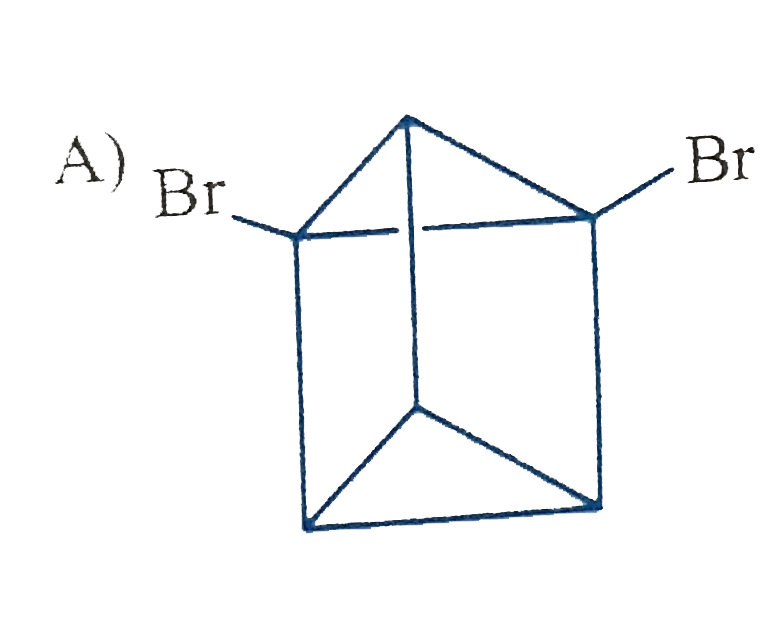Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which is considered to be earth's protective umbrella' Why? |
|
Answer» Solution :(i) At high altitudes in the atmosphere CONSISTS of a layer of ozone `(O_3)` which ACTS as an umbrella for harmful UV radiations. Ozone is considered to be earth.s PROTECTIVE umbrella. (ii) It protects US from harmful effects ofUV-radiations of the SUN such as skin cancer. (iii) Ozone layer prevent the UV radiations to reach the earth surface. So it acts as an umbrella for the Earth. |
|
| 2. |
There are two types of iodine titrations (a) Iodometric & (b) Iodimetric, Iodometric method is indirect method of I_(2) estimation. Any oxidant which liberates I_(2) from KI solution, the liberated iodine is estimated by titrating it with Na_(2)S_(2)O_(3) solution as : I_(2)+2Na_(2)S_(2)O_(3)rarr2NaI+Na_(2)S_(4)O_(6) 100 mL of .x. M K_(2)Cr_(2)O_(7)solution is added to excess of KI solution in acidic medium. The liberated iodine required 50 mL of 0.1N Na_(2)S_(2)O_(3) solution. The value of x is |
|
Answer» `21.58%` `PXX(3)/(249.5)xx1=(10)/(1000)xx0.5xx1=0.4158` implies % Purity = 41.58% |
|
| 3. |
Which one of the following is the likely bond angles of sulphur tetrafluoride molecule? |
|
Answer» `120^(@),80^(@)` |
|
| 4. |
Which one of the following alkali metals gives hydrated salts ? |
|
Answer» Li |
|
| 5. |
The strength of elctrolytes is expressed in terms of degree of dissociation alpha For strong electrolyte alpha is close to oneand for weak electrolytes alpha is quite small. According to Ostwald Dilution Law alpha=sqrt((K)/(C)) For an acid [H^(+)] = sqrt(K_(a)C) For a base [OH^(-)] =sqrt(K_(b)C The relative strengths of acids or bases can be compared in terms of the square roots of their K_(a) " or " K_(b) values. At infinite dilution , the percentage ionisation of both strong and weak electrolytes is : |
| Answer» SOLUTION :Both strong and weak acidsare almost COMPLETELY ionised at infinite dilution. | |
| 6. |
The strength of elctrolytes is expressed in terms of degree of dissociation alpha For strong electrolyte alpha is close to oneand for weak electrolytes alpha is quite small. According to Ostwald Dilution Law alpha=sqrt((K)/(C)) For an acid [H^(+)] = sqrt(K_(a)C) For a base [OH^(-)] =sqrt(K_(b)C The relative strengths of acids or bases can be compared in terms of the square roots of their K_(a) " or " K_(b) values. The dissociation constant of monobasic acids A,B and C are 10^(-4), 10^(-6) " and " 10^(-10) respectively. The concentration of each is 0.1 M . Which is correct order or their pH values ? |
|
Answer» `A LT B lt C` |
|
| 7. |
Which of the following statements regarding van der Waals' constants a and b is not correct? A. The constant a is a measure of van der Waals' forces. B. A gas with a lower value of a possesses a greater tendency to get liquefied. C. b is a measure of effective size of molecules. D .b is the excluded volume per mole. |
