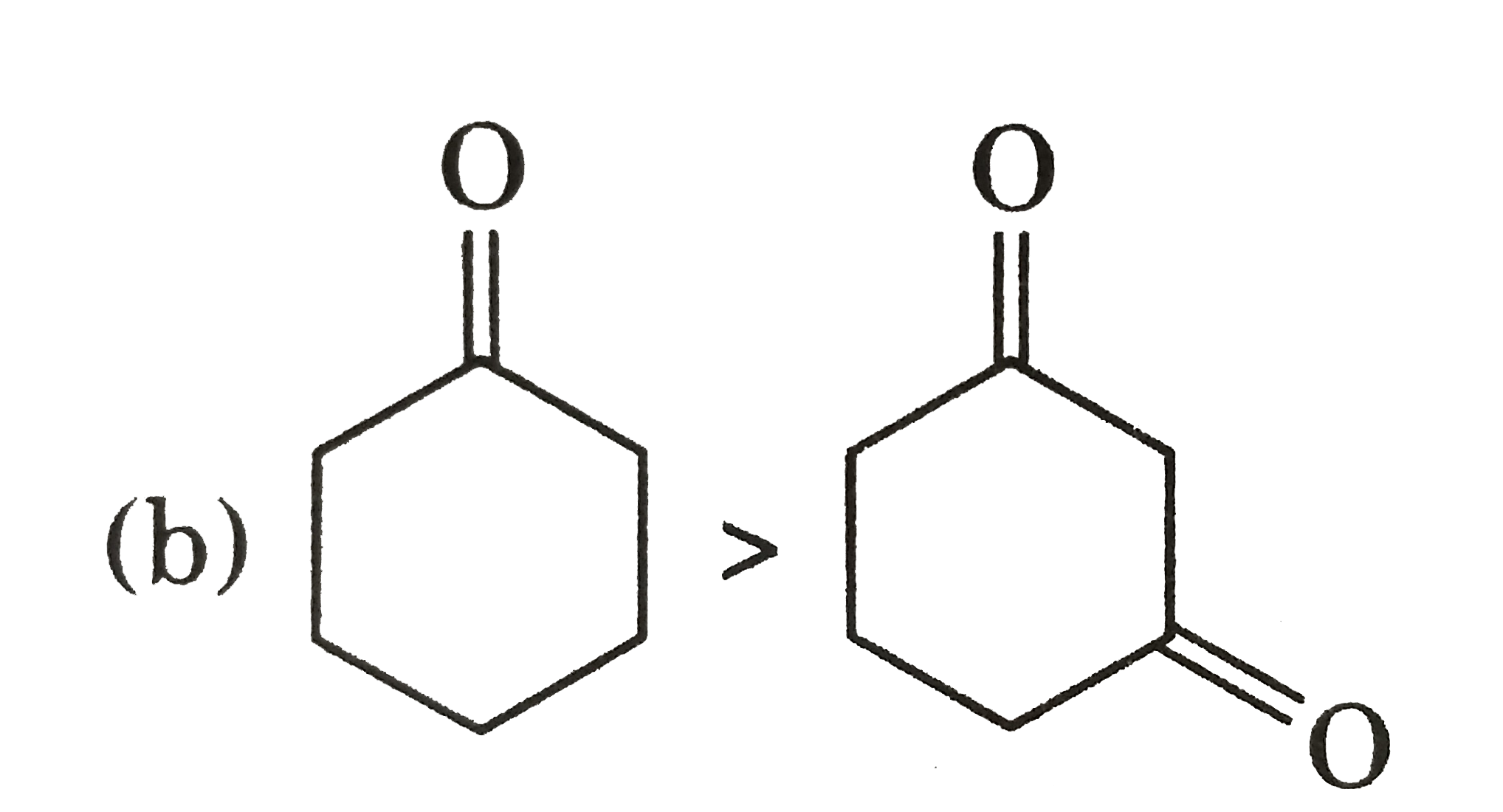
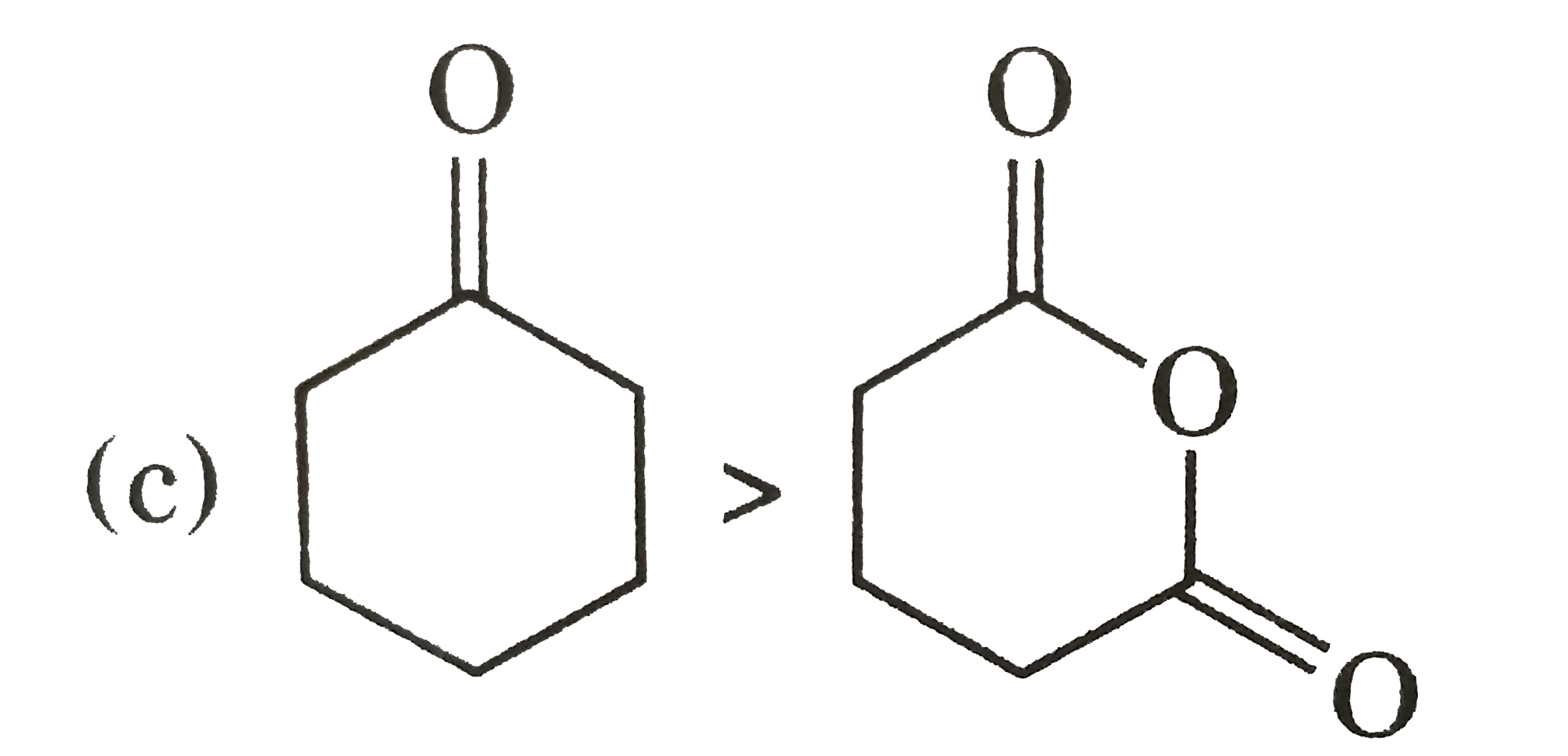
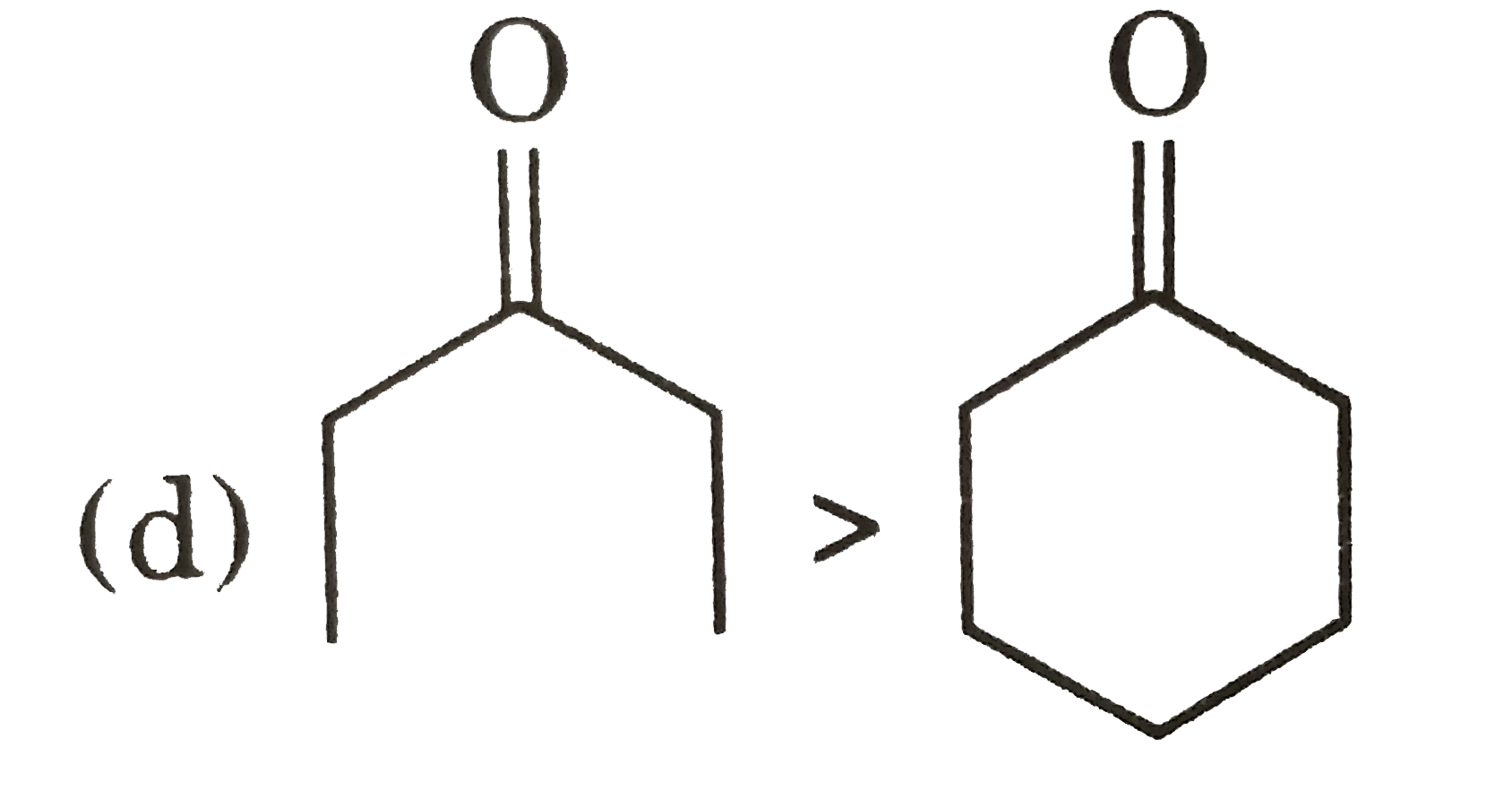
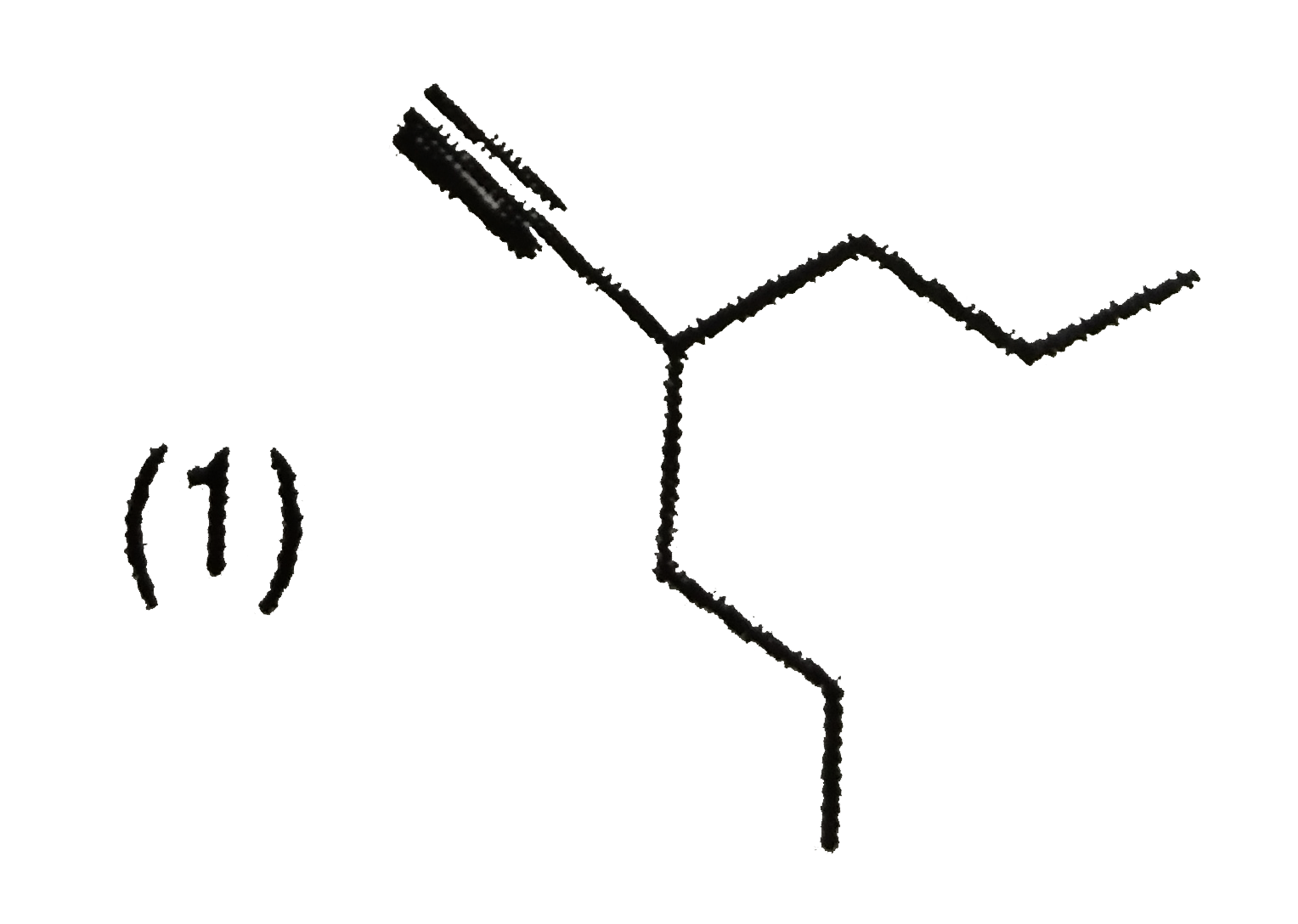
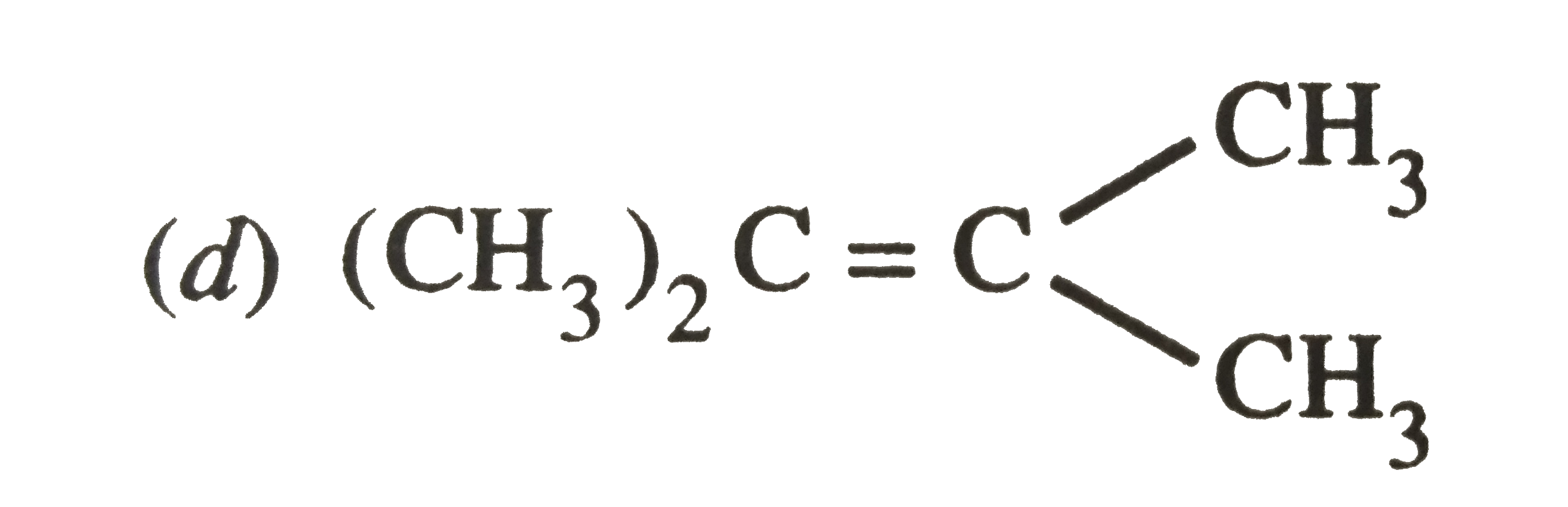Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The solubility of AgCl in water and 0.1 NaCl, in which one it is more ? |
| Answer» SOLUTION :The SOLUBILITY of AGCL is more in WATER and LESS in AgCl. | |
| 2. |
The velocityof electronin orbitdepends on whichfactors ? |
|
Answer» SOLUTION :As thepositivechargeof NUCLEUSINCREASE THEVELOCITY of electronincrease. Asthe principlequantumnumberincreasethevelocityof electronincrease. |
|
| 3. |
What is a semiconductor ? Mention the two main types of semiconductor. |
| Answer» SOLUTION :Solids which have conductivity INBETWEEN those of CONDUCTORS and insulator , i.e., ranging from `10^(-6)` to `10^4 "ohm"^(-1) m^(-1)` are called semiconductors. The two main types are n-type and p-type. | |
| 4. |
Which of the following gas when passed through dilute blood will impart a cherry red colour to the solution ? |
|
Answer» `CO_(2)` |
|
| 5. |
Which among the following is/are correct about chromium ? |
|
Answer» Its electronic configuration is `3d^4 4s^1` Total spin = Number of unpaired electron `xx 1/2 = 6 xx 1/2 = 3` Spin multiplicity `= (2sum s+1) = 2 xx 5 xx 1/2 + 1= 6` Magnetic moment `=sqrt(N(n+2))` where n= number of unpaired electrons. `=sqrt(6xx8) = sqrt(48) ` B.M. |
