Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
When an ideal gas undergoes undestrained expansion, no cooling occurs because the molecules |
|
Answer» Are above the inversion temperature |
|
| 2. |
What is syngas ? Why is it called so? |
| Answer» Solution :MIXTURES of CO and `H_(2)` is CALLED syngas. It is so named because it is used for the synthesis of methanol and a number of hydrocarbons. | |
| 3. |
Which of the following ions perform important biological functions in maintenance of the ion balance and nerve impulse conduction? |
|
Answer» `Li^(+) RB^(+)` |
|
| 4. |
Volume strength of 1.5 N H_2O_2 is |
|
Answer» 1.5 = `1.5 xx5.6`= 8.4 `underset((2xx34g))(2H_(2)O_(2))to2H_(2)O+underset((22.4liters))(O_(2)dowmarrow)` Volume strength of Hydrogen peroxide `= ("Normality "xx "Equivalent WEIGHT of "H_(2)O_(2)xx2.24)/(68)` |
|
| 5. |
Which elements of group shows anamolous behaviour. Explain with examples. |
|
Answer» Solution :First element of each group shows anamolous behaviour. Ex. Li elements of group-1, Be element of group-2. BORON to fluorine elements of group 13 to 17. Li and Be from different covalent compound than other elements of group. DIAGONAL Relationship : In fact the behaviour of lithium and beryllium is more similar with the second element of the following group i.e., magnesium and aluminium, respectively. This SORT of similarity is commonly REFERRED to as diagonal relationship in the periodic properties. |
|
| 6. |
The stabilisation of a half filled d-orbital is more prounced than that of the p-orbital why? |
|
Answer» Solution :(i) Symmetry : The half filled orbitals are more symmetrical than partially filled ORBITAL and this symmetry leads to GREATER stability. (II) Exchange energy : The ELECTRONIC with same spin in the different orbitals of the same subshell can exchange their position. Each such exchange energyand this is known as exchange energy. Greater the number of exchanges, greater the exchange energy and greater the stabitity. In d - orbital 10 exchanges are possible but in p - orbital 6 exchange are possible. |
|
| 7. |
Which of the following reaction defines Delta H _(f)^(@) ? |
|
Answer» `C_("diamond") + O_(2)(g) RARR CO_(2)(g)` |
|
| 8. |
Which halides of element of group 13 exist as dimer in vapour state ? |
|
Answer» AL |
|
| 9. |
Which one of the following test is used to detect phosphorous in an organic compound? |
|
Answer» a)Silver NITRATE test |
|
| 10. |
What transition in the hydrogen spectrum would have the same wavelength as the Balmer transition, n = 4 to n = 2 of He^(+) spectrum ? |
