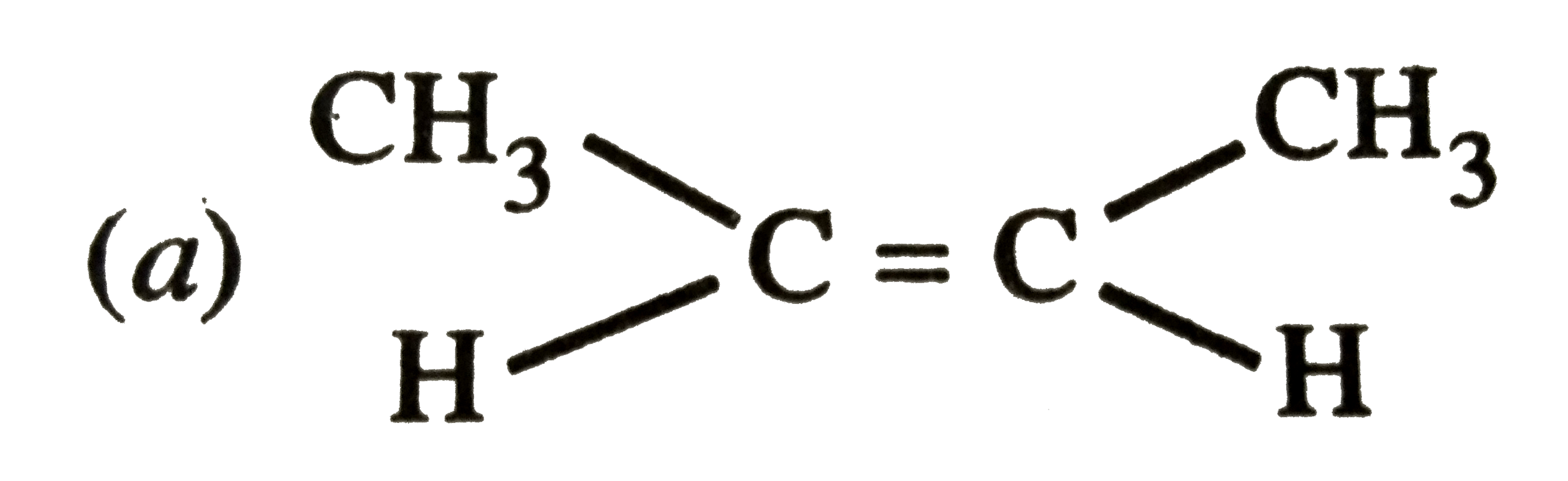Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
What is the difference inenergyof 2sorbitalsof hydrogenand 2sorbitalof otheratoms ? |
| Answer» SOLUTION :Electronof 2SORBITAL of HYDROGEN `LT`electronof2sorbitalsof otheratom | |
| 2. |
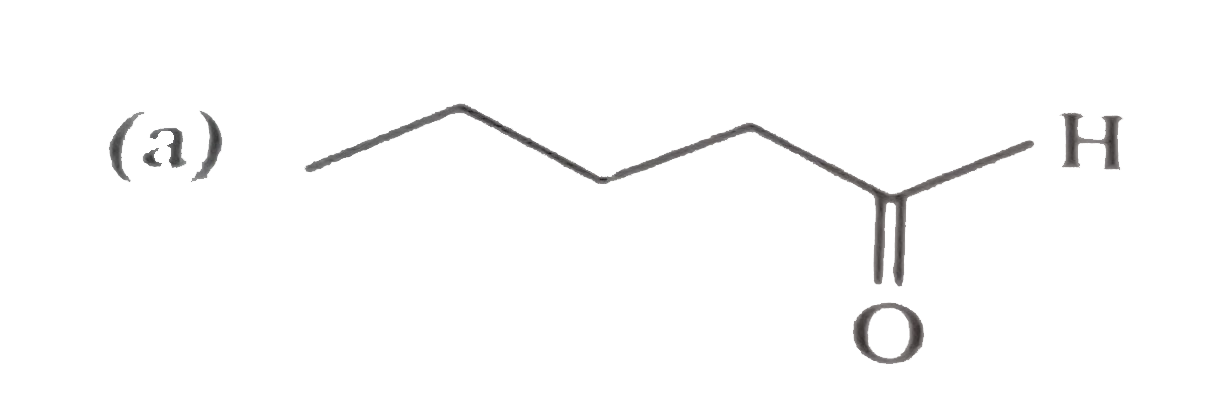
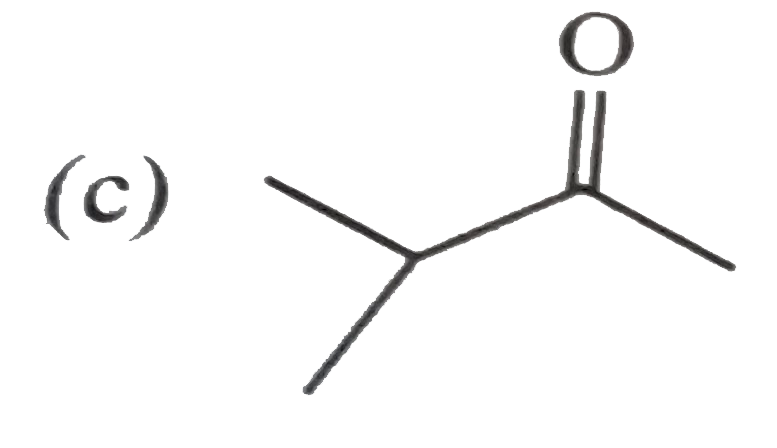
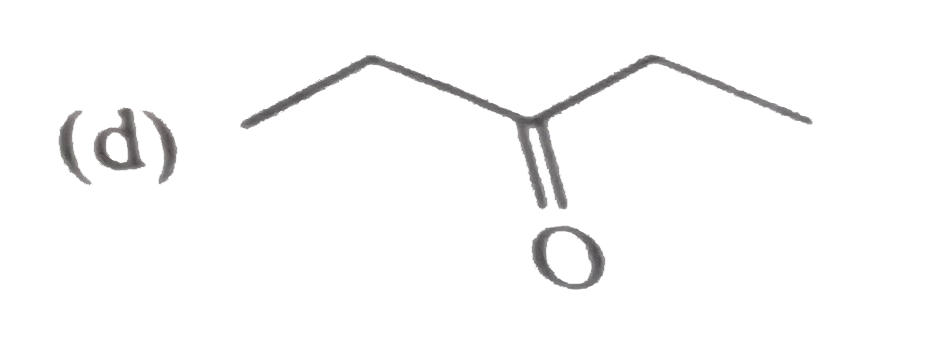
Unknown compound (A) C_(5)H_(10)O gives positive test with 2,4-DNPbut negative test with Tollen's reagent . It also give yellow precipitate with I_(2)//NaOH. (A) is : |
|
Answer»
|
|
| 3. |
When a mixture of anhydrous Na_2CO_3 and NaHCO_3is heated to 100^@C, a loss in mass is recorded. This is due to |
|
Answer» DECOMPOSITION of `Na_2CO_3` ALONE |
|
| 4. |
The stability of 2,3-dimethyl but-2-ene is more than 2-butene. This can best be explained in terms of |
|
Answer» STERIC effect 
|
|
| 5. |
Which of the following equations depict the oxidizing nature of hydrogen peroxide ? |
|
Answer» `MnO_(4)^(2-)toMn^(2+)` |
|
| 6. |
Which metals is present in chlorophyll? How does this metals react with N_(2) ? |
| Answer» Solution :MG is pressent in chlorophyy. `N_(2)` REACTS with Mg to form MAGNESIUM NITRIDE. | |
| 7. |
The sun of the numbers of lone pairs of electrons on each central atom in the following species is …….. [TeBr_(6)]^(2-),[BrF_(2)]^(+), SNF_(3) and ]XeF_(3)]^(-) (Atomic number : N= 7, F=9 , S=16, Br=35, Te=52, Xe=54) |
|
Answer» In `[TeBr_(6)]^(2-) T = (1)/(2) (6 + 6 + 2 ) = 7 , B = 6 ` `thereforelp = 7 - 6 = 1 ` In `BrF_(2)^(+) , T= (1)/(2) = ( 7 + 2 - 1) = 4 , B = 2 ` ` thereforelp = 4 - 2 = 2 ` In ` XeF_(3)^(-), T = (1)/(2) (8 + 3 + 1) = 6 , B = 3 ` `therefore lp = 3 ` STRUCTURE of `SNF_(3) ` is  where S has zero where S has zerolone pair of electrons. `{:("Species" ,[TeBr_(6)]^(2-), BrF_(2)^(+),SNF_(3), XeF_(3)^(-)),("Lone pairs",1,2,0,3):}` on central atom Total number of lone pairs of electrons = 6 |
|
| 8. |
What are the demerties of long form periodic table. |
|
Answer» Solution :(i)POSITION of hydrogen is not defined till now. (II)Lanthanides and actinides still FIND place in the bottom of the table. |
|
| 10. |