|
| 6. |
Which one of the following statements about the zeolites is false ? |
|
Answer» They are used as cation exchangers |
|
| 7. |
What is r.m.s speed of O_(2)molecule if its kinetic energy is 2 k cal "mol"^(-1)? |
|
Answer» `7.24 xx 10^`2 m/sec `sqrt((2 xx 2 xx 10^3 xx 4.18)/(32 xx 10^(-3)) = 7.24 xx 10^(2)` sec. |
|
| 8. |
Which one of the following has least acidic character? |
|
Answer» HCOOH |
|
| 9. |
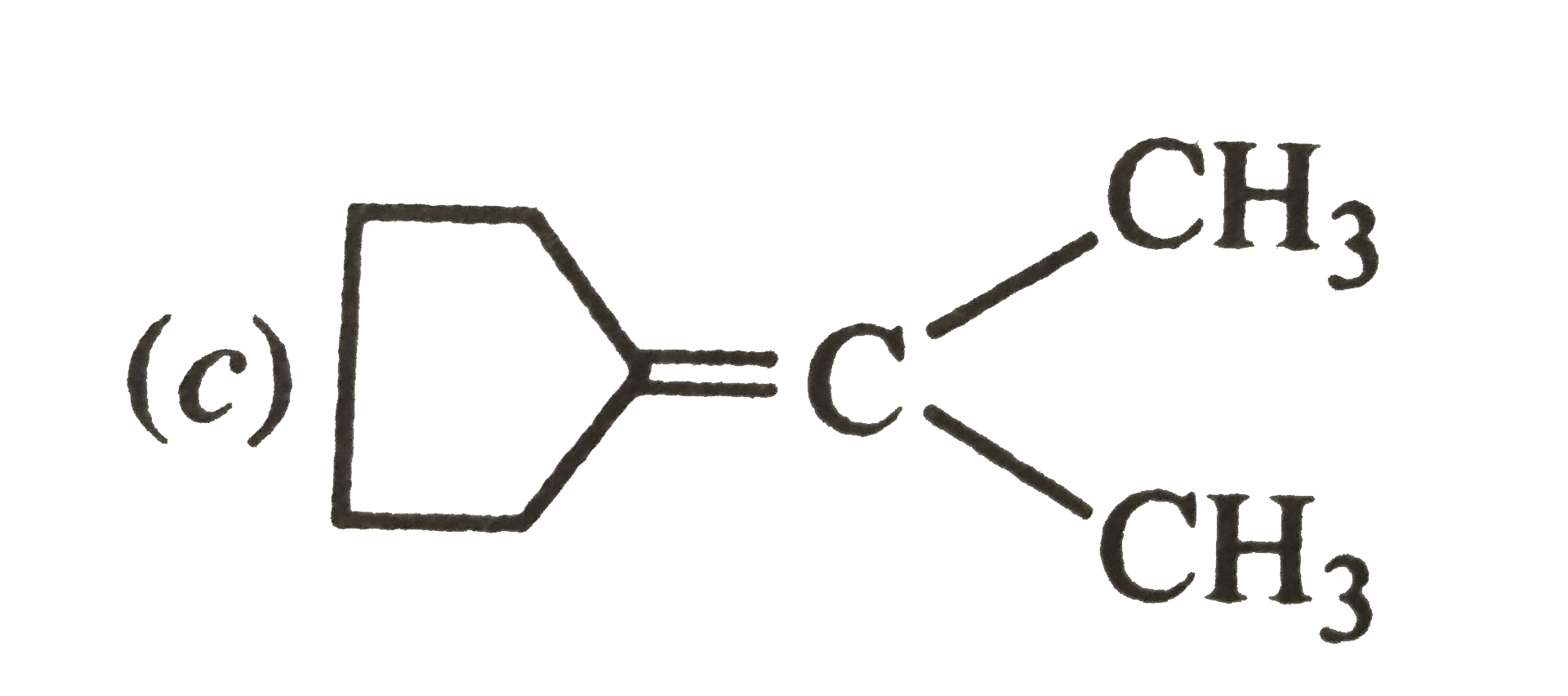
Which of the following alkenes on ozonolysis give a mixture of ketones only ? |
|
Answer» `CH_3-CH=CH-CH_3`  On the other hand, alkenes (a) and (B) give a mixture of two aldehydes. 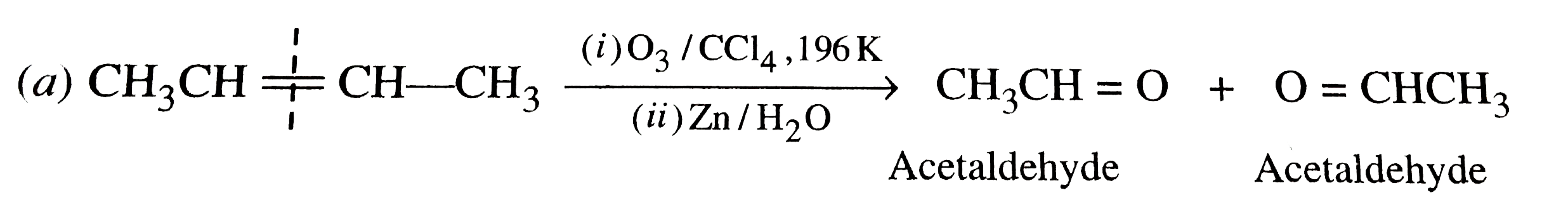 (b)`CH_3-undersetunderset(CH_3)|CH-CH=CH_2 underset((ii)Zn//H_2O)OVERSET((i)O_3//C Cl_4, 196 K)to underset"2-Methylpropanal"(CH_3-undersetunderset(CH_3)|CH-CH=O)+underset"Methanal"(O=CH)` |
|
| 10. |
Which of the following statements are incorrect? |
|
Answer» `Li^(+)` has minimum DEGREE of HYDRATION among alkali METAL cations. `Li^(+) gt Na^(+) gt K^(+) gt Rb^(+)gtCs^(+)` |
|
| 11. |
which quantum number tends to specify the orientation in space for an orbital ? |
| Answer» SOLUTION :MAGNETIC QUANTUM NUMBER | |
| 12. |
Write the isomers of the compound whose molecular formula is C_(4)H_(9)Br. |
Answer» SOLUTION : . .
|
|
| 13. |