|
Answer» Solution :For H - like particles in general `bar(v) = (2pi^92) m Z^(2) e^(4))/(CH^(3)) ((1)/(n_(1)^(2)) - (1)/(n_(2)^(2))) = RZ^(2) ((1)/(n_(1)^(2)) - (1)/(n_(2)^(2)))` `:.` For `He^(+)` spectrum, for Balmer transition, n = 4 to n = 2 `bar(v) = (1)/(lamda) = RZ^(2) ((1)/(2^(2)) - (1)/(4^(2))) = R xx 4 xx (3)/(16) = (3R)/(4)` For hydrogen spectrum `bar(v) = (1)/(lamda) = R ((1)/(n_(1)^(2)) - (1)/(n_(2)^(2))) = (3)/(4)R or (1)/(n_(1)^(2)) - (1)/(n_(2)^(2)) = (3)/(4)` which can be so for `n_(1) = 1 and n_(2) = 2`, i.e., the transition is form n = 2 to n = 1 |
|
| 11. |
What are the uses of hydrogen peroxide? |
|
Answer» Solution :(i) `H_(2)O_(2)` is used in WATER TREATMENT to OXIDIZE pollutants. (ii) `H_(2)O_(2)` is used as a mild antiseptic. (iii) `H_(2)O_(2)` is used as a bleach in textile, paper and hair-care industry. |
|
| 12. |
The standard readuction potential at 298 k for the followng half cell reaction are Zn^(2)+2e^(-)rarrZn(s),E^(@)=-0.7623 V Cr^(3+)(aq)+3e^(-)rarrCr(s),e^(@)=-0.740 V Cr^(3+)(aq)+3e^(-)rarrCr(s),E^(@)=-0.70 v 2H^(+)(aq)+2e^(-)rarrH_(2)(g),E^(@)=0.0V F_(2)+2e^(-)rarr 2f^(-)(aq)=2.87V which of the following is the strongest reducing agent |
|
Answer» CR (s) |
|
| 13. |
Which of the following alkenes are more stable than |
|
Answer»
|
|
| 14. |
Which has maximum viscosity between ethanol and H_(2)O ? |
| Answer» SOLUTION :`H_(2)O`. Because INTERMOLECULAR H - bond is STRONG in `H_(2)O`. | |
| 15. |
Which one is primary alcohol ? |
|
Answer» Butene-2-ol |
|
| 16. |
What is bleaching powder? How is it prepared? |
| Answer» Solution :Bleaching powder is `CA(OCI)_(2)` It is prepared by treating chlorine with MILK of LIME. `2Ca(OH)_(2) + 2CI_(2) to CaCl_(2) + underset("Bleaching powder")(Ca(OCI)_(2) + 2H_(2)O` | |
| 17. |
Which of the following will give hydrogen peroxide on hydrolysis |
|
Answer» `H_(2)S_(2)O_(8)` `H_(2)SO_(5)+H_(2)O to H_(2)SO_(4)+H_(2)O_(2)` `H_(3)PO_(5) + 2H_(2)O toH_(2)PO_(4) + H_(2) O_(2)` `HCLO_(4)`on hydroliysisdoes notproduced`H_(2)O_(2)` |
|
| 18. |
Which of the following hydrides are colvalent polymeric? |
|
Answer» Ca |
|
| 19. |
Which of the following statements in incorrect:- |
|
Answer» ENZYMES are in COLLOIDAL state |
|
| 20. |
Why AI(HO)_(3) is insoluble in excess of NH_(4)OH but soluble in NaOH? |
|
Answer» Solution :NaOH is strong alkali than `NH_(4)OH and AI(OH)_(3)` being amphoteric DISSOLVES in NaOH due to the FORMATION os soluble `NaAIO_(2)`. `I(OH)_(3)+NaOHrarrNaAIO_(2) + 2H_(2)O` |
|
| 21. |
Which one of the following statements is not correct ? |