|
Answer» The constant a is a MEASURE of VAN der WAALS' forces. |
|
| 8. |
What is the mass in grams of a chlorine atom, Cl? |
|
Answer» SOLUTION :The atomic weight of Cl is 35.5 AMU, so the MOLAR mass of Cl is 35.5 g/mol. DIVIDING 35.5 g ( per mol) by ` 6.023 xx 10^(23)` gives the amss of one atom. Mass of a Cl atom`=(35.5g)/(6.023 xx 10^(23))` ` = 5.90 xx 10^(-23) g ` |
|
| 9. |
Write the alkenes that give the following compounds on ozonolysis: (i) CH_(3)COCH_(2)CH_(3) + CH_(3)CHO (b) Hunderset(O)underset(||)CCH_(2)CH_(2)CH_(2)underset(O)underset(||)CCH_(2)CH_(2)CH_(3) (c)CH_(3)-underset(O)underset(||)CCH_(2)CH_(2)underset(O)underset(||)CH+Hunderset(O)underset(||)CH+CH_(3)CHO |
Answer» Solution :`CH_(3)CH_(2)UNDERSET(CH_(3))underset(|)C+ O = underset(H)underset(|)C-CH_(3) underset(ZN, H_(2)O)overset(O_(3))larr CH_(3)CH_(2)underset(CH_(3))underset(|)C =CHCH_(3)` 
|
|
| 10. |
Which is the conjugate acid of weak base NH_3 ? |
|
Answer» Solution :The CONJUGATE ACID of `NH_3` is `NH_4^+` `NH_4^+` is a strong because it conjugate acid of WEAK base. `NH_4^(+) + H_2O HARR NH_3 + H_3O^+` `NH_4^+` is strong acid than `H_2O`. |
|
| 11. |
Which of the following is correct regarding the Lewis concept of acids and bases ? |
|
Answer» It cannot explain the cases when a species is donating as WELL as accepting electron pairs. |
|
| 12. |
When phenolphthalein is used as the indicator in a titration of an HCl solution with a solution of NaOH, the indicator undergoes a colour change from colourless to poink at the end point of the tiration. This colour change occurs abruptly because |
|
Answer» Phenolphthalein is a very strong acid that is capable of rapid dissociation |
|
| 13. |
Which of the following hydrocarbons is not formed when Wurtz reaction takes place between ethyl iodide and propyl iodide? |
|
Answer» Butane |
|
| 14. |
Which is not the characteristic of Pi- bond |
|
Answer» pi bond are formed when a SIGMA bond already exists |
|
| 15. |
Thetype of substituion reactions of benzenoid hyderocarbons are : |
|
Answer» elemination |
|
| 16. |
What are Psi and Psi^(2) ? |
|
Answer» SOLUTION :(i) `Psi`itself has no physical MEANING but it represents an atomic orbital. (II) `Psi^(2)` is related to the probabilty of fiding the electrons within a GIVEN volume of space . |
|
| 17. |
The type of H-bonding present in ortho nitro phenol and p-nitro phenol are respectively |
|
Answer» inter MOLECULAR H-bonding and |
|
| 18. |
What happens when Aluminium is treated with dilute NaOH |
|
Answer» Solution :AL reacts with dilute NAOH to form sodium tetrahydroxo aluminate(III). Hydrogen gas is liberated in the process. `2Al_((s)) + 2NaOH_((AQ)) + 6H_2O_((l)) to 3H_(2(g)) + 2Na^(+) [Al(OH)_4]_((aq))^(-)` |
|
| 19. |
