Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which one of the following reactions represents the oxidising property of H_(2)0_(2)? |
|
Answer» `2KMnO_(4)+3H_(2)SO_(4)+5H_(2)O_(2)toK_(2)SO_(4)+2MnSO_(4)+8H_(2)O+5O_(2)` |
|
| 2. |
What is the pH at the equivalence point in a titration of 0.2 M NH_(3) (aq) with 0.02 M HBr (aq).For the ammonia K_(b) = 1.8 xx 10^(-5) |
|
Answer» `5.48` |
|
| 3. |
X + "water" rarr ethyne, so what is compound X? |
|
Answer» SODIUM carbide |
|
| 4. |
What is the combining weight of oxygen in N_(2)O_(3)and N_(2)O_(5)respectively ? |
| Answer» Answer :C::D | |
| 5. |
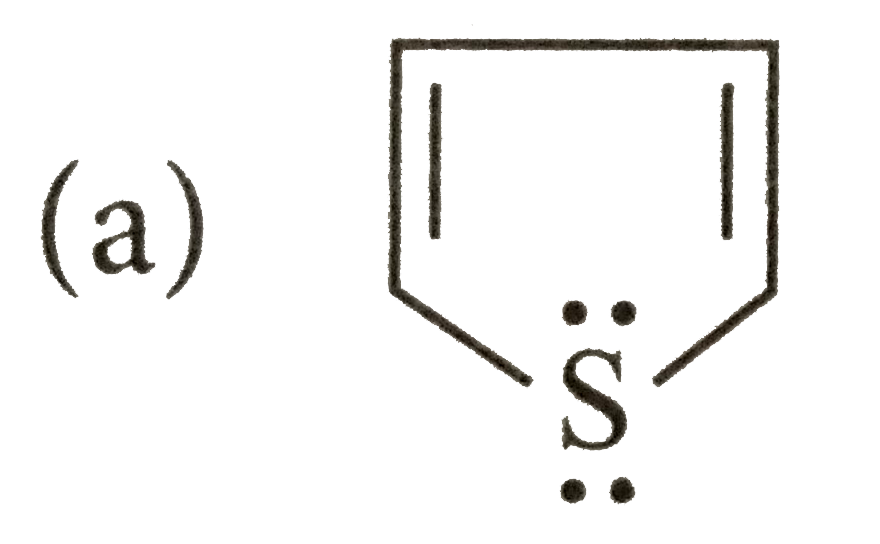
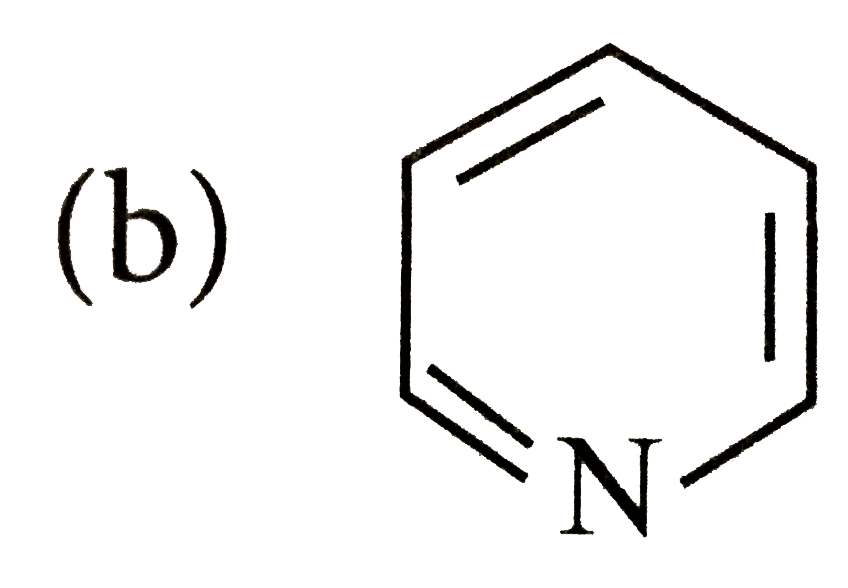
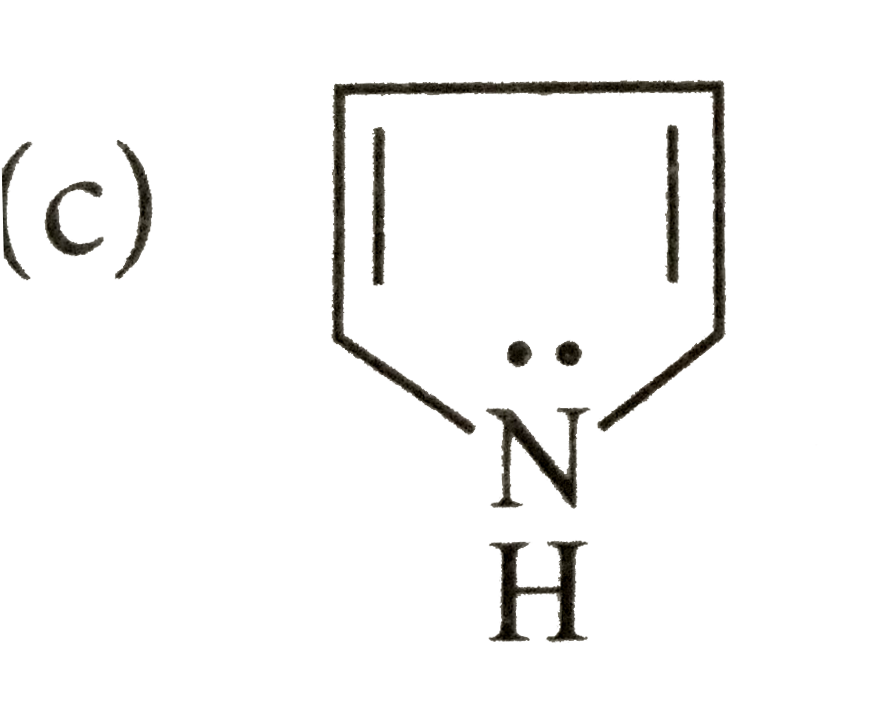
Which of the following species does not show aromaticity? |
|
Answer»
 4n+2=4 or 4n=4-2=2, n=0.5 |
|
| 6. |
The species which can best serve as an initiater for the cationic polymerisation is |
|
Answer» `LiAlH_(4)` |
|
| 7. |
Which of the following structures permits cis-trans isomerism? |
|
Answer» `X_(2)C = CY_(2)` |
|
| 8. |
Which are the correct IUPAC names of the following compounds ? overset(H_3C-CH_2-CH_2-CH_2-underset(CH_3)underset|underset(H_3C-C-CH_3)underset|underset(CH_2)underset|"CH"-CH_2-CH_2-CH_2-CH_2-CH_3) |
|
Answer» 5-(2'-2'-Dimethylpropyl)decane |
|
| 9. |
Which of the following oxides is not expectedto reactwith sodium hydroxide ? |
|
Answer» CaO |
|
| 10. |
What type of stoichiometric defect is shown by : (i) ZnS (ii)AgBr? |
|
Answer» Solution :(i)ZnS SHOWS Frenkel DEFECT because its ions have a large DIFFERENCE in size (II) AGBR shows both Frenkel and Schottky defects |
|
| 11. |
Write the main product of given reaction : (i) C_(6)H_(5)CH=CH_(2) overset(HBr)rarr (ii) C_(4)H_(10)overset("Air "Delta)rarr (iii) CH_(3)-underset(CH_(3))underset(|)(C)=CH_(2) overset(Br_(2))rarr (v) (CH_(3))_(2)CHCHBrCH_(3) underset(KOH, Delta)overset("Ethanol")rarr (vi) CH_(3)CH=CHCH_(3)+HBr rarr (vii) CH_(3)-overset(CH_(3))overset(|)(C)=CH_(2)+HBr rarr (viii) CH_(3)-CH=overset(CH_(3))overset(|)(C)-CH_(3)-HCl rarr (ix) 3-Methyl but-1-ene+HBr (x) CH_(3)CH=CHCH_(3) underset(KMnO_(4))overset("Cold ", Na_(2)CO_(3))rarr (xi) 2, 3-Dimethyl but-2-ene overset("Bayer test")rarr (xii) Cyclonexanol + alcoholic KOH+ Delta |
|