|
Answer» Alcohols are weaker acids than water. |
|
| 22. |
Which of the following groups has highest inductive effect? |
|
Answer» `CH_(3)-` |
|
| 23. |
Which will form 2,2-Dibromopropane with HBr ? |
|
Answer» `CH_2=CH-CH_3`<BR>`CH_3-C-=CH` |
|
| 24. |
Which property is the same for 1.0g samples of H_(2) and CH_(4) in separate 1.0L containers at 25^(@)C? |
|
Answer» PRESSURE in I is the MAXIMUM. |
|
| 25. |
Write Lewis symbols for the following atoms and ions: S and S^(2-) , Al and Al^(3+) , H and H^(-) |
|
Answer» Solution :`""_(16)"S"+2,8,6" "THEREFORE` Lewis symbol `=:overset(* *)S:,S^(2-) "ion" =[:UNDERSET(* *)overset(* *)S:]^(2-)` `""_(13)AL = 2,8,3" "therefore` Lewis symbol `=*overset(*)Al.,Al^(3+)" ion"=[Al]^(3+)` `""_(1)H =1" "therefore` Lewis symbol `=H., H^(-)" ion" = overset(*)H*` |
|
| 26. |
What are decarboxylation reactions given an example |
| Answer» Solution :WHENA MIXTUREOF SODIUMSALT ofcarboxylicacidand sodaline isheatedan alkane isformedduringthisprocess`CO_(2)` molccule is climinated processprocess and this processprocessknow asdecarboxylation | |
| 27. |
What makes a glass different from a solid such as quartz ? Under what conditions could quartz be converted into glass ? |
| Answer» SOLUTION :Glass is an amorphous solid in which the CONSTITUENT particles (`SiO_4` TETRAHEDRA) have only a SHORT range order and there is no long range order. In quartz. the constituent particles (`SiO_4` tetrahedron) have short range as well as long range order. On melting quartz and then cooling it RAPIDLY, it is converted into glass | |
| 28. |
which among the following pairs of ions cannot be separated by H_(2)S in the presence of dilute HCl ? |
|
Answer» `Bi^(3+) , Sn^(4+)` |
|
| 29. |
Which of the followingare isoelectronicspeciesl.e.Thosehavingthe samenumberof electron ? Na^(+)K^(+)Mg^(2+) ,Ca^(2+) ,S^(2-) Ar |
|
Answer» SOLUTION :`NA^(+)Mg^(2+)` bothhave `10^(e )`so theyare ISOELECTRONIC `K^(+)Ca^(2+)S^(2-)Ar18 e^(-)` areisoelectronic . |
|
| 30. |
What is formula of V_(C ), P_(C ) T_(C ) ? |
| Answer» Solution :`V_(C )=3B, P_(C )=(a)/(27b^(2)), T_(C )=(8A)/(27Rb)` | |
| 31. |
Whatis theCORRECTbond-linestructuralformulaforCH_(3)CH_(2)CH_(2)CH_2 CH_(3)? |
| Answer» | |
| 32. |
What is positive and negative deviation of real gas at constant pressure for pV to p ? |
| Answer» SOLUTION :If curve of GRAPH is down from ideal GAS then DEVIATION will be negative. If curve of graph is up from ideal gas then deviation will be positive. | |
| 33. |
The reagent commonly used to determine harness of water titrimetrically is : |