Why are alkali metals always univalent? Which alkali metal ion forms largest hydrated ion in aqueous solution? |
| Answer» Solution :They are always univalent because after losing one electron, they acquire NEAREST inert gas configuration. Lit forms largest hydrated CATIONS because it has the highest HYDRATION energy. | |
| 14. |
X overset("Soda lime ")rarrY overset(Cl_(2)//uv-"light")rarrZ-overset(2Na)underset("dry either")rarr CH_3 - CH_(2) - CH_(2) - CH_3+2NaCl Then X, Y and Z include |
|
Answer» Sodium ethanoate, ETHANE and EHTYL chloride |
|
| 15. |
What are electrophile? Give an example. |
|
Answer» Solution :The electron DEFICIENT SPECIES are CALLED electrophiles. EG: `Br^(+)` or `CH_(3)^(+)` or `NO_(2)^(+)` or any suitable example. |
|
| 16. |
Total volume of atoms present in a face-centred cubic unit cell of a metal is (r is atomic radius) |
|
Answer» `12/3pir^(3)` Volume of the atom =`4//3pir^(3)` Total volume in the unit cell `=4times4/3pir^(3)=16/3pir^(3)` |
|
| 17. |
What are the reasons behind the Moseley's attempt in finding atomic number? |
|
Answer» Solution :(i) The number of electrons INCREASES by the same numbers as the increase in the atomic number. (ii) As the number of electrons increase , the electronic structure of the ATOM changes. (iii)Electrons in the OUTERMOST shell of an atom (valence shell electrons) DETERMINE the CHEMICAL properties of the elements. |
|
| 18. |