Answer» Solution :(i) `underset("Phenyl ethene")underset("(2-styrene")(C_(6)H_(5)CH=CH_(2))overset(HBr)rarr underset("1-Bromo-phenylehane")(C_(6)H_(5)-underset(Br)underset(|)(CH)-CH_(3))` (ii) `underset("Butane")(C_(4)H_(10))+(13)/(2)O_(2) underset(Delta)overset("Air")rarr 4CO_(2(g)) + 5H_(2)O_((g))` (III) `underset("2-Methylpropene")(CH_(3)-underset(CH_(3))-underset(|)(C)=CH_(2))underset(C Cl_(4))overset(Br_(2))rarrunderset("2-Methyl propane")underset("1,2-Dibromo")(CH_(3)-underset(CH_(3))underset(|)overset(Br)overset(|)(C)-CH_(2)Br)` (iv)  (vi) `underset("2-Butene")(CH_(3)CH=CHCH_(3))+HBr rarr underset("2-Bromobutane")(CH_(3)CH_(2)-underset(Br)underset(|)(CH)-CH_(3))` (vii)`underset("2-Methyl propene")(CH_(3)-overset(CH_(3))overset(|)(C)=CH_(2)+HBr)rarr underset("2-Bromo-2-methyl-propane")(CH_(3)-underset(Br)underset(|)overset(CH_(3))overset(|)(C)-CH_(3))` (viii) `underset("2-Butene")(CH_(3)CH=CHCH_(3))+HBr rarr underset("2-Bromobutane")(CH_(3)CH_(2)-underset(Br)underset(|)(CH)-CH_(3))` `underset("2-Methyl propene")(CH_(3)-overset(CH_(3))overset(|)(C)=CH_(2)+HBr)rarr underset("2-Bromo-2-methyl-propane")(CH_(3)-underset(Br)underset(|)overset(CH_(3))overset(|)(C)-CH_(3))` (ix)3-Methyl but-1-ene 
|
|
| 12. |
Which orbital in each of the following pairs is lower in energy in a many-electron atom? (a) 2s, 2p (b) 3p, 3d (c) 3s, 4s (d) 4d, 5f |
|
Answer» <P> SOLUTION :(a) `2s LT 2p` (b) `p lt 3d` (C) `3s lt 4s` (d) `4d lt 5f` |
|
| 13. |
Which of the following units represents the largest amount of energy? |
|
Answer» Calorie |
|
| 14. |
Which of the following when treated with a Grignard reagent yield (s) an alkane? |
|
Answer» `H_2O` |
|
| 15. |
Thermochemical equations. |
|
Answer» Solution :A balanced chemical equation together with the value of its `Delta_(R) H` is called a thermochemical equation. It would be necessary to remember the FOLLOWING conventions regarding thermochemical equations. (1) The coefficients in a balanced thermochemical equation refer to the number of moles reaction. (2) The numerical value of `Delta_(r) H^( Theta)` refers to the number of moles of SUBSTANCES specified by an equation. Standard ENTHALPY change `Delta_(r ) H^( Theta)` will have units as `" kj mol"^(-1)`. e.g., `Fe_(2) O_(3(s)) + 3H_(2(g)) to 2Fe_(2(g)) to 2Fe_((s)) + 3H_(2) O _((l))` From standard enthalpy of formation, `Delta_(f) H^( Theta) (H_(2) O) = -285.83` kj/mol `Delta_(f) H^( Theta) (Fe_(2) O_(3) )=-824.2` kj/mol `Delta_(f) H^( Theta) (FE) and Delta_(f) H^( Theta) (H_(2) ) =0` Then, `Delta _(f) H^( Theta) = [3(-285.83)] - [1(-824.2)]` `= -33.3` kj/mol If we had balanced the equation differently, for example, `(1)/(2) Fe_(2) O_(3 (s)) + (3)/(2) H_(2(g)) to Fe_((s)) + (3)/(2) H_(2) O_((l))` `Delta_(r) H_(2) ^( Theta) = [(3)/(2) (-285.83)]-[(1)/(2) (-824.2)]` `=(-428.7 + 412.1) "kj mol"^(-1)` `=-16.2 "kj/mol"=(1)/(2) Delta_(r) Delta H_(1)^( Theta)` It shows that enthalpy is an extensive quantity. (3) When a chemical equation is reversed, the value of `Delta_(r) H^( Theta)` is reversed in sign. `N_(2(g) ) + 3H_(2(g)) to 2NH_(3(g)) , Delta_(r) H^( Theta) =-91.8 "kj/mol"` `2NH_(3(g)) to N_(2 (g)) + 3H_(2 (g)) , Delta_(r) H^( Theta) = +91.8 "kj/mol"` |
