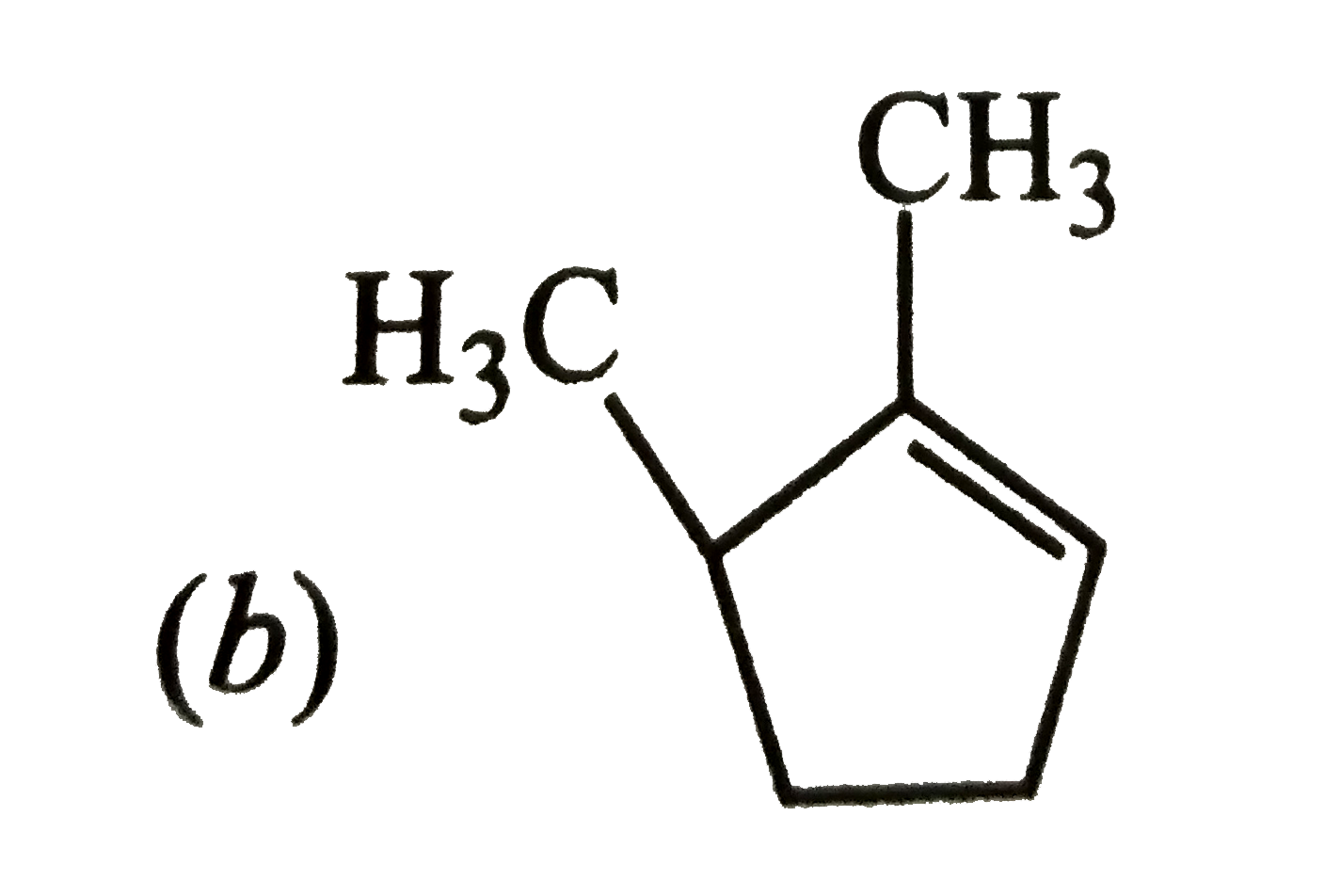Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which of the following ions are not isoelectronic with Ar? |
| Answer» Answer :a | |
| 2. |
Titration curves for 0.1M solution of three weak acids HA_1,HA_2 and HA_3 with ionization constants K_1, K_2 and K_3 respectivelyare plotted as shown in the figure. Which of the following is/are true?(##AKS_ELT_AO_CHE_XI_V01_B_C05_E02_135_Q01.png" width="80%"> |
|
Answer» `K_2 =(K_1+K_3) //2` ACID strength ` ALPHA (1)/( pH)` |
|
| 3. |
What are abnormal colligative properties? |
| Answer» Solution :The experimental values of COLLIGATIVE properties in most of the cases RESEMBLE closely to those OBTAINED theortically by their formula. However in some cases experimental values of colligative properties differ widely form those obtained theortically. Such experimental values are REFFERED to as abnormal colligative properties. | |
| 4. |
Which one is polyprotic acid : |
|
Answer» `CH_(3)COOH` |
|
| 5. |
Which one among the following is the standard for atomic mass? |
|
Answer» `""_(1)H^(1)` |
|
| 6. |
Which of the following compounds contain after the carbon atoms in the same hybridisation state? |
|
Answer» `H-C -= C -C -= C-H`   `because` EVERY CARBON have one DOUBLE bond `because` EVERY CARBON have one DOUBLE bond
