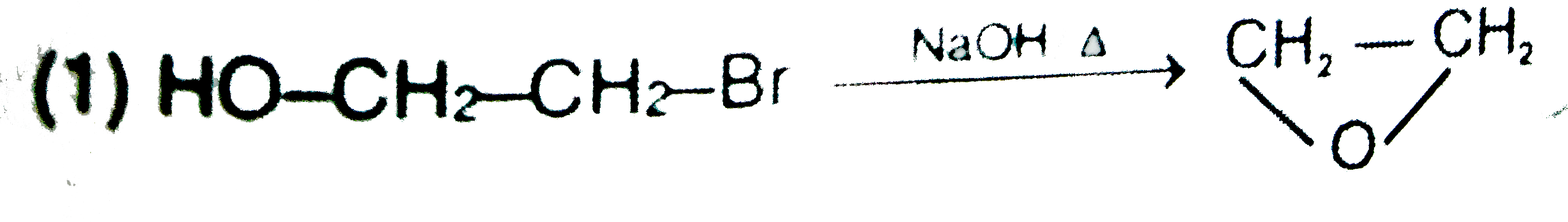Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which element in periodic table has highest ionisation energy. (IE) ? |
|
Answer» |
|
| 2. |
Write the resonating structures for the following molecules. N_(2) O |
Answer» SOLUTION :
|
|
| 3. |
The work function (phi) of some metals is listed below. The number of metals which will show photoelectric effect when light of 300 nm wavelength falls on the metal is {:("Metal",Li,Na,K,Mg,Cu,Ag,Fe,Pt,W),(phi(eV),2.4,2.3,2.2,3.7,4.8,4.3,4.7,6.3,4.75):} |
|
Answer» i.e., `E = ((6.6 xx 10^(-34) Js) (3 xx 10^(8) ms^(-1)))/((300 xx 19^(-9) m))` `= 6.6 xx 10^(-19) J = (6.6 xx 10^(-19))/(1.6 xx 10^(-19)) EV = 4.12 eV` `:.` Metals showing photoelectric effect will be Li, Na, K and Mg only i.e., 4 metals, (which have WORK FUNCTION less than 4.12 eV) |
|
| 4. |
Which of the following is not a correct statement about electrochemical series of reduction potentials ? |
|
Answer» The standard ELECTRODE POTENTIAL of hydrogen is 0.00 volts. |
|
| 5. |
Water pollution is due to |
|
Answer» A agricultural DISCHARGES |
|
| 6. |
Which of the following is not chemical combinations |
|
Answer» `C+O_(2) to CO_(2)` |
|
| 7. |
What are dimensions of Planck's constant. What other physical quantity has the same dimensions ? |
| Answer» Solution :DIMENSIONS of PLANCK's constant, `H = kg m^(2) s^(-1)` Angular MOMENTUM `= mvr = kg (m s^(-1)) m = kg m^(2) s^(-1)` has the same dimensions (Remember from Bohr's POSTULATE, `mvr = n (h)/(2pi)`) | |
| 8. |
When 3*06of solid NH_(4)HS is introduced into a two - litre evacuated flask at 27^(@C,30% of the solid decomposes into gaseous ammonia and hydrogen sulphide. (i) Calculate K_(c) and K _(p) for the reaction at 27^(@)C. (ii) What would happen to the equilibrium when more solid NH_(4)HSis introduced into the flask ? |
|