What is frequency of first line of Balmer series for H-atom ? |
|
Answer» `3.29 xx10^(15) s^(-1)` `therefore` form n=3 to n=2 electron transition `V=(triangleE)/(h)` `=(2.18 xx10^(-18)j)/(6.626xx10^(-34)js)(1)/(3^(2))-(1)/(2^(2))` `=-0.4569 xx10^(15)` `=-4.57 xx10^(14)s^(-1)` |
|
| 19. |
Which one of the following is a hydrophobic sol |
|
Answer» STARCH solution |
|
| 20. |
What is a sigma bond ? |
| Answer» Solution :SIGMA bond is a bond FORMED by the OVERLAPPING of ATOMIC ORBITAL along the axis. | |
| 21. |
Which element of group 2 is used in treatment of cancer ? |
|
Answer» Magnesium |
|
| 22. |
Which of the following solutions will have the highest specific conductance ? |
|
Answer» 0.001 N |
|
| 23. |
Write short note on the following bond parameter . Bond angle |
| Answer» Solution :Bond angle : Covalent bonds are directional in NATURE and are oriented in specic directions in space. This directional nature creates a FIXED angle between two covalent bonds in a MOLECULE and this angle is termed as bond angle . | |
| 24. |
When the frequency of the incident radiation on a metallic plate is doubled, KE of the photoelectrons will be |
|
Answer» doubled If `KE_(2)` were equal to `2 hv - 2 W_(0), KE_(2)` would have been `2 XX KE_(1)`. As `(2 hv - W_(0)) gt (2 hv - 2 W_(0))`, hence, `KE_(2) gt 2KE_(1)` |
|
| 25. |
What happens when boricacid is heated ? |