|
|
| 7. |
Which of the following compound has maximum volatility- |
|
Answer»

|
|
| 8. |
What is the composition of the compound formed by the reaction of carbon with fluorine ? |
| Answer» SOLUTION :`(CF)_n` | |
| 9. |
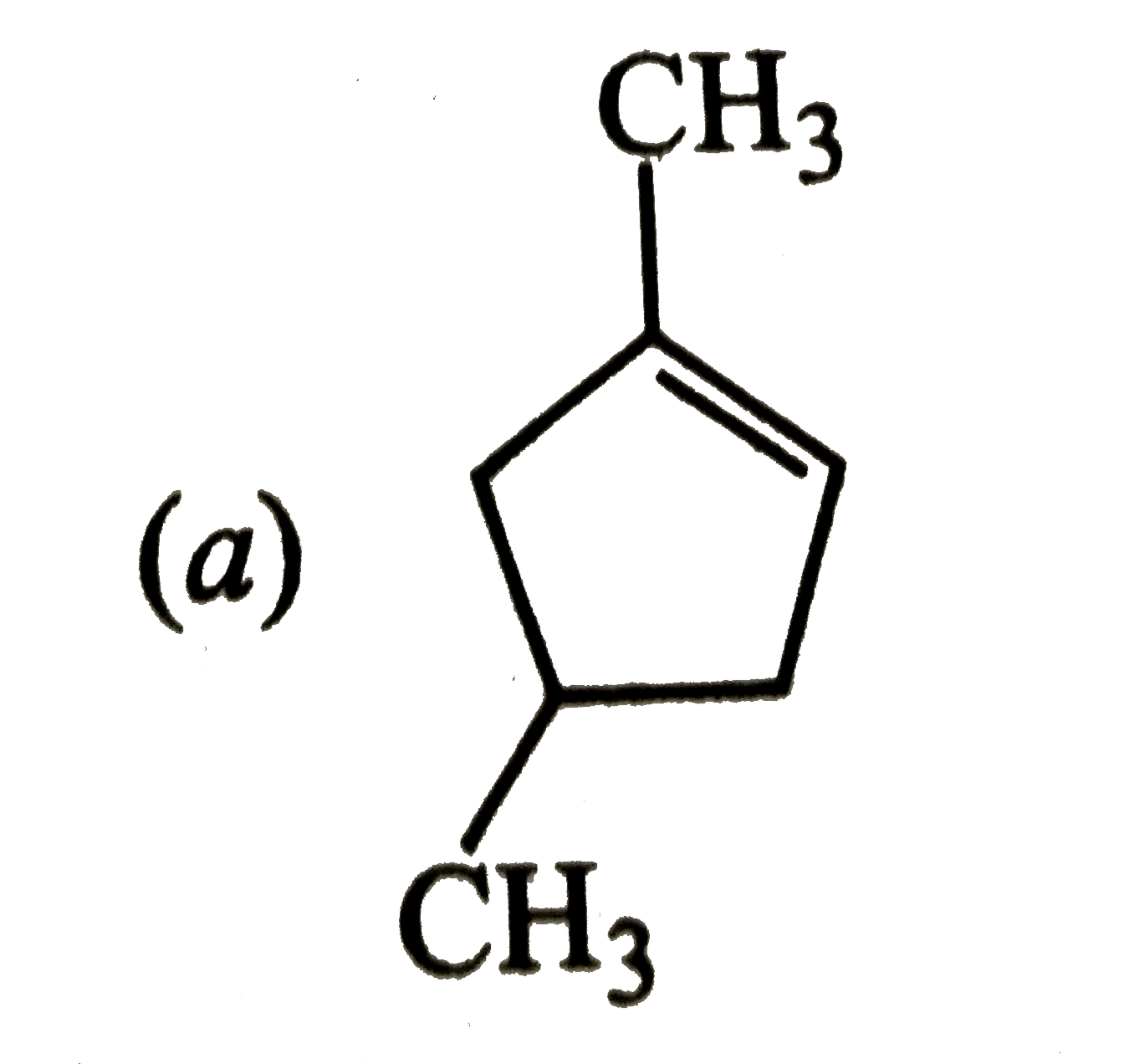
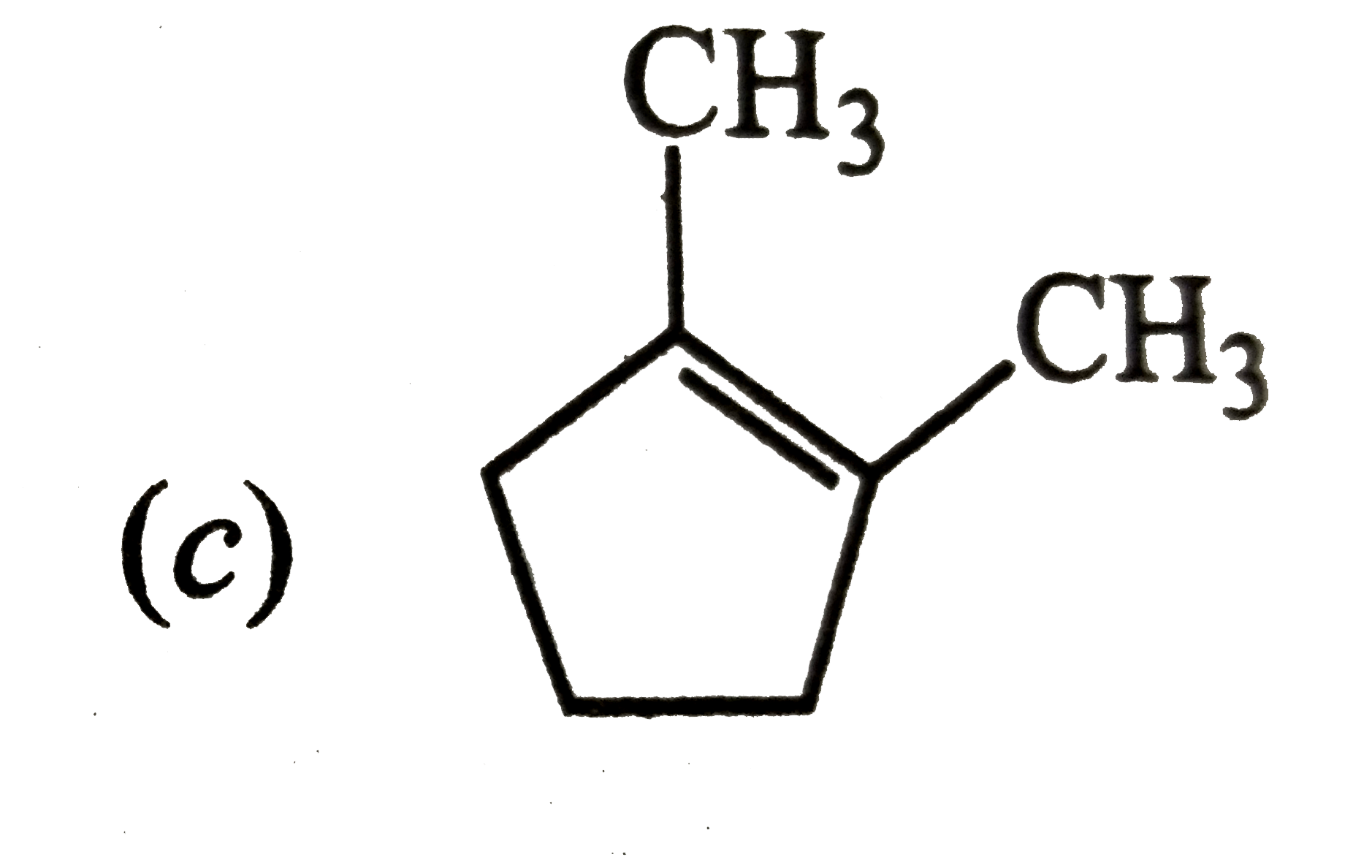
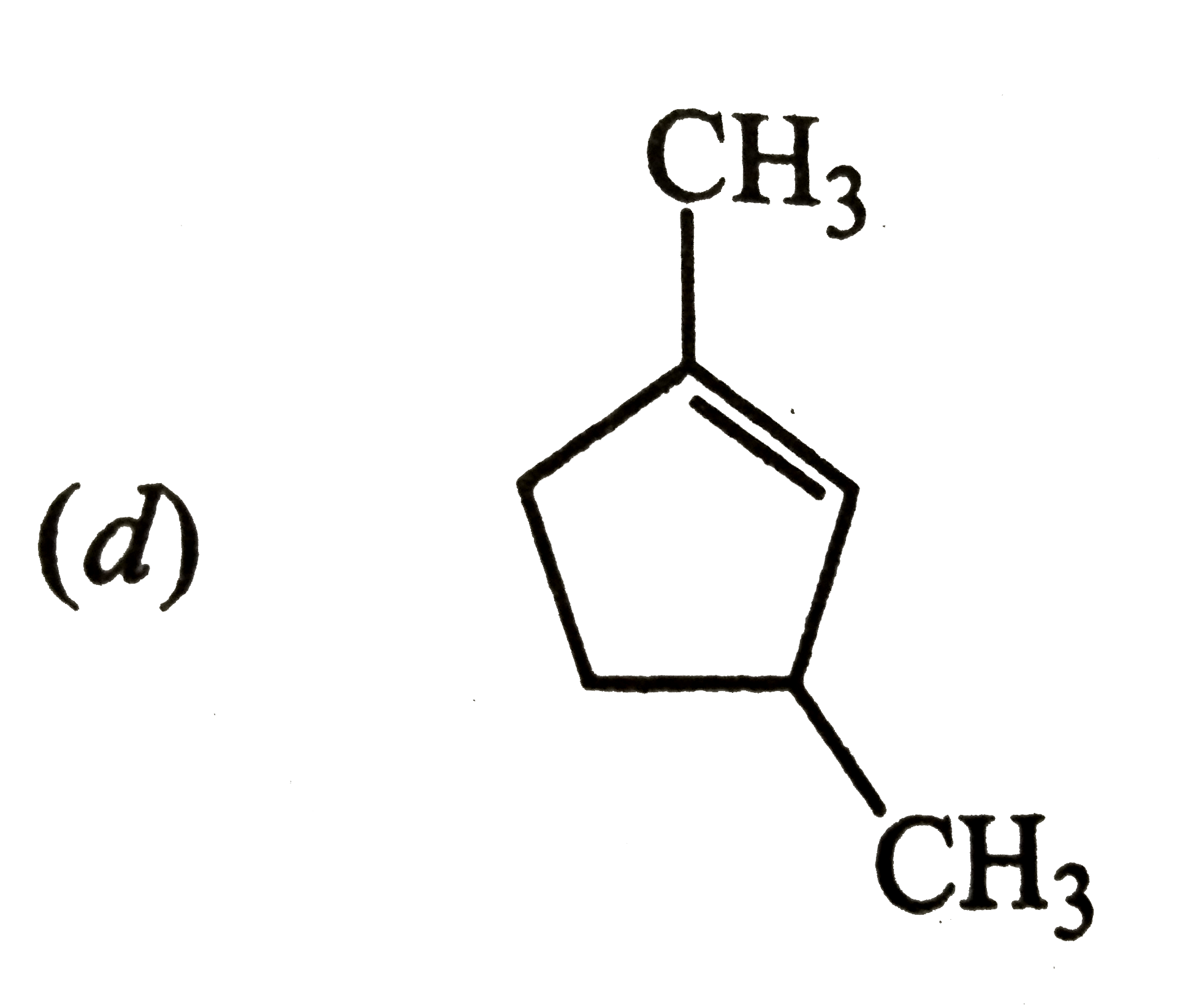
Which of the following is the most stable cycloalkane? |
|
Answer»
|
|
| 10. |
Which of the following is the strongest oxidising agent ? |
|
Answer» `F_(2)`<BR>`Cl_(2)` |
|
| 11. |
Which is the shape of carbocation? |
|
Answer» Planar |
|
| 12. |
Which of the following orbital(s) lie in the xy-plane? |
|
Answer» `d_(x^(2)-y^(2))` |
|
| 13. |
Which are important ingredients present in Portland cement ? |
| Answer» Solution :Important ingredients present in Portland cement are dicalcium silicate `(Ca_(2)SiO_(4))` 26%, tricalciumsilicate`(Ca_(3)SiO_(5)) 51%` and tricalciumaluminate `(Ca_(3)Al_(2)O_(6))`11%. | |
| 14. |
Which of the following compounds exhibits stereo isomerism |
|
Answer» 2-methyl butene-1  2-methyl butanoic acid has ONE CHIRAL CENTRE. So it exhibits STEREO isomerism |
|
| 15. |
Which one of the following combinations will give the highest stability to a nucleus with atomic number Z and mass number N |