|
| 17. |
Which las holds good for lighter elements up to calcium? |
|
Answer» NEWLAND's Octaves |
|
| 18. |
What is the ratio of most probable velocities of hydrogen molecules at 50 K and oxygen molecules at 800 K ? |
| Answer» SOLUTION :`1 : 1` | |
| 19. |
The velocity associated with a proton moving in a potential difference of 1000 V is 4.37 xx 10^(5) m s^(-1). If the hockey ball of mass 0.1 kg is moving with this velocity, calculate the wavelength associated with this velocity? |
| Answer» Solution :`lamda = (h)/(mv) = (6.626 xx 10^(-34) kg m^(2) s^(-1))/((0.1lh) (4.37 xx 10^(5) MS^(-1))) = 1.516 xx 10^(-28) m` | |
| 21. |
Which of the following groip has less electron attracting capasity then hydrogen? |
|
Answer» `-CH_(3)` (`""^(-)I` effect) `= -NO_(2) gt -CN gt -COOH gt -F gt - Cl gt- Br gt -I gt -OH gt -OCH_(3) gt -C_(6)H_(5) gt -H` |
|
| 22. |
Which of the following statement(s) is/are corect |
|
Answer» Water equalies the strength of `NAOH, KoH,CsOH` and `Ba(OH)_(2)` `pK_(a) +- 1, :. pK_(a) = 5` If indicator is `50%` ionized, then `["Hin"] = ["In"^(-)]` `:. pH = pK_("In"), pK_(a) = 10^(-5)` |
|
| 23. |
Which of the following metals cannot be extracted by smelting process ? |
|
Answer» Pb |
|
| 24. |
Zinc reacts with hot and concentrated NaOH and forms |
|
Answer» `H_(2)` |
|
| 25. |
What is biosphere? |
| Answer» SOLUTION :Biosphere includes the lithosphere, HYDROSPHERE and atmosphere integrating the living ORGANISMS PRESENT in the lithosphere, hydrosphere and atmosphere. | |
| 26. |
Which has the maximum and minimum mass ? (A) 25.6 g of oxygen (atomic mass = 16) (B) 2.56 gram atom of sodium (atomic mass = 23) (C) 0.256 gram molecule of water (molecular mass = 18) (D) 0.256 gram atom of iodine (atomis mass = 127) |
|
Answer» (B) 1.0 GRAM atom of Na = 23.0 g 2.56 gram atom of `Na = 2.56 xx 23.0 = 58.88` g (C) 1.0 gram molecule of `H_(2)O = 18.0 g` 0.256 gram molecule of `H_(2)O = 0.256 xx 18.0 = 4.608 g` (D) 1.0 gramatom of iodine `= 127.0 g` 0.256 gram atom of iodine `= 127.0 xx 0.256 = 32.512 g` Maximum MASS of = BMinimum mass = C. |
|
| 27. |
Which of the following is an example of non-benzenoid aromatic compound ? |
| Answer» Solution :azulene | |
| 28. |
What are real gases ? |
| Answer» Solution :REAL GAS are those which do not FOLLOW at the gas laws at all temperature and pressure. | |