| Answer» Solution :`4H_(3)BO_(3) OVERSET(370K)underset(-4H_(2)O)rarr 4HBO_(2) underset(-H_(2)O)overset(410 K)rarr H_(2)B_(4)O_(7) overset(" Redheat ")rarr 2B_(2)O_(3) +4H_(3)BO_(3)` | |
| 27. |
Which one of the following compounds hassp^(2) hybridisation ? |
|
Answer» `CO_(2)` and involves`SP^(2)` HYBRIDISATION . |
|
| 28. |
Which of the following reactions are not feasible? |
|
Answer» `HC-=CH + KOH to ` |
|
| 29. |
Which of the following pair of ions makes the water hard |
|
Answer» `NA^(+), SO_4^(2-)` |
|
| 30. |
Which type of isotops of C_(4)H_(8) are possible ? Give their names and structure. |
Answer» Solution :(i) `("Saturated CYCLIC HYDROCARBON")/("CYCLOBUTANE")`  (ii) Unsaturated hydrocarbon : 
|
|
| 31. |
Which one of the following correctly represents the variation of electronegativity (EN) with atomic number (Z) of group 13 elements? |
|
Answer»
|
|
| 32. |
Which molecule has higher enol content |
|
Answer» `CH_(3)-underset(O)underset(||)(C)-CH_(3)` |
|
| 33. |
When will DeltaH= Deltau for chemical reaction? |
| Answer» Solution :When `Deltan_((G)) = 0, DELTAH= DELTAU`. | |
| 34. |
The species having pyramidalshape is |
|
Answer» `SO_(3)` bond pair. THEREFORE , no. of bond pairs around the CENTRAL S atom `= 2 (S-F)+ 1 (S = O)=3 ` No. of electron pairs around S atom `= ( 6 + 2 + 0 //2 = 4 ` No .of lone pairs ` 4-3= 1` Now accordingto VSEPR theory , a molecule with threebond pairs and one lone pair mustbe pyramidal. 