|
Answer» Even Z and odd N |
|
| 16. |
What are hybridisation states of each carbon atom in the following compounds? CH_(2)=C=O, CH_(3)CH=CH_(2), (CH_(3))_(2)CO, CH_(2)=CHCN, C_(6)H_(6) |
|
Answer» Solution :`overset(SP^(2))CH_(2)=overset(sp)C=O` `overset(sp^(3))CH_(3)-overset(sp^(2))CH=overset(sp^(2))CH_(2)` `overset(sp^(3))CH_(3)-underset(sp^(2))overset(O)overset(||)C-overset(sp^(3))CH_(3)` `overset(sp^(2))CH_(2)=overset(sp^(2))CH-overset(sp)C=N` 
|
|
| 17. |
When H_(2)O_(2) is added to ice cold solution of acidified potassium dichromatic in ether and the contents are shaken and allowed to stand. |
|
Answer» a blue COLOUR is obtained in ether due to formation of `Cr_(2)(SO_(4))_(3)` |
|
| 18. |
What happends when : (a) Aqueous solution of lead acetate and sodium sulphide are mixed with each, (b) A few drops of sodium nitroprusside are added to the aqueous solution containing sodium suphide. (c) An aqueous solution containing sodium iodide is treated with a few drops of CS_(2) followed by excess of chlorine water. (d) Phoshorus is fused with sodium peroxide |
|