Answer» Solution :` {:((i),NH_(4)HS(s),HARR,NH_(3)(s),+,H_(2)S(g)), ("Intial amount",3*06 g,,,,), (,=3*06//51 "mole",,,,),(,=0*06 "mole",,,,):}` `{:("At eqm.",0*06-30/100xx0*06,0*018 "mole",0*018"mole"), (,=0*06-0*018,,), (,=0*042 "mole",,):}` ` {:("Eqm.conc",1,0*018//2,0*018//2),(,("being solid"),=0*009,=0*009"mol"L^(-1)):}` ` K_(c) = ([NH_(3)][H_(2)S])/([NH_(4)HS])=(0*009 xx 0*009)/1 = 8*1 xx10^(-5)"" ( [NH_(4)HS(s)] = 1 ) ` ` K_(p)= K_(c) (RT)^(Delta n_(g)) = 2 -0 =2` (II)As`K_(c) = [NH_(3)][H_(2)S] `and does not depend upon the amount of `NH_(4)HS`(s) . hence , therewill be no effect on equilibrium when more solid `NH_(4)HS ` is added. |
|
| 9. |
What is order of intermolecular interaction in solid, liquid and gas ? |
| Answer» Solution :Gas `to` LIQUID `to` Solid : ORDER of INTERMOLECULAR interaction INCREASE. | |
| 10. |
Which of the following anions is present in the chain structure of silicates ? |
|
Answer» `(Si_(2)O_(5)^(2))_(N)` |
|
| 11. |
Which of the following hydrogen bonds is relatively weake? |
|
Answer» N........ H-N |
|
| 12. |
Which of the following pairs of ions are isoelectronic and isostructural ? |
|
Answer» `SO_(3)^(2-) , NO_(3)^(9-)` `CIO_(3)^(-) to sp^(3) to ` Pyramidal` to 17 + 24 + 1 = 42 e^(-)` `CO_(3)^(2-) to sp^(2) to ` TRIANGULAR planar ` to 6 24 + 2= 32 e^(-)` ` NO_(3)^(2-) to sp^(2) to ` Triangular planar ` to 7 + 24 + 1 = 32 e^(-)` Thus, `SO_(3)^(2-) and CIO_(3)^(-)` are isoelectronic and isostructureal . |
|
| 14. |
Which of the following are correct about the reactionA + 2B hArr C + D |
|
Answer» `K_p=(n_C xx n_D)/(n_A xx (N_B)^2) xx ((P)/(RT))^(-1)` `=(n_(C)n_(D))/(n_(A)n_(B)^(2))((V)/(RT))=(n_(C)n_(D))/(n_(A)n_(B)^(2))((SUM n)/(P))` |
|
| 15. |
Which of the following pairs is /are correctly matched ? I. Carbocation : electrophile II. Free radical : paramagnetic III. Carbene : Incomplete octet IV. Carbanion : Incomplete octet Select the correct answer using the codes given below : |
|
Answer» Only I |
|
| 17. |
Which of the following ions is not having the configuration of neon |
|
Answer» `F^-` `F^(-) =1s^2 2s^2 2p^6` `Na^(+)=1s^2 2s^2 2p^6` `Mg^(++)=1s^2 2s^2 2p^6` `Cl^(-)=1s^2 2s^2 2p^6 3s^2 3p^6` |
|
| 18. |
Whichtypeof metalusedin photoelectriccell giveexample . |
| Answer» SOLUTION :Themetalwhichhas lowionizationenthalpyused inphotoelectriccelle.g. CS | |
| 19. |
Which type of force of atraction exist in HF ? |
|
Answer» London force |
|
| 20. |
