Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
'X’ moles of N_2 gas at S.T.P. conditions occupy a volume of 10 litres, then the volume of ‘2x’ moles of CH_4 at 273^@C and 1.5 atm is |
| Answer» Answer :B | |
| 2. |
Which type of chemical reaction does given by arene compounds ? Give there suitable examples and reactions. |
|
Answer» Solution :Benzene gives electrophilic ADDITION and electrophilic substitution reactions. They also gives oxidation reactions. (A) Electrophilic addition reactions are comparatively difficult. (i) Hydrogenation : Reaction with HYDROGEN at high temperature and/or at high pressure, in presence of nickel catalyst. (ii) Chlorination : They perform addition reactions with chlorine in presence of uv radiation. (B) Electrophilic substitution reaction : Benzene possess STABILITY, so substitution reaction in benzene is difficult with reactions, the reaction occurs by liberation of electrons from the electrophiles. (i) Nitration : A nitro group is introduced into benzene is heated with a mixture of concentrated nitric acid and concentrated sulphuric acid (nitrating mixture). Such reaction substitute H of benzene with `-NO_(2)`. (ii) Sulphonation : A mixture of benzene and fuming `H_(2)SO_(4)` or oleum `(H_(2)S_(2)O_(7))` or `(H_(2)SO_(4) + SO_(3))` when heated at 353 K temperature one hydrogen of benzene is replaced to give benzene sulphonic acid, the reaction isknown as sulphonation. (iii) Halogenation : Arenes react with `Cl_(2)` and `Br_(2)` (halogens) in the presence of a Lewis acid like anhydrous `FeCl_(3), FeBr_(3)` or `AlCl_(3)` to give chlorination and bromination respectively (halogenation). `{:(phi + Cl_(2) overset(FeCl_(3))rarr phi " Cl" ),(phi + Br_(2) overset(FeCl_(3))rarr phi " Cl"):}}` where `phi = C_(6)H_(5)` (iv) Friedel-Crafts ALKYLATION reaction : When benzene is treated with an alkyl halide in the presence of anhydrous aluminium chloride, alkybenene is formed. So , addition of alkyl group in benzene in place of H is known as Friedel-Crafts alkylation. (v) Friedel-Crafts acylation reaction : The reaction of benzene with an acyl halide (RCOCl) or acid anhydride `((RCO)_(2)O)` in the presence of Lewis acids `(AlCl_(3))` yields acyl benzene. So, addition of acyl group `(RCO-)` in benzene in place of H is known as Friedel-Crafts alkylation. (C) Oxidation reactions : (i) Reductive ozonolysis : When benzene is treated with `O_(3)` in presence of `Zn + H_(2)O`, the ozonolysis and oxidation of benzene is carried out. (ii) Combustion reaction : when arenes compounds are combusted in presence of enough amount of oxygens, they GET combusted by following reactions : `C_(x)H_(y) + (x+(y)/(4)) O_(2) rarr x CO_(2) + (y)/(2) H_(2)O` |