|
|
| 35. |
Which of the following conclusions could not be derived from Rutherford's alpha-particle scattering experiment ? |
|
Answer» Most of the space in the atom is empty |
|
| 36. |
What are hybridisation states of each carbon atom in the following compounds ? CH_(2) = C = O, CH_(3)CH = CH_(2), (CH_(3))_(2)CO, CH_(2) = CHCN,C_(6)H_(6). |
|
Answer» SOLUTION :`overset(sp^(2))(C)H_(2) = overset(sp)(C) = O, ""overset(sp^(3))(C)H_(3) - overset(sp^(2))(C)H = overset(sp^(2))(C)H_(2), "" overset(sp^(3))(C)H_(3) - underset(sp^(2))overset(O)overset(||)(C)-overset(sp^(3))(C)H_(3)` `overset(sp^(2))(C)H_(2)=overset(sp^(2))(C)H - overset(sp)(C) -= N`, 
|
|
| 37. |
Write the structural formulae of all the possible isomers of C_2H_2Cl_2 and indicate which of them is non-polar ? |
| Answer» SOLUTION :(i)cis-1,2-dichloroethene , (II) trans-1,2-dichloroethene and (III)1,1-dichloroethene, (ii) is NON POLAR | |
| 38. |
Which of the following classes of organic compounds show metamerism? |
|
Answer» Ethers |
|
| 39. |
What would be the electronic configuration of HeH^(-) molecular ion ? Calculateits bond order and comment on its stability . |
|