What are the possible isomers for the formula C_(4)H_(9)Cl ? Give their structures and IUPAC names. |
|
Answer» Solution :`C_(4)H_(9)Cl` (i) `CH_(3)-CH_(2)-CH_(2)-CH_(2)Cl` 1-chloro BUTANE (II) `CH_(3)-CH_(2)-UNDERSET(Cl)underset(|)(CH)-CH_(3)` 2-chloro butane (iii) `CH_(3)-underset(CH_(3))underset(|)(CH)-CH_(2)Cl` 1-chloro-2-methyl PROPANE (iv) `CH_(3)-underset(Cl)underset(|)overset(CH_(3))overset(|)C-CH_(3)` 2-chloro-2methyl propane |
|
| 20. |
Which question represents charge because equation for the solution of H_(2)S in water? |
|
Answer» `2[H^(+)]=[S^(-2)]+[HS^(-)]+[OH^(-)]` |
|
| 21. |
Which of the following are the correct reasons for anomalous behaviour of lithium ? |
|
Answer» Exceptionally SMALL SIZE of its atom |
|
| 22. |
When burning magnesium splinter is placed in a jar containing the following gases then it continues to burn in |
|
Answer» `N_(2)` |
|
| 23. |
Two flasks A and B have equal volume. Flask A contains hydrogen at 300 K while flask B has an equal mass of CH_(4) at 600 K. which flask contains larger number ofmoleculars? b. In which flask is the pressure greater and by how many times? |
|
Answer» Solution :a. A has larger NUMBER of molecules B. pressure is higher in B C. molecules move faster in B d. B. |
|
| 24. |
Which of the following statements are correct. Answer on the basis of Fig. given below. |
|
Answer» The two bridged HYDROGEN ATOMS and the two boron atoms lie in ONE PLANE. 
|
|
| 25. |
What is the difference between a quantum and a photon ? |
| Answer» Solution :The SMALLEST packet of energy of any radiation is CALLED a quantum whereas that of LIGHT is called PHOTON | |
| 26. |
When conc. H_(2)SO_(4) was added into an unknown nitrate salt present in a lest-tube, a brown gas (A) was evolved. This gas intensified when copper turnings were also added into this test-tube. On cooling, the gas (A) changed into a colourless gas (B). (a) Identify the gases (A) and (B). (b) Write the equations for the reactions involved. |
|
Answer» Solution :When a nitrate salt is heated with conc. `H_(2)SO_(4)`, vapours of `NHO_(3)` are first produced which undergo decomposition to produce nitrogen dioxide `(NO_(2))` which is a brown GAS (A). When Cu turnings are added, the gas (A) intensified due to the reduction of `HNO_(3)` by Cu. `underset("Sod. nitrate")(2NaNO_(3))+H_(2)SO_(4)overset("Heat")tounderset(("Colourless"))underset("Nitcid acide")(2HNO_(3))+Na_(2)SO_(4)` `4HNO_(3)overset("Heat")tounderset("Brown gas A")underset("Nitrogen dioxide")(4NO_(2))+O_(2)+_2H_(2) O` `underset("Copper turnings")(Cu)+4HNO_(3)overset("Heat")toCu(NO_(3))_(2)+underset("Nitrogen dioxide(A)")(2NO_(2))+2H_(2)O` `NO_(2)` is an ODD electron species (7+8+8=23 electrons). In the valence shell, it has only 7 elecrons and hene is less stable. To become more stable by having 8 electrons in the valence shell, it undergoes dimerization to form a colourless gas, `N_(2)O_(4)`.  THUS, (A) is nitrogen dioxide `(NO_(2))` and (B) is dinitrogen tetroxide `(N_(2)O_(4))`. |