Which of the following hydrocarbons has the lowest dipole moment ? |
|
Answer»
|
|
| 11. |
Which of the following contains the maximum number of atoms? |
|
Answer» 10g of `CaCO_(3)` `=0.1N_(0)` MOLECULES `=0.1N_(0)XX5` atoms 2) `4gH_(2)=(4)/(2)=2" moles "=2N_(0)xx2` atoms 3) `9gNH_(4)NO_(3)=(9)/(80)=0.1125` moles `=0.1125N_(0)xx9` atoms 4) `1.8gC_(6)H_(12)O_(6)=(1.8)/(180)=0.01` moles `=0.01N_(0)xx24` |
|
| 12. |
When heavy water reacts with calcium carbide, the product formed is |
|
Answer» Acetylene |
|
| 13. |
Which of the following involves gain of five electrons ? |
|
Answer» `MnO_(4)^(-) RARR Mn^(2+)` |
|
| 14. |
Which of the following alcohols is most soluble in water? |
|
Answer» n-Butyl alcohol |
|
| 15. |
Which of the following statements are correct i) Both carbides of Be and Al react with water to produce methane gas ii) Be and Al dissolves in alkali to from berrylates and aluminates iii) Be forms many complexes when compared with remaining II_(A)- group elements. |
|
Answer» I, II are correct |
|
| 16. |
What do you mean by electrovalence? |
| Answer» SOLUTION :Number of IONS lost (GAINED) by ATOMS during the formation of IONIC bond. | |
| 17. |
Which elements are known as transition element and inner transition element ? |
|
Answer» SOLUTION :ELEMENT of d-block are known as transition ELEMENTS. Elements of f-block are known as inner transition elements. |
|
| 18. |
Which reducing agent, would you use to carry out the following transformation. |
|
Answer» `LiAIH_(4)` |
|
| 19. |
Which of the following metal sulphides have maximum solubility in water ? |
|
Answer» `HGS (K_(SP) = 10^(-54))` |
|
| 20. |
What is the pH of basic buffer ? How it is prepared ? |
|
Answer» SOLUTION :The pH of BASIC BUFFER is `gt` 7.0 it is PREPARED by weak base and its salt with STRONG acid in equal proportion. e.g. `(NH_4OH + NH_4Cl)` is basic buffer pH: 9.25 |
|
| 21. |
Which of the following is /are correct?\ |
|
Answer» `Al_(2)O_(3)`reacts with CaO but not with `SiO_(2)` |
|
| 22. |
Which of the following represents the correct IUPAC name for the compounds concerned ? (a) 2, 2-Dimethylpentane or 2-Dimethylpentane (b) 2, 4, 7-Trimethyloctane or 2, 5, 7-Trimethyloctane (c) 2-Chloro-4-methylpentane or 4-Chloro-2-methylpentane (f) But-3-yn-1-ol or But-4-ol-1-yne. |
