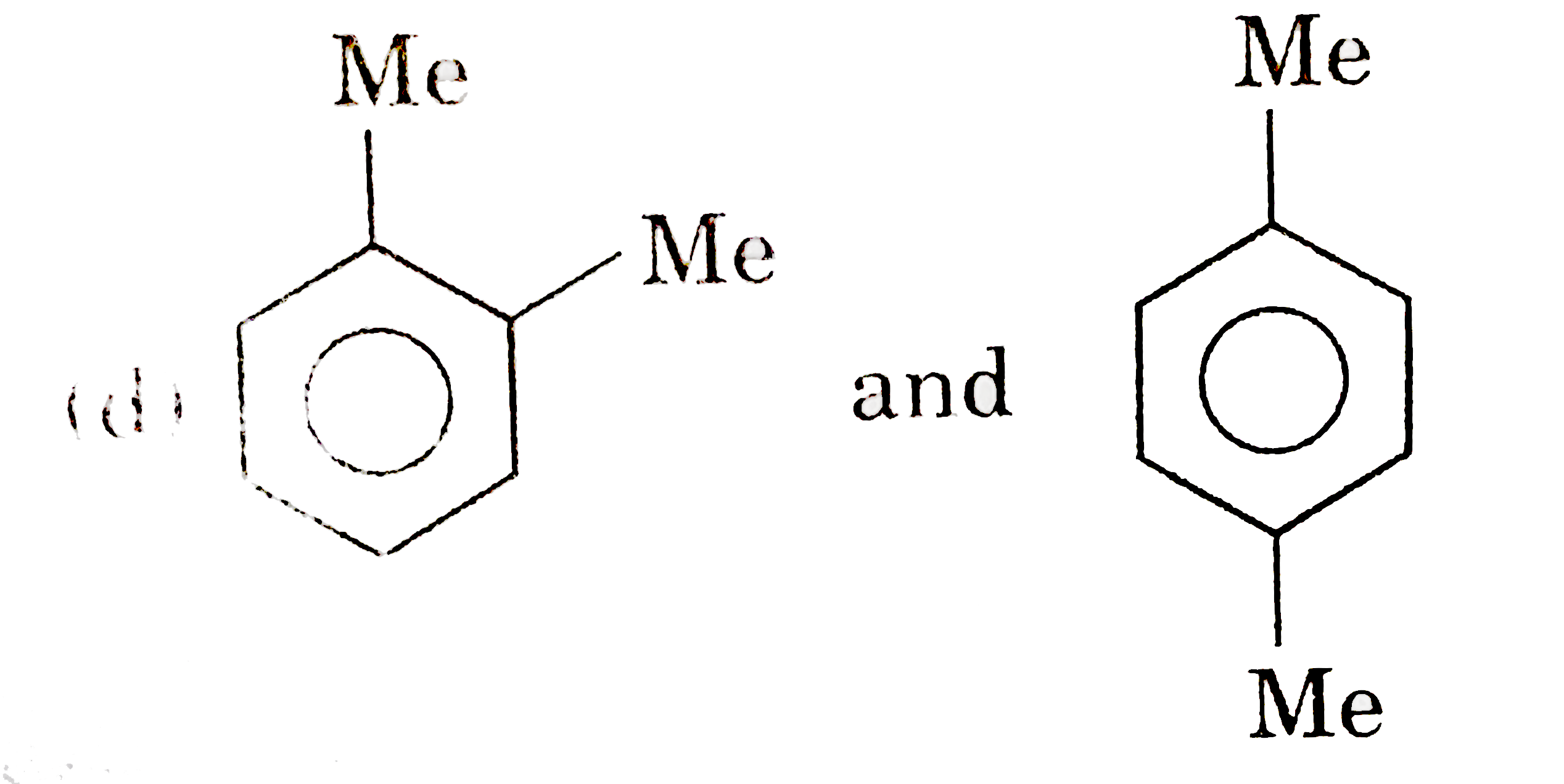
Which of the following pairs of compounds are functional isomers? |
|
Answer»
|
|
| 39. |
Which of the following is electron-deficient ? |
|
Answer» `PH_(3)` |
|
| 40. |
Which is a characteristic of a catalyst? |
|
Answer» It CHANGES the equilibrium point |
|
| 41. |
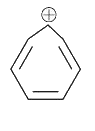
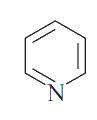
which of the following species have six Pi constructed electrons? |
|
Answer»
|
|
| 42. |
Which of the following can be used for the isolation of aluminium ? |
|
Answer» `Al_(2)O_(3) + 3C overset(Delta)rarr 2AL + 3CO uarr` `AlCl_(3) + 3Na rarr 3NaCl+Al`. |
|
| 43. |
Which of the following hydrides is an electron-rich hydride? |
|
Answer» HF |
|
| 44. |
Which of the following order is correct regarding stability (I) CH_3 - CH = C = CH-CH_3 (II) CH_3 - CH = CH -CH = CH_2 (III) CH_2 = CH - CH_2 - CH =CH_2 |
|
Answer» `I gt II gt III` |
|
| 45. |
If the velocity of the electron is 1.6xx10^(6) "ms"^(-1) , calculate the de-Broglie wavelength associated with this electron . |
|
Answer» `3.2xx10^(-7)` m `(8 xx 10^(-7))/(lambda_B) = 1/4 implieslambda_B = 32 xx 10^(-7) m ` |
|
| 46. |
Which one of the following acids would you expect to be the strongest? |
|
Answer» `I-CH_(2)COOH` |
|
| 47. |
What is maxwell Boltzman distribution curve ? |
| Answer» Solution :It is indicate number of molecules of given speed. `((IN)/(N))` or graph of number of molecules against speed of fixed TEMPERASTURE which isd known as MOLECULE DISTRIBUTION CURVE. | |
| 48. |
Which one of the following will have the highest coagulation power for a ferric hydroxide sol.? |
| Answer» Answer :D | |
| 49. |
Which one of the following is called ferric ferrocyanide? |
|
Answer» A)`Na_(4)[Fe(CN)_(6)]` |
|
| 50. |
What is K_(p) for the equation 2Cl_((g)) hArr Cl_((g)) ? When the system contains equal numberof Cl_((g)) atom and Cl_(2(g)) molecules at 1 bar and 300 k |
|
Answer» `X_(Cl)=(1)/(2), PCl=P_("TOTAL")` mole fraction of = `Cl=(1)/(2), ""^(X)Cl_(2)=(1)/(2), PCl_(2)=P_("total")` mole fraction of `Cl_(2)=(1)/(2), PCl_(2) = P_("total") xx Cl_(2)=(1)/(2) :. K_(P)=(PCl_(2))/((PCl)^(2))=((1)/(2))/(((1)/(2))^(2))=2` |
|