| 29. |
What will be the pH of 1M NaNO_(3)solution at 25^(@)C? |
| Answer» Solution :`NaNO_(3)` is a salt of STRONG acid and strong base. HENCE, its solution will be NEUTRAL. At `25^(@)C`, its pH will be = 7 . | |
| 30. |
Which of the following are cumulative dienes? |
|
Answer» `CH_3 - CH = C = CH-CH_2` 
|
|
| 31. |
Which of the following hydrides is electron precise hydride ? |
|
Answer» `B_2H_6` |
|
| 32. |
Write the general outer electronic configuration of d-block elements. |
| Answer» SOLUTION :Non-availabillity of d ORBITALS | |
| 33. |
The salt responsible for permanent hardness of H_(2)O is |
|
Answer» `Na_(2)SO_(4)` |
|
| 34. |
Which is pure basic oxide |
|
Answer» `Al_2O_3` |
|
| 35. |
Which has maximum density between Dry or air containing moisture ? Why ? |
| Answer» SOLUTION :Dry air, because dry air contains `N_(2)(28), O_(2)(32), CO_(2)(44)` having maximum molecular MASS but air of moisture contain `O_(2), CO_(2), N_(2)` and `H_(2)O(18)` having LES molecular mass. | |
| 37. |
What volume of 10 M HCl and 3M HCl should be mixed to get 1 L of 6M HCl solution ? |
|
Answer» Let the VOLUME of 10 MHCl = x L `:.` the volume of `3MHCl = (1-x)L` `M_(1)V_(1)+M_(2)V_(2)=M_(3)V_(3),10xx x+3xx(1-x)=6xx1` `10x +3-3x=6or7x=3orx=3//7=0.428L` `:.` Volume of 10 MHCl = 0.428 L = 428 mL Volume of 3 M HCl = (1000 - 428)= 573 mL |
|
| 38. |
The sulphate of metal M contains 20% of M. this sulphate is isomorphic with ZnSO_(4)*7H_(2)O. Which of the following are true about metal M- |
|
Answer» atomic MASS of METAL is 24 |
|
| 39. |
Which of the following is a correct statement:- |
|
Answer» Surface tension of a liquid decreases with increase in temperature |
|
| 40. |
The systematic name of (CH_(3))_(2) CHCOOH is: |
|
Answer» 2-Propanoic ACID 2-Methylpropanoic acid |
|
| 41. |
Which of the following can effectively remove all types of hardness of water? |
|
Answer» SOAP |
|
| 42. |
What is the difference between distillation, distillation under reduced pressure and steam distillation ? |
|
Answer» Solution :Distillation is EMPLOYED in CASE of volatile liquids ASSOCIATED with non-volatile IMPURITIES. Distillation under reduced pressure is carried to purify liquids which decompose at their boiling point temperatures. Steam distillation is done for the steam volatile associated with water immiscible impurities. For more DETAILS, consult section 12.38. |
|
| 43. |
Water is a useful solvent. Which properties make water as a good solvent? |
| Answer» Solution :High DIPOLE MOMENT and high dielectric CONSTANT make water as a USEFUL solvent | |
| 44. |