Answer» Solution :(a) A black precipitate of lead sulphide is formed. `Na_(2)S + underset("Lead acetate")((CH_(3)COO)_(2) Pb rarr underset(("Black ppt")) (PbS )+ underset("Sod. Acetate")(2CH_(3)COONA)` (B) The solution acquires a violet colour. `Na_(2)S + underset("Sod. nitroprusside")(Na_(2) [Fe(CN)_(5)NO]) rarr underset(("violet colour"))(Na_(4) [Fe(CN)_(5) NOS])` (c) The oily layer of `CS_(2)` acquires a violet colour. chlorine being more reactive than iodine displaces the same from the salt solution `2NaI + Cl_(2) rarr 2NaCl + I_(2)` (d) Sodium phosphate is formed on fusion. `5Na_(2) O_(2) + 2P overset(("Fuse"))rarr underset("Sod. phosphate")(2Na_(3)PO_(4) +2Na_(2)O)` |
|
| 19. |
Which one of the following is a gas |
|
Answer» `BF_(3)` |
|
| 20. |
What is the ionic product Q_(sp) ? Explain by example. |
|
Answer» Solution :When the solution of two sparingly SOLUBLE salt mix together than the product of IONS of sparingly soluble salt is called solubillish product. If `AgNO_3` and NaCl mix together `Q_(sp)=[AG^+][CL^-]` `[Ag^+]=AgNO_3` `[Cl^-]=NaCl` |
|
| 21. |
Which of the following orbitals are degenerate ? 3d_(xy), 4d_(xy), 3d_(z^(2)), 3d_(yz) , 4d_(yz) ,4d_(z^(2)) |
| Answer» Solution :`3d_(xy) , 3d_(Z2), 3d_(YZ)` and `4d_(xy) , 4d_(yz) , 4d_(z2)` | |
| 22. |
Use Markovnikov's rule to predict the product of the following reactions : (i)HCl with CH_3 C Cl=CH_2and (ii)HCl with CH_3CH=C (CH_3)_2 |