Answer» Solution :Total NUMBER of ELECTRONS in `HeH^(-) = 2 + 1 + 1 = 4therefore ` Its electronic configuration will be `sigma _(1s)^(2)sigma _(1s)^(**2)` Bond order = `(1)/(2) (2-2) = 0 ` . Hence , this ION does not exist . |
|
| 40. |
Which of the following possess almost same properties due to lanthanide contraction? |
| Answer» Solution :Zn, HF | |
| 41. |
what information is observed from positive , zero and negative volumes of change in entropy ? |
|
Answer» SOLUTION :`DeltaSlt0` = The PROCESS is non - spontaneous `DeltaSlt0` = The process exist in EQUILIBRIUM. `DeltaSlt0` = The process is spontaneous |
|
| 42. |
Which of the following is/are incorrect order of percentage enolic content? |
|
Answer»
|
|
| 43. |
What is formula of compound if elements are given containing ? (i) Sodium and oxygen (ii) Aluminium and bromine (iii) Silicon and oxygen(iv) Phosphorus and chlorine(v) 19^(th)element and iodine (vi) 18^(th)element and oxygen |
Answer» SOLUTION :
|
|
| 44. |
Which of the following contains unpaired electrons ? |
|
Answer» `NO_2` |
|
| 45. |
What is the potential drop through which an electron , witha de Broglie wavelength of 1.5 "Å" , should be accelerated , if its de Broglie wavelength should be reduced to 1 "Å" ? |