What is oxidation reaction ? Explain it with example. |
|
Answer» SOLUTION :.In which reaction oxygen/ELECTRONEGATIVE elements is added or hydrogen/electropositive element is remove this TYPE of reaction is known as oxidation reaction... `2Mg_((s))+O_(2(G))to2MgO_((s))` `S_((s))+O_(2(g))toSO_(2(g))` The elements Mg and S are oxidised on account of addition of oxygen to them. Mg is oxidised by electronegative elements such as `F_(2),Cl_(2)` `Mg_((s))+F_(2(g))toMgF_(2(s))` `Mg_((s))+Cl_(2(g))toMgCl_(2(s))` `2K_(4)[Fe(CN)_(6)]_((aq))+H_(2)O_(2(aq))tounderset("Potassium ferrocyanide")(2K_(3)[Fe(CN)_(6)]_((aq))+2KOH_((aq)))` Removal of K form potassium ferrocyanide is known as oxidation. |
|
| 45. |
What is the structure of H_(2)O_(2)? Draw a shematic diagram indicating the shape o the molecule clearly ? |
Answer» Solution :Hydrogen peroxide has open-book structure. It has non-planner structure. In the crystal, the DIHEDRAL angle `111.5^(@)"REDUCE to"90.2^(@)` on accountof hydrogen BONDING. There is single bond between two OXYGEN ATOM in hydrogen peroxide. 
|
|
| 46. |
The state of a gas can be described by quoting the relationship between |
|
Answer» pressure, VOLUME, TEMPERATURE The ideal gas equation is `pV= n RT`. Thus, `p, V, T and n` describe the state of the system. |
|
| 47. |
Which is the conjugate acid of H_2SO_4 ? |
|
Answer» `HSO_4^-` |
|
| 48. |
Write the rules for assigning oxidation number. |
|
Answer» Solution :(1) All atoms in the elemental or MOLECULES state have zero oxidation state. (2) Elements ofgroup I and II in the periodic table always have +1 and +2 oxidation states respectively (3) HYDROGEN has +1 oxidation state in all its compounds except metal hydrides, where it is -1. (4) Oxygen has been ASSIGNED as oxidation number of -2 in all its compounds except perocides and oxygen fluoride. In peroxide it is -1 and in oxygen fluoride it is +2. (5) Halogens generally have -1 oxidation state Except fluorine. other halogens may have positive oxidation states in their oxides and inter halogen compounds (Fluorine always has -1 state). (6) The algebraic SUM of the oxidation number of all the elements in a compound is zero and that in an ion is EQUAL to the net charge onthe ion. |
|
| 49. |
When 1 molof CO_(2)(g) occupying a volumeof 10 L at 27^(@) isallowedto expand under adiabaticconditions, temperaturefallsto -123^(@)C. Hence,final volume of the gas will be : |
|
Answer» 20L For`CO_(2)` ( triatomic gas) , `gamma=1.33` `:.( 150)/( 300) = ((10)/( V_(2)))^(0.33) = ((10)/( V_(2)))^(1//3)` or `(1)/(2)=((10)/( V_(2)))^(1//2)` or`(10)/( V_(2)) = (1)/(8) `or`V_(2) = 80 L` |
|