| Answer» SOLUTION :(i)`CH_3 C CL=CH_2+HCL to CH_3C Cl_2CH_3` (ii) `CH_3CH=C (CH_3)_2 + HCl to CH_3CH_2 -C Cl(CH_3)_2` | |
| 23. |
Which of the following will produce aromatic compound as product ? |
|
Answer» `CH_3 - C -= C underset(Delta)OVERSET("Red HOT FE TUBE")to` 
|
|
| 24. |
WhichiondoesnotposseselectroncofigurationlikeNe? |
|
Answer» `Na^(+)` `F^(-)to (9+1) = 10 `electrons `Mg^(2+)to (12- 2) = 10`electron `CI^(-) to (17+ 1)= 18`electrons |
|
| 25. |
What is the hybrid state of carbon in ethyne, graphite and diamond ? |
| Answer» SOLUTION :`sp^2 , sp^3` | |
| 26. |
Which of the following statement 1s wrong about electrons ? |
|
Answer» it is a particle |
|
| 27. |
What volume of 3 molar HNO_3is needed to oxidize 8g of Fe^(2+)"to " Fe^(+3) ? HNO_3 , gets converted to NO |
|
Answer» SOLUTION :eq. `HNO_3 =Fe^(2+)` `V_1N_1= V_1N_1` `V_1M_1n_1 = W/Exx1000` `V xx 3 xx 3 = (8)/((56)/1) xx1000` `V=(Fxx1000)/(56xx9)=15.8`ml ( `~~16` ml) = 15.8 ml |
|
| 28. |
What is the molecular mass of water? |
|
Answer» Solution :Molecular MASS =Real mass of one molecular `6.022 XX 10^(23)` water molecules=18g One water molecular =? Mass of a molecule `=(18)/(6.022 xx10^(23))=2.99 xx 10^(-23)g` `=2.99 xx 10^(-26)kg` |
|
| 29. |
Which laws give information about Dalton's atomic theory ? According to Dalton's atomic theory which other laws give information about atomic principle. |