|
| 3. |
Which of the followingis a Lewis acid ? |
|
Answer» `AlCl_(3)` |
|
| 4. |
Which of the following is correct IUPAC name for the following compound? CH_(3) - (CH_(2))_(4) - underset(underset(CH_(3))(|))(CH) - underset(underset((CH_(2))_(2))(|))overset(overset(CH_(3))(|))(C )- CH_(2) - CH_(3) |
|
Answer» 6, 7-dimethyl-7-n propylonane |
|
| 5. |
Why are carboncompounds relatively inert ? |
| Answer» SOLUTION :Carbon-carbon BOND DISSOCIATION ENTHALPY isquite HIGH. | |
| 6. |
Which of the following equilibrium will shift to right side on increasing the temperature ? |
|
Answer» `CO_((G)) + H_(2)O_((g))hArrCO_(2(g)) + H_(2(g))` In this REACTION in the forward reaction. So on increasing temperature reaction will proceed in forward DIRECTION. |
|
| 7. |
Which one of the following is non-reducting? |
|
Answer» `H_(2)S` |
|
| 8. |
Which orbit would be the first to have g subshell :- |
|
Answer» `3^(RD)` |
|
| 9. |
Which of the following are the correct axial distance and axial angles for rhombohedral system ? |
|
Answer» `a = B = C, alpha = beta = gamma != 90^(@)` |
|
| 10. |
Which of the following molecules are chiral |
|
Answer»

|
|
| 11. |
Which of the following is an oxidation and reduction reaction? |
|
Answer» `BaO_(2)+H_(2)SO_(4) to BaSO_(4)+H_(2)O_(2)` |
|
| 12. |
The volume of 2.8 g of carbon monoxide at 27^(@)C and 0.821 atm pressure is |
|
Answer» <P>30 L |
|
| 13. |
What is the maximum number of electrons that can be theoretically present in the seventh orbit? |
|
Answer» 49 |
|
| 14. |
What are degenerate orbitals ? |
| Answer» SOLUTION :ORBITALS having same ENERGY, i.e., BELONGING to the same sub-shell | |
| 15. |
Which metals are used for storage of hydrogen |
|
Answer» PD, Pt |
|
| 16. |
Write down the reactions involved during the formation of photochemical smog. |
|
Answer» Solution :When fossil fuels are burnt, a variety of pollutants are emitted into the earth.s troposphere. Two of the hydrocarbons and nitric oxide build up SUFFICIENTLY high level. A chain reaction occurs from their interaction with sunlight in which NO is converted into nitrogen dioxide `(NO_2)`. This `NO_2` in turn absorbs energy from sunlight and breaks up into nitric oxide and free oxygen atom.  Oxygen atoms are very reactive and combine with the `O_2` in air to produce ozone. `O_((g)) + O_(2(g)) HARR O_(3(g))"".........