|
Answer» 110 volts |
|
| 46. |
Which of the following is not true about the voids formed in 3 dimensional hexagonal close packed structure ? |
|
Answer» A tetrahedral void is formed when a sphere of the second LAYER is present above triangular void in the first layer (d) is not true because octahedral void is combination of TWO triangular voids one with vertex UPWARD and the other with vertex DOWNWARD and not their overlap |
|
| 47. |
Which sub shell is filled up progressively in actinoids |
|
Answer» `1.4f` |
|
| 48. |
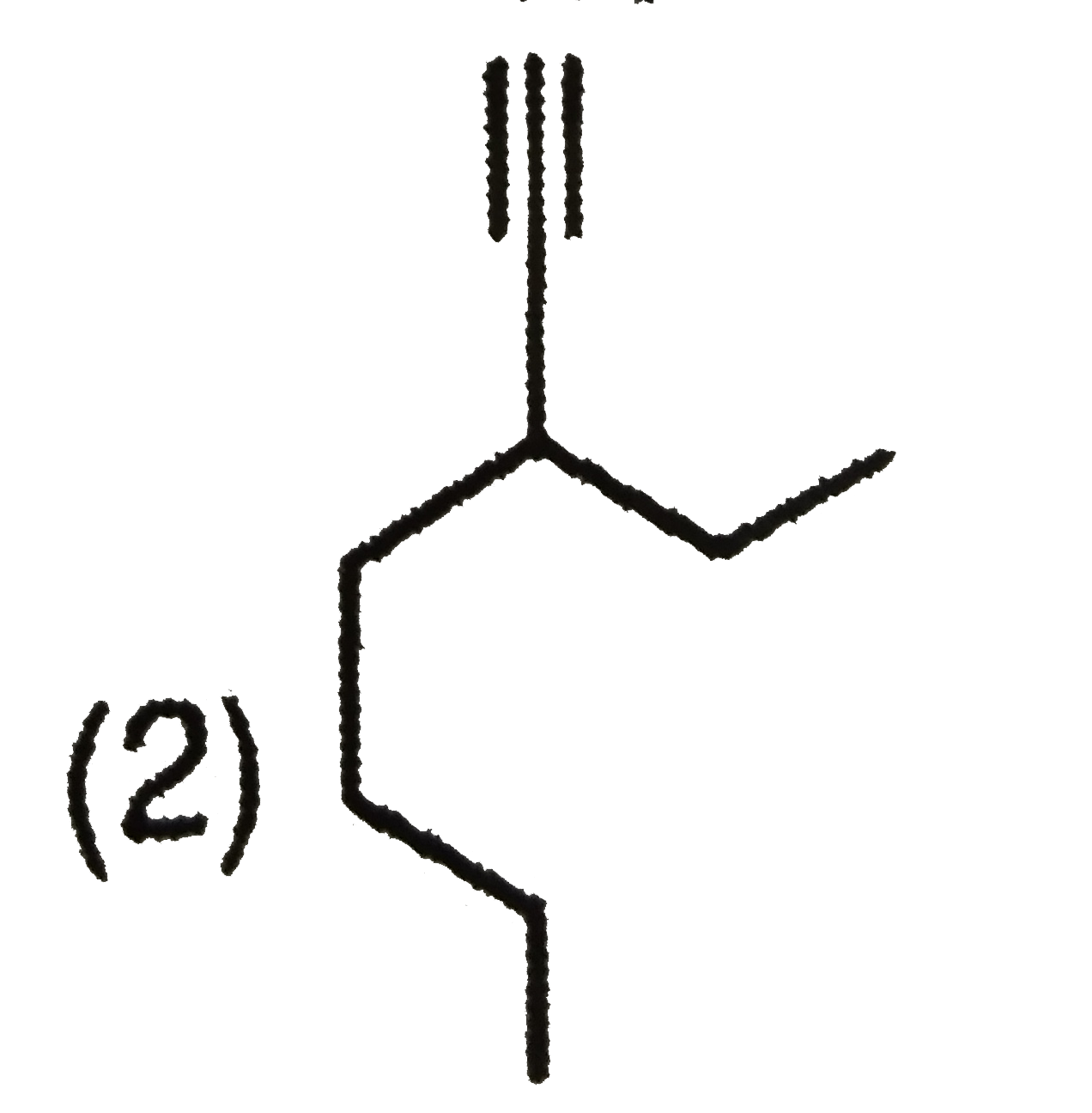
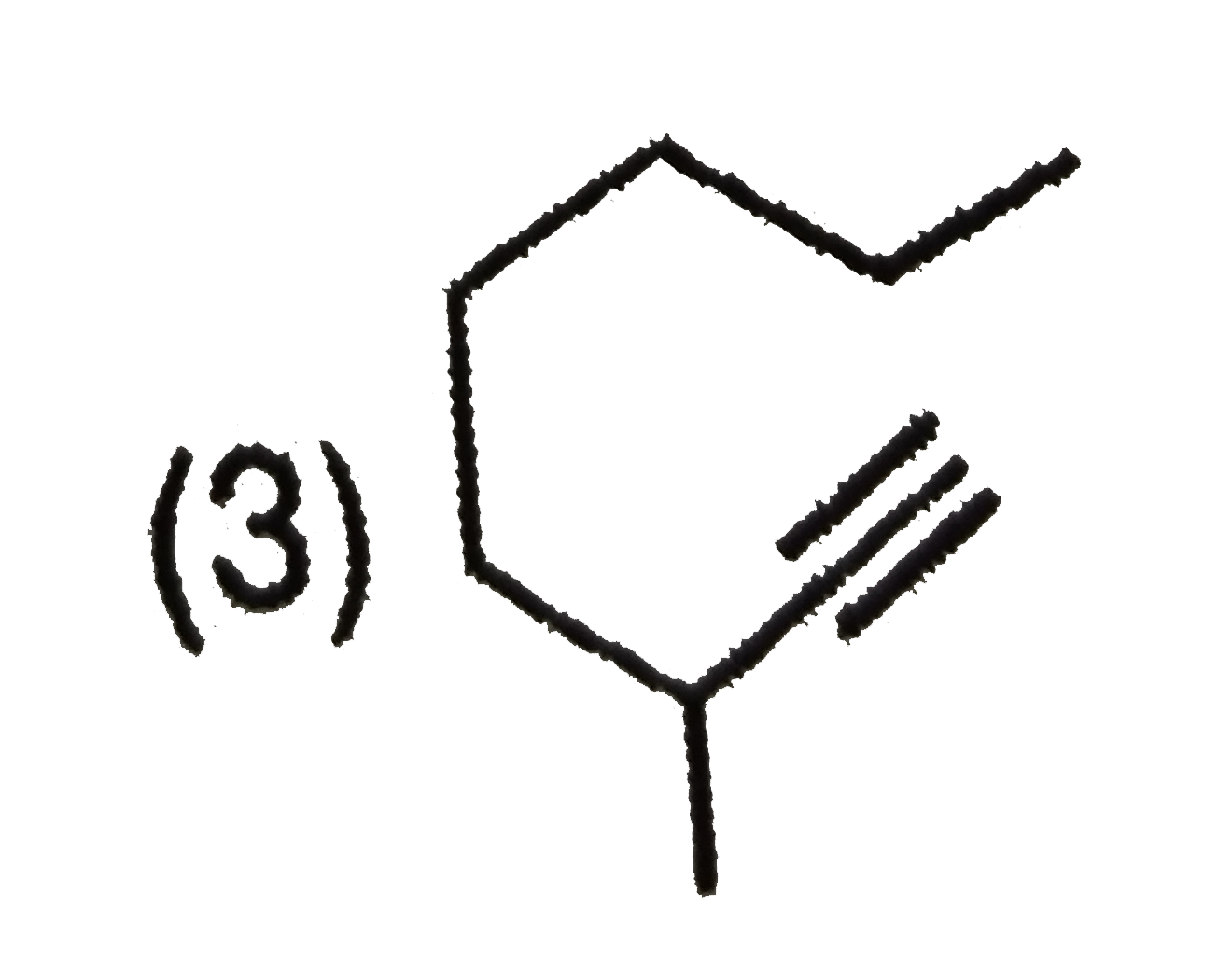
Whci alkyne will give 3-Ethyl heptane on catalytichydrogenation |
|
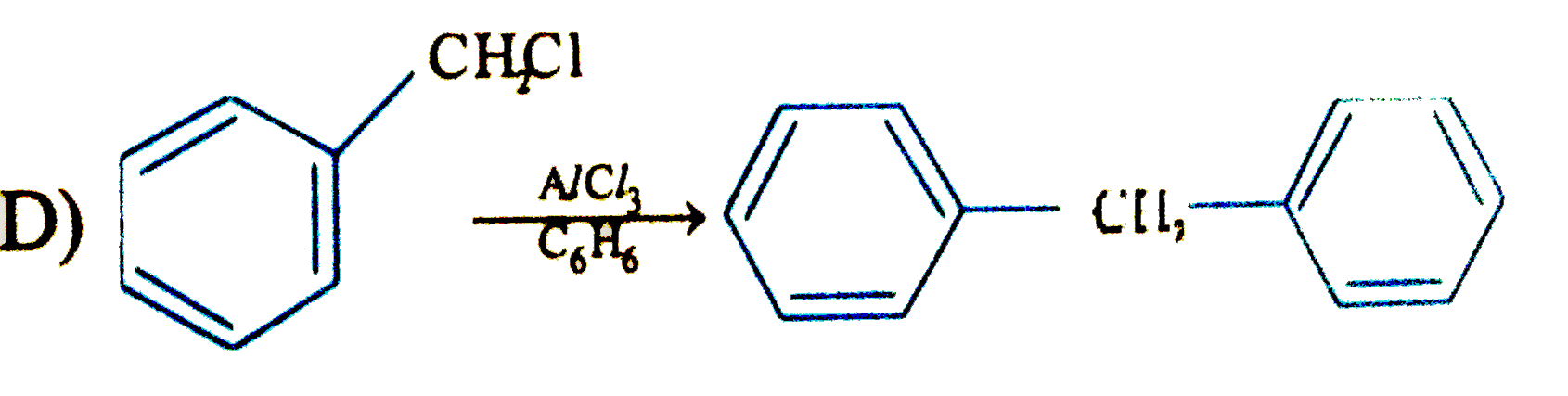
Answer»
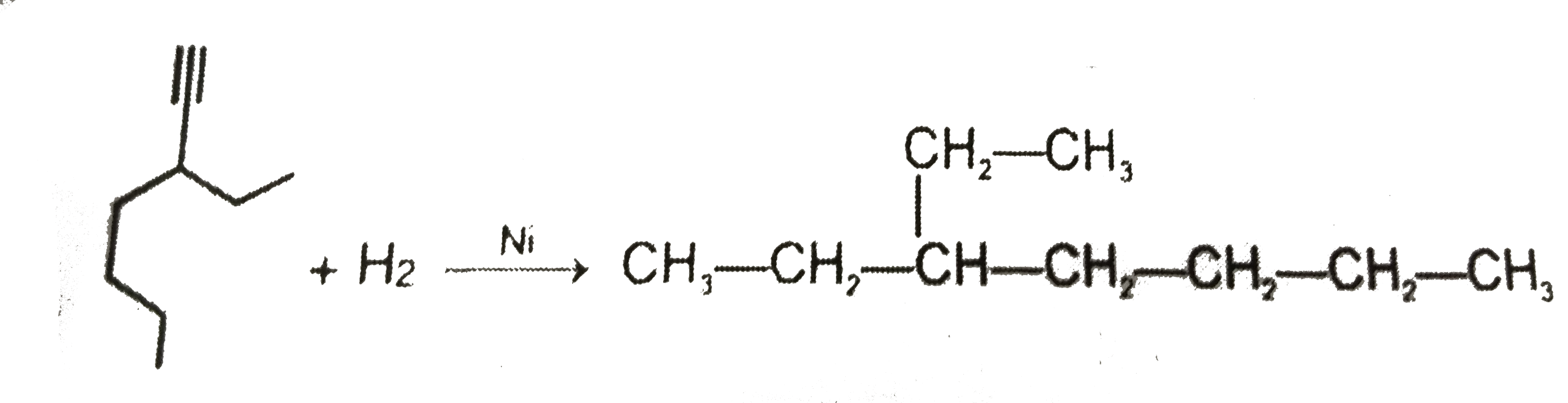 have TWO types of chemically DIFFERENT hydrogen atom so it forms two MONOCHLORO ISOMERS on photochemical chlorination. have TWO types of chemically DIFFERENT hydrogen atom so it forms two MONOCHLORO ISOMERS on photochemical chlorination.
|
|
| 49. |
Which of the following cases, reactant and product are correctly matched. |
|
Answer»
|
|
| 50. |
Write first elements of the period 1 to 7. |
Answer» SOLUTION :
|
|