|
Answer» Law of definite PREPARATION. Law of MULTIPLE proportion and Avogradro law give explanation of Dalton.s atomic THEORY. |
|
| 30. |
Which isotope of hydrogen emitted beta-particle ? |
| Answer» Solution :The TRITIUM isotope of HYDROGEN emitted the `BETA` -particles. | |
| 31. |
Which of the following molecule contain no pi bond? |
|
Answer» `SO_(2)`  Water `(H_(2)O)` contains only `sigma` bonds and no `PI` bonds. |
|
| 32. |
Three solutions eaach of 100 mL containing 0.4 M As_(2)S_(3),5M NaOH and 6MH_(2)O_(2), respectively were mixed to form AsO_(4)^(3-) and SO_(4)^(2-) as products. Q. When the above solution is allowed to stand for some time what volume of O_(2) will be obtained STP? |
|
Answer» 0.112 L `underset(40)(H_(2)O_(2)tounderset(0)(H_(2)O)+(1)/(2)underset(0)(O_(2))` `{:(mmol es),(of H_(2)O_(2)),(l eft)]:}-(1)/(2)xx40=20m" mol of "O_(2)` THUS, mmol es of O_(2)formed`=40xx(1)/(2)=20` Volume of `O_(2)` at `STP=20xx10^(-3)xx22.4L` `(because1 " mol of "O_(2) at STP=22.4L)` `=0.448L` |
|
| 33. |
The value of the gas constant R is |
|
Answer» `0.082dm^(2)ATM` |
|
| 34. |
Three solutions eaach of 100 mL containing 0.4 M As_(2)S_(3),5M NaOH and 6MH_(2)O_(2), respectively were mixed to form AsO_(4)^(3-) and SO_(4)^(2-) as products. Q. Percentage strength of the H_(2)O_(2) solution left after reaction is |