(ii)` The ozone formed in the above reaction (ii) reacts rapidly with the `NO_((g))` formed in the reaction (i) to regenerate `NO_2. NO_2` is a brown gas and at sufficiently high levels can contribute to haze. `NO_((g)) + O_(3(g)) to NO_(2(g)) +O_(2(g)) ......... (iii)` Ozone is a TOXIC gas and both `NO_2` and `O_3` are strong oxidising agents and can react with the unburnt hydrocarbons in the polluted air to produce chemicals such as formaldehyde, acrolein and peroxyacetyl nitrate (PAN). `3CH_4 + 2O_3 to underset("Formaldehdye")(3CH_2 =O) + 3H_2 O` `CH_2 = underset("Acroline")(CHCH)=O "" underset("Peroxyacetyl nitrate (PAN)")(CH_3underset(O)underset(||)C OO NO_2)` |
|
| 17. |
Which one of the following order represents the correct sequence of the increasing basic nature of the given oxides ? |
|
Answer» `MGO lt K_(2)O lt Al_(2) O_(3) lt Na_(2)O` Hence , the INCREASING correct order of basic nature is `Al_(2)O_(3) lt MgO lt Na_(2)O lt K_(2)O`. |
|
| 18. |
Thehalide that is highly soluble in H_(2)O among the following is |
|
Answer» `BeF_(2)` |
|
| 19. |
Which of the following is only acidic in nature |
|
Answer» `Be(OH)_(2)` |
|
| 20. |
Which of the following hydrocarbons has lowest dipole moment? |
|
Answer»
 Trans ISOMER have lowest DIPOLEMOMENT both dipoles are equal and OPPOSITE DIRECTION. |
|
| 21. |
When solid lead iodide is added to water, the equilibrium concentration of I^(-) becomes 2.6xx10^(-3)M. What is K_(sp) for PbI_(2) ? |
|
Answer» `2.2xx10^(-9)` Thus, `PB^(2+)` ion concentration is half of the `I^(-)` ion concentration `[I^(-)]=2.6xx10^(-3)M:.[Pb^(2+)]=1.3xx10^(-3)M` `K_(SP)=[Pb^(2+)][I^(-)]^(2)` `=(1.3xx10^(-3)) (2.6xx10^(-3))^(2)=8.8xx10^(-9)`. |
|
| 22. |
Which of the following statements are true about HCOOH ? |
|
Answer» It is a STRONGER acid than `CH_(3)COOH` |
|
| 23. |
Which alkali metal and which alkaline earth metal is radioactive ? Give their atomic numbers also . |
| Answer» SOLUTION :Fr , Z = 87 and RA , Z = 88. | |
| 24. |
When an electron revolves in a stationary orbit then |
|
Answer» it ABSORBS ENERGY |
|
| 25. |
What are the major causes of water pollution ? Explain. |
|
Answer» Solution :Major causes of water pollution are as below: (i) Pathogens : The most serious water pollutants are the disease causing agents called pathogens. Pathogens include bacteria and other organisms that enter water from domestic sewage and animal excreta. Human excreta contain bacteria such as Escherichia coli and Streptococcus faecalis which cause gastrointestinal diseases. (ii) Organic wastes : The other major water pollutant is organic matter such as leaves, grass, trash etc. They pollute water as a consequence of run off. Excessive phytoplankton growth within water is also a cause of water