|
Answer» OXALIC acid |
|
| 34. |
Which of the following is /are correct about redox reactio? MnO_(4)^(ϴ)+H^(ϴ)toMn^(+2)+S_(4)O_(6)^(-2) |
|
Answer» 1mol of `S_(2)O_(3)^(2-)`is oxidised by 8 mol of `MnO_4^(-)` `2S_(2)O_(3)^(2-)rarrS_(4)O_(6)^(2-)(n=2/2=1)` Eq of `MnO_(4)^(-)-=` Eq of `S_2O_(3)^(-)` `5xx` MOLES of `MnO_(4)^(-)-=1xx` moles of `S_2O_(3)^(2-)` `:.1` mol of `MnO_(4)^(-)-=1xx` moles of `S_(2)O_3^(2-)` `:.1 ` mol of `S_(2)O_(3)^(2-)=5` mol of `MnO_4^(-)` b) pH change from 4 to 10 (acidic to strongly BASIC ) `e^(-)+MnO_(4)^(-)rarrMnO_(4)^(2-)(n=1)` `S_(2)O_3^(-2)rarr2SO_(4)^(-2)+8e^(-)(n=8)` Eq of `MnO_4^(-) =" Eq of " S_(2)O_3^(2-)` `:.1` mol of `S_2O_(3)^(2-)=1/8 ` mol of `MnO_4^(-)` HENCE with change of pH from 4 to 10, will change the stiochiometry of reaction and also changes the product. c) pH change from 4 to 7 (acidic to neutral medium ) `3e^(+)+MnO_(4)^(-)RARR MnO_(2)(n=3)` `S_(2)O_3^(2-) rarr 2HSO_(4)^(-)+8e^(-) (n=8)` Hence it will also effect the stoichiometry of reaction and nature of product. d) At `pH = 7,S_(2)O_(3)^(2-)` is oxidised to `HSO_4^(-)` ion |
|
| 35. |
What happens when white phosphrous is heated with concetrated NaOH solution in an inert atmosphere of CO_(2) ? |
|
Answer» Solution :White phosphours REACTS with NaHO to form PHOSPHINE. `underset("Phosphorus")(P_(4))+3NaOH+3H_(2)Ounderset(CO_(2)"atmosphere")overset("Heat")tounderset("Phoshine")(PH_(3))+underset("Sod. hypopsphite")(3NaH_(2)PO_(2))` |
|
| 36. |
What is sodalime? |
| Answer» SOLUTION :When quick lime or calcium oxide is mixed with SODIUM hydroxide gives SOLID MIXTURE KNOWN as sodalime [CaO + NaOH] | |
| 37. |
The threshold limit value or CO in the air is nearly ____whereas that of CO_2 is nearly _________ |
|
Answer» |
|
| 38. |
Tin plague is |
|
Answer» Conversion of stannous SALT into STANNIC salt |
|
| 39. |
Which of the following is not a part of green chemistry? |
|
Answer» Photichemisty |
|
| 40. |
X underset((ii) dil. H_(2)SO_(4))overset((i) HCN)rarr2-Hydroxypropinoinc acid The structure of X is |
|
Answer» `CH_(3)COOH` 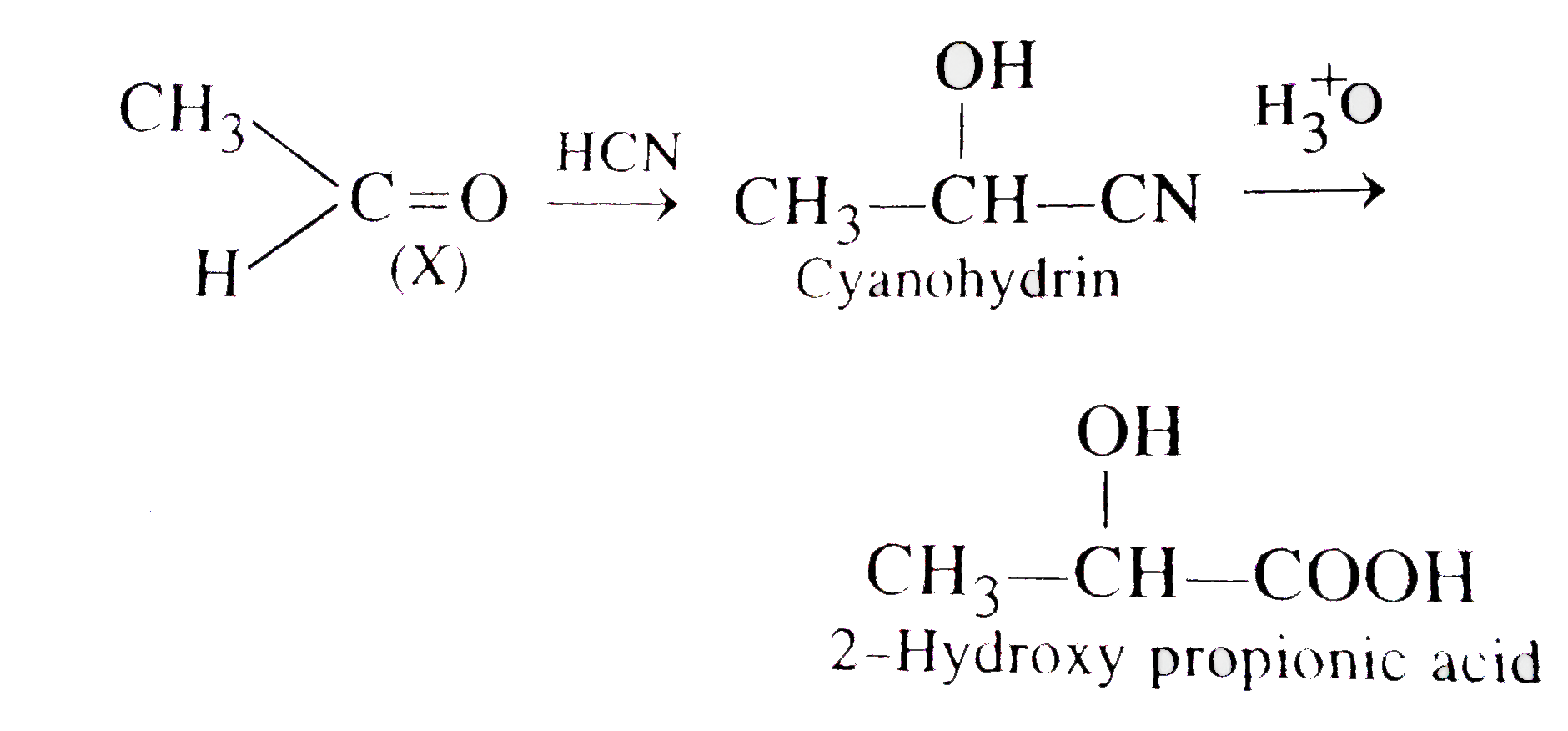
|
|
| 42. |
When an atom is placed in the magnetic field, then due to the presence of electrons in it |
|
Answer» the ORBITALS do not orient themselves relative to the MAGNETIC field |
|
| 43. |
What is the present by mass of titanium in rutile, a mineral that contains Ti and oxygen if structure can be described as a closest packed array to oxide ions with titanium ions in one-half of the octahedral holes ? What is the oxidation number of Ti ? (Atomic mass of Ti=48) |