|
Answer» `0.017%` `N of H_(2)O_(2)` left`=(2XX2)/(15)=(4)/(15)N` `1N of H_(2)O_(2)=(1eq)/(L)H_(2)O_(2)=5.6V` (Volume STRENGTH)`=1.7%` `(4)/(15)NH_(2)O_(2)=1.7xx(4)/(15)=0.452%` |
|
| 35. |
Which compound would give 5-keto-2-methylhexanal on ozonolysis ? |
|
Answer»
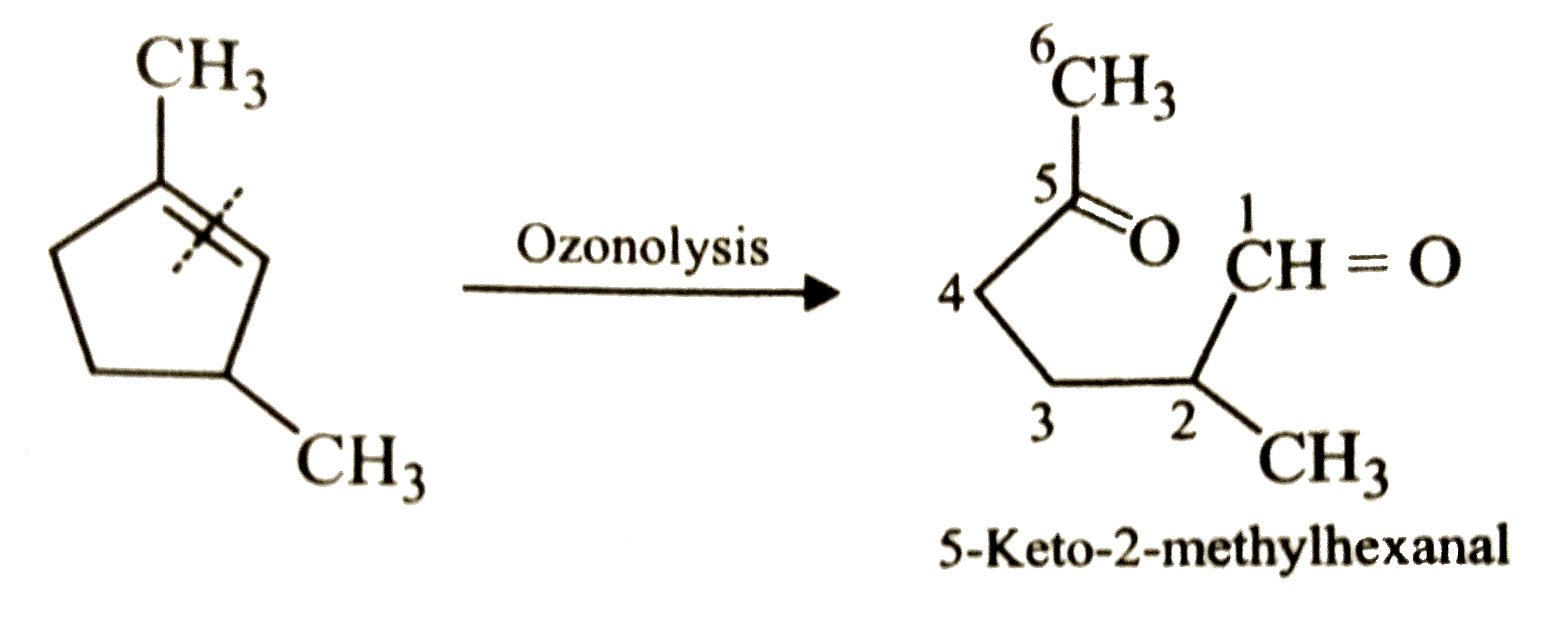
|
|
| 36. |
Which of the following can form cis-transisomers |
|
Answer» `C_(2)H_(5)Br` |
|
| 37. |
Which of the following statements is true about Ca(OH)_(2) ? |
|
Answer» It is used in the preparation of BLEACHING powder |
|
| 38. |
What is the value of bond angle inPCl_(5) ? |
|
Answer» <P> SOLUTION :There are three P - CL are in one plane. Its angleCl - P - Cl is `120^(@)`. Two P - Cl is at `90^(@)` . |
|
| 39. |
What is used for drying alcohol and non-acidic gases and why? |
| Answer» SOLUTION :CALCIUM is used for drying ALCOHOL and non-acidic gases bacause Ca does not REACT with alcohol. | |
| 40. |
Write the structural isomers of an alkene having M.F. C_4H_8. How can you distinguish between them ? |
Answer» SOLUTION :`C_4H_8` has three structural isomers : 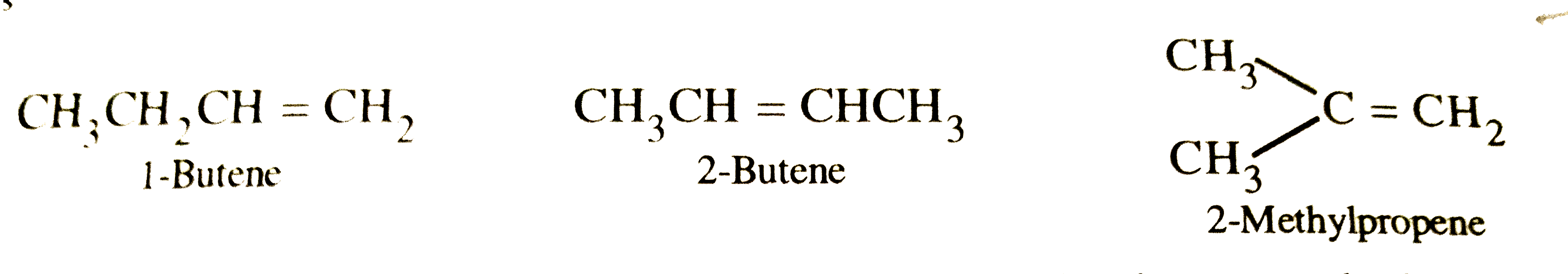 These can be easily distinguished on the BASIS of their REDUCTIVE OZONOLYSIS products because each of them gives different products : 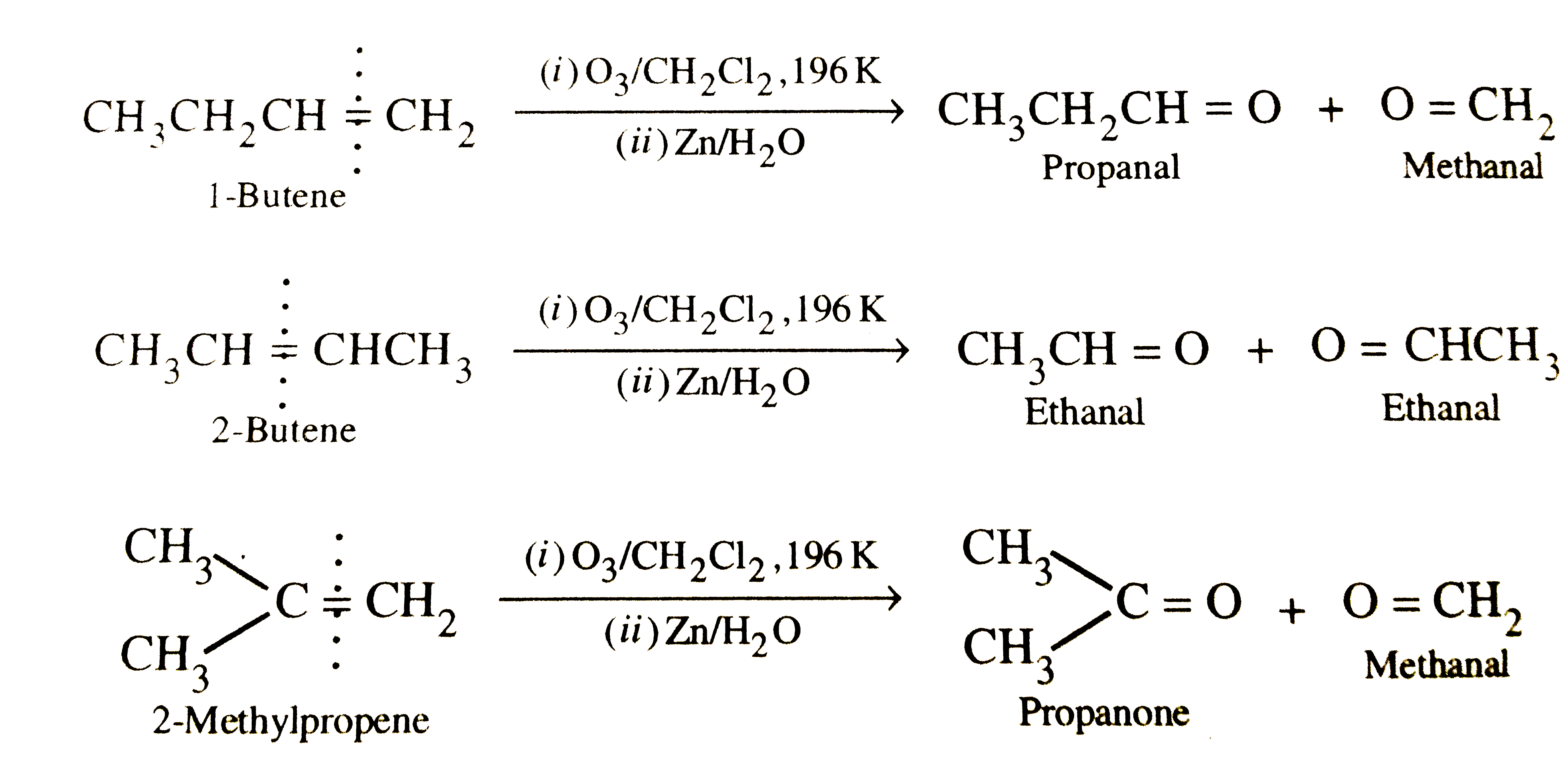