pollution. These wastes are biodegradable. The large population of bacteria DECOMPOSES organic matter present in water. They consume oxygen dissolved in water. The amount of oxygen that water can hold in the solution is limited. The concentration of dissolved oxygen in cold water is upto 10 ppm, whereas oxygen in air is about 200,000ppm. Because of this a moderate amount of organic matter when decomposes in water can DEPLETE the amount of dissolved oxygen. The concentration of dissolved oxygen in water is very important for aquatic life. If the concentration of dissolved oxygen of water is below 6 ppm, the growth of fish gets inhibited. Oxygen reaches water either through atmosphere or from the process of PHOTOSYNTHESIS carried out by many aquatic green plants during day light. Photosynthesis STOPS during night but the pplants continue to respire. It resulting in reduction of dissolved oxygen. Microorganisms also used dissloved oxygen for the oxidation of organic matter. So, too much of organic matter is added to water, all the available oxygen is used up. Thus, oxygen dependent aquatic life to die. Anaerobic bacteria which do not require oxygen, begin to break down the organic waste and produce chemicals which are harmful to human health. Aerobic bacteria which require oxygen degrade the organic wastes and KEEP the water depleted in dissolved oxygen. |
|
| 26. |
The sulphates of which of the following metals dissolve in water . SrSO_(4) , K_(2)SO_(4) , BeSO_(4) , Li_(2)SO_(4) , MgSO_(4) , BaSO_(4) , Na_(2)SO_(4) , CaSO_(4) , Rb_(2)SO_(4). |
|
Answer» |
|
| 27. |
Which of the following is heterocyclic compound ? |
|
Answer»
|
|
| 28. |
Write IUPAC and common name of the following: (i) CH_(2)OH- CH_(2)-CH_(2)OH (ii) CH_(2)OH- CHOH - CH_(2)OH (iii) CHO- CHO (iv) Cl- CH_(2)-COOH (v) CH_(3)COOCH_(3) (vi) CH_(3)COO CH_(2)CH_(3) (vii) HCOOH (viii) COOH-COOH (ix) CH_(2)Cl_(2) (x) CHCl_(3) (xi) CHCl_(2) C Cl_(3) |
Answer» SOLUTION :
|
|
| 29. |
Which of the following statement is correct regarding following reaction ? |
|
Answer» Major PRODUCT is endocyclic ALKENE formed ACCORDING to Saytzeff. 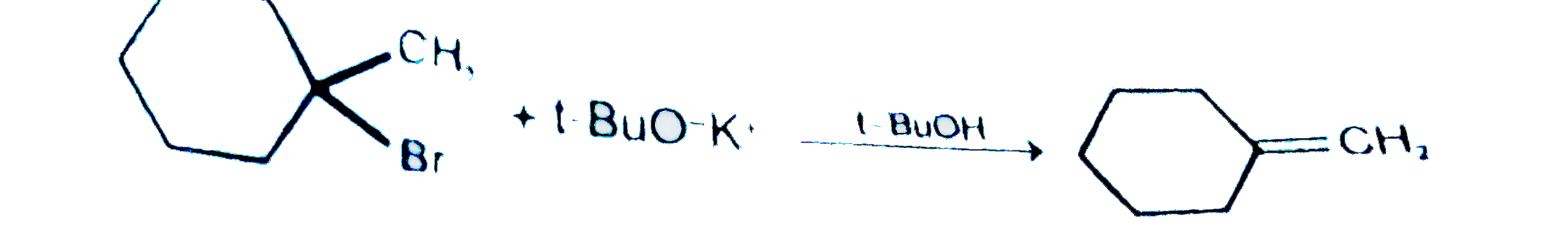
|
|
| 30. |
What causes the tempoary and permanent hardness of water ? |
| Answer» Solution :Presence of bicarbonates of calcium and magnesium , i.e., `(Ca(HCO_(3))_(2)` and `Mg(HCO_(3))_(2)` in water CAUSE temporary HARDNESS and presence of soluble chloride and sulphates of calcium and magnesium , i.e., `CaCl_(2), CaSO_(4), MgCl_(2) and MgSO_(4)` in water causes PERMANENT hardness. | |
| 32. |
Total asymmetric C present in following compound are: |