|
| 27. |
Which of the following species has the shortest bond length ? NO, NO^(+), NO^(2+),NO^(-) |
|
Answer» Solution :The electronic configurationsand the bond orders of these species are as follows : (NO has7 + 8 = 15 electrons) `NO: KK sigma_(2s)^(2)sigma_(2s)^(**2)sigma_(2p_(z))^(2)pi_(2p_(x))^(2) pi_(2p_(x))^(**1) ""` Bond order `= (1)/(2) (8-3) = 2.5 ` `NO^(2+)` : One electron is removed from `pi_(2p_(x)) ^(**)` of NO, Bond order =` (1)/(2) (8-4) = 2 ` Thus , ` NO^(+)` has the highest bond order and , therefore, the SHORTEST bond length . |
|
| 28. |
What is the of hydrogen peroxide in 1L of 3M solution |
|
Answer» 10.2 g |
|
| 29. |
Why are potassium and caesium, rather than lithium used in photochemical cells ? |
|
Answer» Solution :(i)Due to low ionisation energy K and Cs eject. electrons when exposed to light. (II) Lithium which has highest ionisation enthalpy AMONG alkali meta LS cannot be used in photo ELECTRIC cells because it does not RELEASE electrons when exposed to light |
|
| 30. |
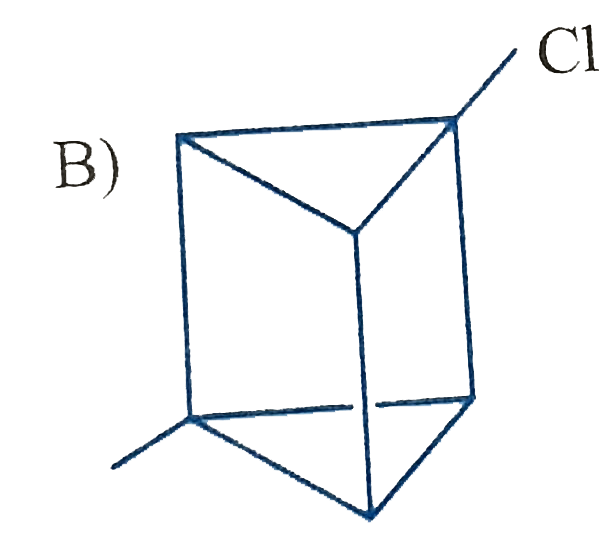
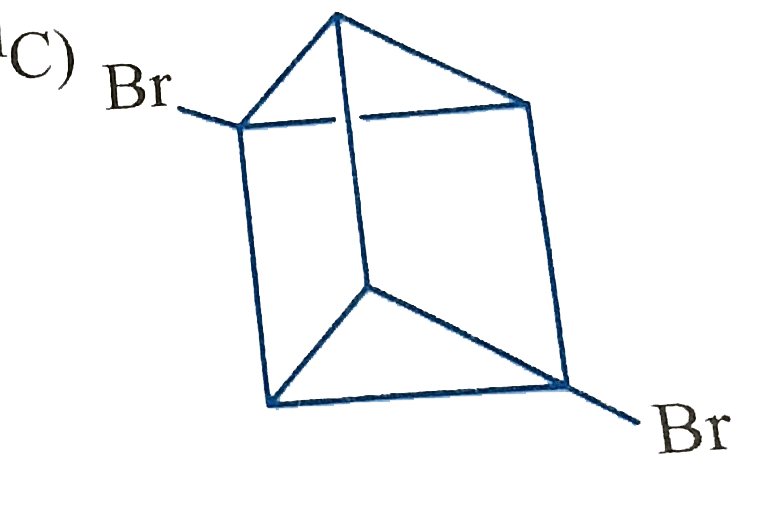
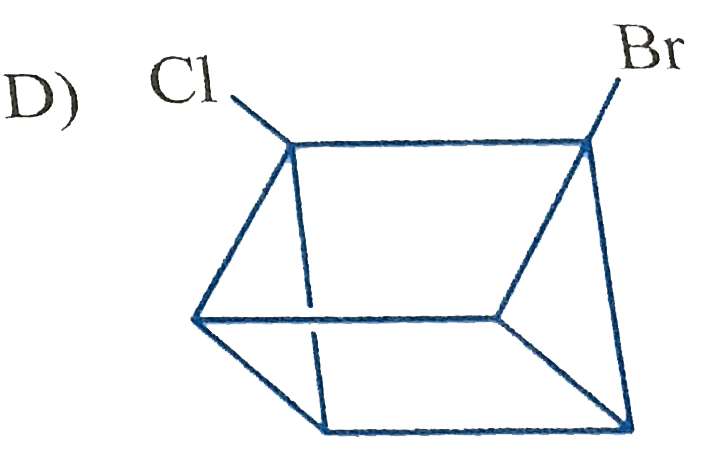
Which of the following cycloalkanes will show cis-trans isomerisin? |
|
Answer»
|
|
| 31. |
Which of the fillowing pairs cannot exist in between two structural isomers |
|
Answer» FUNCTIONAL and position |
|
| 32. |
Which of the following is attacking spcies in conversion Y to P? |
|
Answer» `:CCl_(2)` |
|
| 33. |
The units , nanometer , fermi , angstrom and attometre , arranged in decreasing order are expressed as |
|
Answer» Angstrom , NANOMETRE , FERMI, attometre |
|
| 34. |
What effect on density when pressure pressure at 0.09 mole gas increases at 300 K ? |
| Answer» Solution :At CONSTANT TEMPERATURE, pressure is DIRECTLY proportional to the densith of a fixed mass of the gas. `(d=k^(1)p)`. So DENSITY increase DUE to increasing pressure. | |
| 35. |
What is photon? |
| Answer» SOLUTION :Electromagnetic RADIATIONS are emitted or absorbed or propagated in the FORM of SMALL pockets of energy CALLED photon. | |
| 36. |
What is the coordination number (C.N.) of an atom in (i) primitive cubic unit cell (ii) body-centred cubic unit cell (iii)face-centred cubic unit cell . |
|
Answer» Solution :(i)In a primitive cubic unit cell, each atom has 6 equally spaced nearest NEIGHBOURS . (II)In BCC, an atom at the body centre is surrounded by 8 nearest neighbours situated at the corners of the CUBE. Hence, C. N. =8. (iii) In FCC, an atom is surrounded by 12 nearest neighbours (6 in its own plane, 3 above and 3 below). Hence , C.N. =12 |
|
| 37. |
To which orbit the electron in H atom will jump on absorbing 12.1eV energy ? |
|
Answer» |
|
| 38. |
What is entropy of fusion? |
|