|
Answer» Solution :If no. of oxide ions =n, then octahedral HOLES =n and hence `O^(2-)` ions `=n/2` `therefore TI:O =n/2 :n=1/2:1=1:2` Hence, FORMULA of the oxide is `TiO_2`. Molar mass of `TiO_2`=48+32 =`80 g mol^(-1)` `therefore` % of Ti=`48/80xx100`=60% . Oxidation state of Ti in `TiO_2`=+4 |
|
| 44. |
Which of the following relation is true? |
|
Answer» `C_p GT C_V` |
|
| 45. |
Write the IUPAC names of the products obtained by the ozonolysis of the following compouds : Pent-2-ene |
| Answer» SOLUTION :`CH_3-CH=CH-CH_2-CH_3rarrCH_3-CHO+CH_3-CH_2-CHO` | |
| 46. |
What is the frequency of photon, whose momentum is 1.1xx10^(-23) kg ms^(-2) |
|
Answer» `5xx10^16` HZ `therefore mc^2 =hv` `v=(mc^2)/(h)=(1.1xx10^(-23)xx3xx10^8)/(6.6xx10^(-34))=5xx10^18` Hz |
|
| 48. |
Whatare vicinal dihalides how willyouperpane alkene from vicinal dihaldes |
|
Answer» SOLUTION :(i) THECOMPOUNDIN whichtwohalogenatomsare attachedto adjecentcarbon-atomarecalledas vicinaldihalids (II) When VICINAL dihalidsare warnedwithgramulated zincin methanoltheylosea MOLECULE |
|
| 49. |
Write note on covalent or molecular hydrides. |
|
Answer» Solution :Dihydrogen forms MOLECULAR compounds with most of the p-block elements. Most familiar examples are `CH_4, NH_3, H_2O` and HF. For convenience hydrogen compounds of non metals have also been considered as hydrides. Being covalent, they are volatile compounds. Molecular hydrides are further classified ACCORDING to the relative numbers of electrons and bonds in their Lewis structure into : (i) electron-deficient, (ii) electron-precise, (iii) electron - rich hydrides. An electron-deficient hydride, as the name suggests, has too few electrons for writing its conventional Lewis structure. Example : Diborane `(B_2H_6)` In fact all elements of group 13 will form electron-deficient compounds. They ACT as Lewis acids i.e., electron acceptors. Electron-precise compounds have the REQUIRED number of electrons to write their conventional Lewis structures. All elements of group 14 form such compounds (e.g., `CH_4`) which are tetrahedral in geometry. Electron-rich hydrides have excess electrons which are present as lone pairs. Example : Elements of group 15-17 form such compounds. (`NH_3` has 1-lone pair, `H_2O` -2 and HF-3 lone pairs). They will behave as Lewis bases i.e., electron donors. The presence of lone pairs on HIGHLY electronegative atoms like N, O and F in hydrides results in hydrogen bond formation between the molecules. This leads to the association of molecules. |
|