Answer» 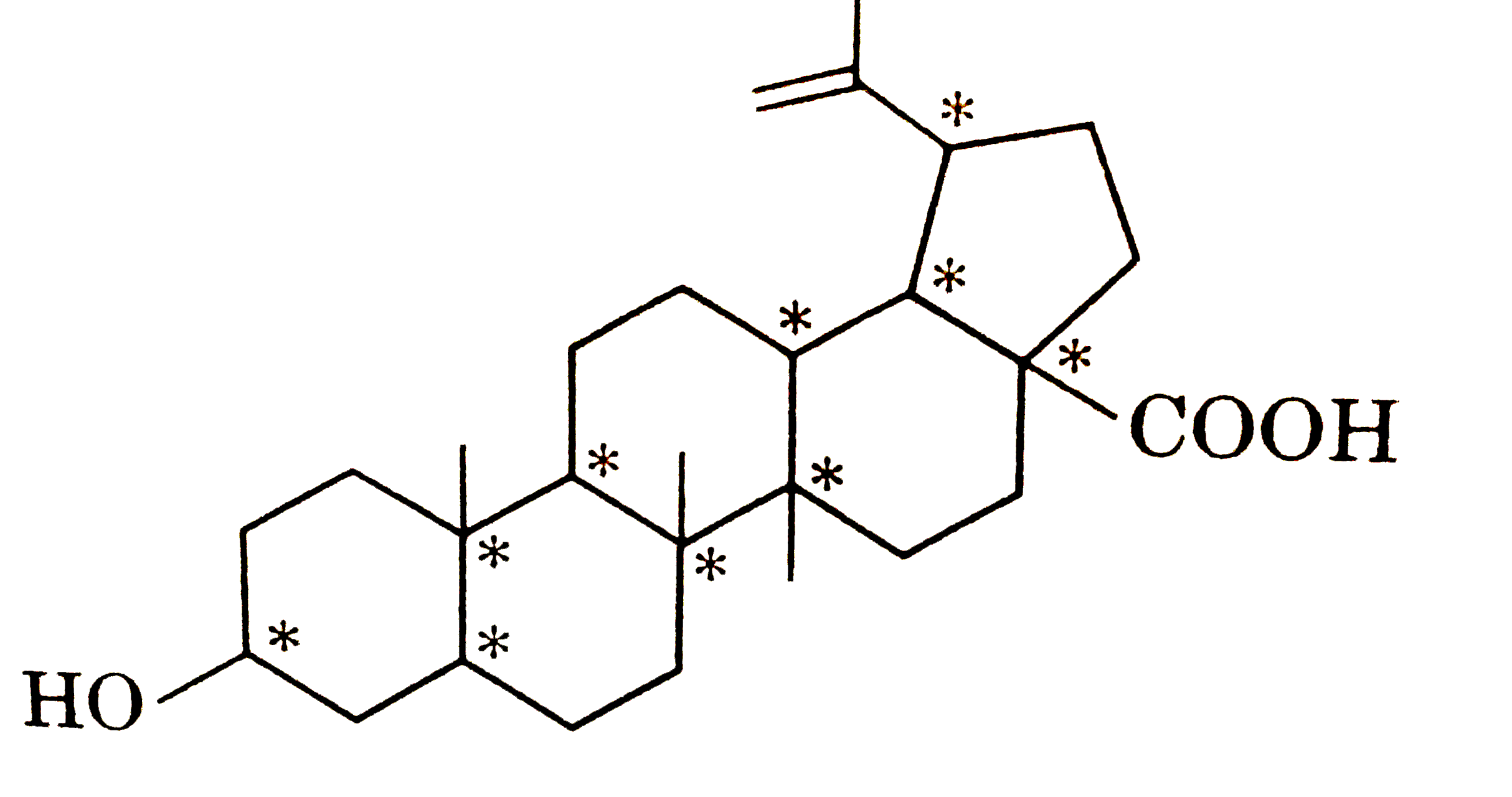
|
|
| 33. |
What is important thing to explain ionisationenthalpy ? |
|
Answer» SOLUTION : (i) Attraction between NOBLE gas and electron. (II) REPULSION between electron-electron. |
|
| 34. |
Why are ionic compounds soluble in water whereas covalent compounds of often insolube in water ? |
|
Answer» Solution :IONIC COMPOUNDS are polar in nature , SINCE water is a polar SOLVENT hence the ionic (polar ) compounds are soluble in polar solvent ( water). But organic ( covalent ) compounds are non - polar in natureand hence they are insoluble in water . |
|
| 36. |
What effects are observed if the level of CO increase in pregnant woman? |
|
Answer» PREMATURE birth |
|
| 37. |
Which of the following alkali metal ions has lowest ionic mobility in aqueous solution? |
|
Answer» `RB^(+)` |
|
| 38. |
What is meant by the term Bond order? Calculate the bond orders in the following. (a) N_(2) "" (b) O_(2)"" (c) O_(2)^(+)"" and (c) O_(2)^(-) |
|
Answer» Solution :Bond order is the MEASUREMENT of the NUMBER of electrons involved in bonds between two atoms in a MOLECULE. `B.O =(" no.of bonding electrons" - "no. of antibondlng electrons ")/(2)` |
|
| 39. |
Which element is more metallic Mg or Al and why? |
|
Answer» |
|
| 40. |
What do you mean by green chemistry? How will it help decrease environmental pollution ? |
|
Answer» Solution :Self-sufficiency in food has been achieved in India since late `20^(th)` century by using fertilizers and pesticides and exploring improved methods of farming, good quality seeds, irrigation etc. Over exploitatoin of soil and excessive use of fertilizers and pesticides have resulted in the deterioration of soil, water and air. Green chemistry is a way of thinking and is about utilising the existing knowledge and principles of chemistry and other SCIENCES to reduce the adverse impact on environment. Green chemistry is a production process that would bring about minimum pollution or deterioration to the environment. The byproducts generated during a process, if not used gainfully, add to the environmental pollution. Such processes are not only environmental unfriendly but also cost-ineffective. The waste generation and its disposal both are ECONOMICALLY unsound. Organic solvents such as benzene, TOLUENE, carbon tetrachloride etc., are highly toxic. One should be careful while using them. Any reaction depends upon physical parameters like temperature, pressure and use of CATALYST. If reactants are fully converted into useful environmental friendly products then there would be no chemical