| Answer» SOLUTION :(a) 2, 2-Dimethylpentane. For TWO ALKYL groups on the same carbon its locant is repeated twice, (b) 2, 4, 7-Trimethyloctane since 2, 4, 7-locant SET is lower than 2, 5, 7. (c)2-Chloro-4-methylpentane. Alphabetical order of substituents. (d) But-3-yn-1-ol. Lower locant for the principle functional group i.e., alcohol. | |
| 23. |
When I_(2) is passed through KCl, KF and KBr solution |
|
Answer» `Cl_(2)` and `Br_(2)` are evolved |
|
| 24. |
Which one of the following is related to thermodynamic equilibrium ? |
|
Answer» All of above |
|
| 26. |
Why are the pi-bonds weaker and more reactive than the sigma-bonds? |
| Answer» Solution :The extent of AXIAL overlapping is greater as compared to SIDEWAYS overlappin HENCE `pi`bonds are weaker and more REACTIVE than `SIGMA`-bonds. | |
| 27. |
The state of hybridization of C_(2), C_(3), C_(5) and C_(6) of the hydroccarbon, {:(""CH_(3)""CH_(3)),("|""|"),(underset(7)(C)H_(3)-.^(6)C-underset(5)(C)H=underset(4)(C)H-underset(3)(C)H - underset(2)(C)-=underset(1)(C)H),("|"),(""CH_(3)):} is in the sequence |
|
Answer» `sp^(3), sp^(2), sp^(2)` and sp THUS, option (d) is correct. |
|
| 28. |
Which of the alkaline earth metal hydroxide are amphoteric in nature. |
| Answer» Solution :BERYLLIUM HYDROXIDE `[Be(OH)_2]` is amphoteric in nature. | |
| 29. |
What is the value of x, y, z from the following reactions ? xS+yHNO_(3)toxSO_(3)+yNO+zH_(2)O |
|
Answer» `3,4,2` |
|
| 30. |
Which of the following is not an example of redox reaction ? |
|
Answer» `CuO+H_2rarr Cu+H_2O` |
|
| 31. |
What is COD ? Which chemical substance is generally used in its measurement ? |
| Answer» Solution :COD stands for CHEMICAL Oxygen DEMAND. It is MEASURED by treating the given sample of water with an oxidizing agent, generally `K_2Cr_2O_7`, in presence of dilute `H_2SO_4` | |
| 32. |
Which of the following statements is not correct for an electron that has the quantum numbers n=2 and m=2 |
|
Answer» The ELECTRON may have the QUANTUM number`s=+1/2` |
|
| 33. |
Which of the following is not an algae nutrient? |
|
Answer» Hg |
|
| 34. |
Which of the following ions interfere with chromyl chloride test? |