|
|
| 41. |
Which of the following equation is known as Van der Waal.s equation ? |
|
Answer» `(PV)/(K)=NRT` |
|
| 42. |
What is Ritz combination principle ? |
| Answer» SOLUTION :`BAR(v)= R ((1)/(n_(1)^(2)) - (1)/(n_(2)^(2)))` where R is called RYDBERG constant | |
| 43. |
Which of thefollowingordersaboutionizationenergyis correct? |
|
Answer» `N gt O gt F ` |
|
| 44. |
Which one of the following is more basic? |
| Answer» Answer :C | |
| 45. |
What are X and Y in the following reactions ? |
Answer» SOLUTION :After the REARRANGEMENT the FOLLOWING PRODUCTS are FORMED. 
|
|
| 46. |
What is bond stabilising energy ? |
|
Answer» |
|
| 47. |
What is the relationship between pK_(ln) and pH at the equivalence point ? |
| Answer» SOLUTION :`pK_(LN) ` = pH at the EQUIVALENCE point. | |
| 48. |
Which of the following come under the view of thermodynamics? |
|
Answer» Predicting the feasibility of chemical CHANGE |
|
| 49. |
Which of the following will have a meso-isomer also ? |
|
Answer» 2-Chlorobutane  2, 3-dichlorobutane have MESO ISOMER due to the presence of plane of SYMMETRY. |
|
| 50. |
Which of the following compound is (meso) compounds? |
|
Answer»

|
|