pollutants introduced in the environment. During a synthesis, care must be taken to choose starting MATERIALS that can be converted into end products with yield approximately upto 100 per cent. This can be achieved by arriving at optimum conditions of synthesis. Synthesis reactions is carryout in aqueous medium since water has high specific heat and low volatility. Water is cost effective, noninflammable and devoid of any carcinogenic effects. |
|
| 41. |
Which has p_(pi) - d_(pi) bonding ? |
|
Answer» `NO_(3)^(-)` bonds involves p-orbital of O and d-orbitals of S SUPPLEMENT Your Knowladge, All others INVOLVE only `p_(pi)-p_(pi)` bonding . |
|
| 42. |
Which of the following statements are correct about the kinetics of this reaction ? |
|
Answer» The RATE of reaction DEPENDS on the concentration of only (II). |
|
| 43. |
Write the reaction to convert propyne to 2,2 - dibromopropane. |
|
Answer» Solution :When PROPYNE is TREATED with HBR, it undergoes Markownikoff RULE by adding across triple bond to form 2,2 - dibromopropane. ` CH_(3) - underset("Propyne")(C equiv CH + HBr) RARR CH_(3)- underset(Br)underset(|)(C ) = CH_(2) underset(M - Rule)overset(HBr)(rarr) CH_(3) - underset(2,2 - "didromopropane")underset(Br)underset(|)overset(Br)overset(|)(C) - CH_(3)` |
|
| 44. |
What will be the pH of a equimolar mixture of CH_3COOH and NaOH solution ? |
| Answer» Answer :D | |
| 45. |
Which is a correct name according to IUPAC system ? |
|
Answer» 2, 3-Dimethylhexane The given IUPAC name is correct. |
|
| 46. |
What will be normality of the solution of the mixture of 40 mL, 0.2 M H_(2)SO_(4(aq)) and 60 mL 0.3 M H_(2)SO_(4(aq)) ? |
|
Answer» 0.26 N `= ((40xx0.2) + (60xx0.3))/(100)` `= (8.0+ 18.0)/(100)` `= 0.26M` Where : `{:(M_(1)=0.2M),(V_(1)=40ML):}}H_(2)SO_(4)` `{:(M_(2)=0.3M),(V_(2)=60mL):}}H_(2)SO_(4)` `v_(3)=(60+40)mL` `= 100 mL` `M_(3)=` Morality of mix solution ` 0.26=` Molarity of mixed `H_(2)SO_(4)` solution MOLECULAR mass of `H_(2)SO_(4) = 2(H) + S + 4 (O)` `= 2(1) + 32+4(16)` `= 98g*MOL^(-1)` BASICITY of `H_(2)SO_(4) = 2= ` no. of `H^(+)` `:.` Equivalent wt. of `H_(2)SO_(4)= 98//2=49` Normality `=("Molarity" xx "Molecular weight")/("Equivalentweight")` `= 0.26xx98)/(49) = 0.52 N H_(2)SO_(4)` |
|
| 47. |
Weight of H_(2)O_(2) present in 560 ml. of 20 vol . H_(2)O_(2) solution is approximately |
|
Answer» 69g |
|
| 48. |
Which of the following configuration is not possible ? |
|
Answer» `2p^(2)` |
|
| 49. |
What is oxidised and what is reduced in the reaction H_2S+Cl_2 to 2HCl+S? |
| Answer» SOLUTION :`H_2S` is oxidised to S and `Cl_2` is REDUCED to HCL. | |