|
Answer» `NO_(3)^(-)`<BR>`Br^(-)` |
|
| 35. |
What is correct about x and y? |
|
Answer» Both are ACHIRAL molecules |
|
| 36. |
What is heat capacity ? |
| Answer» SOLUTION :HEAT required to increase 1 °C temperature of a substance is called heat CAPACITY. | |
| 37. |
Two 22.4 litre containers A and B contains 8 g of O_2 and 8 g of SO_(2) respectively, at 273 K. and 1 atm pressure, then |
|
Answer» NUMBER of MOLECULES in Aand B are same |
|
| 38. |
What is the effect in covalent bond by electron displacement? |
| Answer» SOLUTION :The PERMANENT POLARITY is PRODUCED in BOND | |
| 39. |
which has the highest pK_(a) value ? |
|
Answer»
|
|
| 40. |
What for the abbreviation SI stands? |
| Answer» SOLUTION :INTERNATIONAL SYSTEM of UNITS | |
| 41. |
Which statement is correct regarding green chemistry? |
|
Answer» Avoid USE of catalyst. |
|
| 42. |
What are vicinal dihalides? Give an example. |
| Answer» Solution :Vicinal dihalides, compounds that have halogens on ADJACENT CARBONS, are PREPARED by the reaction between a HALOGEN and an ALKENE. The simplest example is the reaction between ethylene and chlorine to give 1,2-dichloroethane (ethylene dichloride). | |
| 43. |
Which of the following is ionic ? |
|
Answer» `PbF_(4)` |
|
| 44. |
Which of the following statements are true regarding photochemical smog It is oxidsing in nature It is formed when intensity of solar radiation is very high lt is formed by PRN, Ozone and oxides of nitrogen It is formed by the particulate carbon and SO_(2) It is reducing in nature |
| Answer» Answer :D | |
| 45. |
Which one of the following is called halogen family? |
|
Answer» GROUP 17 |
|
| 46. |
What happens to oxidation number of an element during reduction? |
|
Answer» |
|
| 48. |
X' compound (C_(4)H_(8)O) decolorises bromine water react with I_(2) & NaOH it give yellow ppt identify 'X' |
|
Answer» `CH_(3)-overset(O)overset(||)(C)-CH_(2)-CH_(3)` |
|
| 49. |
Which of the following statements is wrong about athode rays? |
|
Answer» They travel in straight LINES towards cathode. |
|
| 50. |
Which of the statements is not true? |
|
Answer» `K_(2)Cr_(2)O_(7)` solution in acidic MEDIUM is orange |
|