Answer» SOLUTION :When one mole of the solid melts at its melting point reversibly the HEAT absorbed is called molar heat of vapourisation. The ENTROPY CHANGE is given by `DeltaS_v = (DeltaH_v)/T_b` where `DeltaH_v` is molar heat of FUSION . `T_b` is melting point. |
|
| 39. |
The Valence Shell Electron pair Repulsion (VSEPR) theory helps in predicting the shapes of covalent molecules. Arrange the bond pair electron and ione pair electron in the decreasing order of the replusive interactions among them. |
| Answer» Solution :1 ONE PAIR-lone pair>lonepair-bond pair>bond pair | |
| 40. |
The Valence Shell Electron pair Repulsion (VSEPR) theory helps in predicting the shapes of covalent molecules. A molecule of the type, AB_3E_2 has three bond pairs and two lone pairs of electrons. Predict the most stable arrangement of electron pairs in the molecule. |
Answer» SOLUTION : the electron PAIRS are oriented towards the CORNERS of a triangular by PYRAMIDAL. The molecule has T-shape |
|
| 41. |
How is bond order related to bond length? |
| Answer» SOLUTION :Bond order is INVERSELY proportional to bond length. i.e, HIGHER the bond order LOWER is the bond length. | |
| 42. |
The strongest acid among the following aromatic compound is |
|
Answer» Ortho-nitrophenol |
|
| 43. |
What are metallic/interstitial hydrides ? How do they differ from molecular hydrides ? |
|
Answer» Solution :Metallic/Interstitial hydride make many d-block and f-block metals. These hydrides are good conductor of electricity. These hydrides are non-stoichiometric EXCEPT SALINE hydride and e.g. being deficient in hydrogen. e.g., `LaH_(2.87) YbH_(2.55) , TiH_(1.5-1.8), VH_(0.56), NiH_(0.6-0.7), PbH_(0.6-0.8)`etc. In these hydrides law of constant composition does not hold good. 
|
|
| 44. |
The standard Gibbs energy change at 300 k for the reaction 2A hArr B + C " is " 2494. 2J . At a given temperature, and time. the composition of the reaction mixture is [A] = 1//2,[B]= 2,[C]= 1//2. The reaction proceed in the (R = 8.314 "J/K mol," = 2.718) |
|
Answer» |
|
| 45. |
Total number of sigma-bond present in the molecula of propyne is |
|
Answer» |
|
| 46. |
The value of Delta H_(transition) of C(diamond) is 1.9 kJ/mol at 25^(@)C. Entropygraphite id higher than entropy of diamond . The implies that. |
|
Answer» C(diamond) isthermodynamicallymoresatblethan (GRAPHITE) at `25^(@)`C. |
|
| 47. |
What amount of silver chloride will be obtained when 20 mL N//20 HCl is made to react with excess of AgNO_(3) ? |
|
Answer» |
|
| 48. |
What is K_H"in " P_( "Solute ") =K_H X_("solute ") ?on what does the value ofK_Hdepends? |
| Answer» Solution :`K_H ` is empireical CONSTANT with the DIMENSION of pressure.The value of ` 'K_H'` depens on the nature of the GASEOUS SOLUTE and solvent | |
| 49. |
Which of the following statements is right regarding geometrical isomers |
|
Answer» All cis-isomers MUST be Z-isomers |
|