Which one following mechanism will be followed when Tartiary butyl chloride is treated with alcoholic KOH? |
|
Answer» `S_(N^1)` MECHANISM |
|
| 21. |
Write the general outer electronic configuration of s-, po, d- and f- block elements. |
|
Answer» Solution :GENERAL electronic configuration of outer most ORBITS : s-block elements : `ns^(1-2)` p-block elements : `ns^(2) np^(1-6)` d-block elements : `(n -1) d^(1-10) ns^(0-2)` f-block elements : `ns^(2) np^(6)` |
|
| 22. |
Which of the following is a beta-elimination reaction ? |
|
Answer»
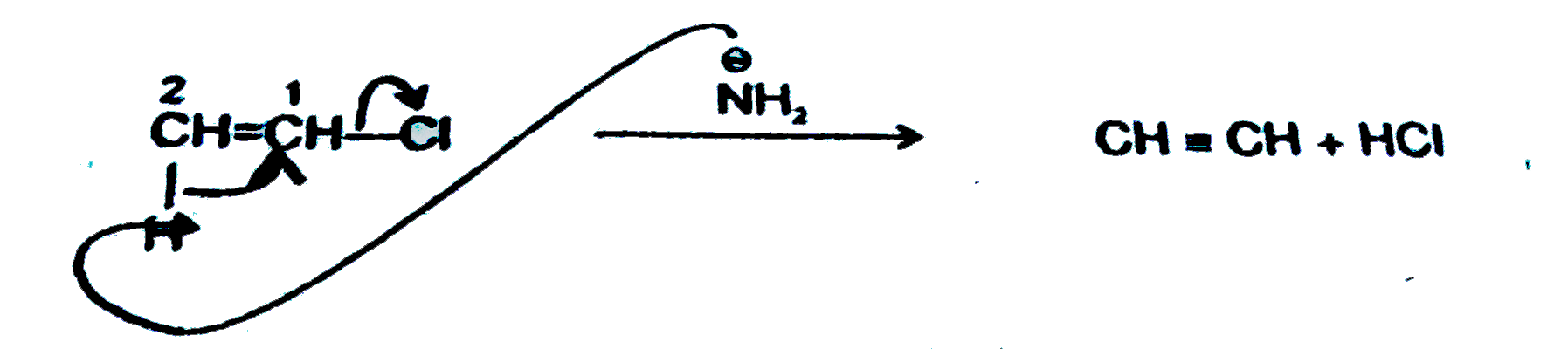 `NaNH_2` is strong BASE so give `beta`-elimination reaction. |
|
| 23. |
Which of the following does not contain a peroxy bond |
|
Answer» `Na_2O_2` |
|
| 24. |
What is the electronic configuration of the elements of group 14 |
|
Answer» `ns^2 np^4` |
|
| 25. |
Write Lewis symbole for the following atoms and ions : S andS^(2 - ): Al and Al^(3+) , and H^(-) |
|
Answer» Solution :`._(16)S = 2, 8, 6 therefore ` Lewis symbol =` : overset (cdotcdot )(S):, S^(2-) ions = [ :UNDERSET(cdotcdot) overset( cdotcdot )S:]^(2-)` ` _(13)Al= 2,8,3 therefore` Lewis symbol`CDOT overset(cdot)Alcdot ,Al^(3+) ions [Al]^(3+)` ` _(1)H = 1therefore ` Lewis symbol= ` H^(cdot ), H^(-) ion = overset(H) (cdot)` . |
|
| 26. |
Write Lewis structure or the following compounds and show formal charge on each atom . HNO_(3), NO_(2), H_(2)SO_(4) |
Answer» Solution :The Lewis structure of the FOLLOWING compounds and formal charge on each ATOM are as  Formal charge on an atom in a Lewis structure = (total number of valence electrons in free atom) - [total number of non-bonding (lonepairs) electrons) `(1)/(2)` [total number of bonding or shared electrons] (i) Formal charge on H = 1 - 0 - `(1)/(2) xx 2 = 0 ` Formal charge on `N = 5 - 0 - (1)/(2) xx 8 = 1`  Formal charge on O (1) = 6 - 4- `(1)/(2) xx 4 = 0 ` Formal charge on`O(2) = 6 - 4 - (1)/(2) xx ` 4 =0 Formal charge on O(3) = 6- 6 - `(1)/(2) xx 2 = - 1 ` (ii)Formal charge on O(1) = 6 -4 - `(1)/(2) xx 4 = 0 ` Formal charge on `N = 5 - 1 - (1)/(2) xx 6 = + 1 ` Formal charge on O(2) `6 - 6 - (1)/(2) xx 2= -1 ` (iii)  Formal charge on H(1) or H(2) = 1 - 0 - `(1)/(2) xx 2 = 0` Formal charge on O(1) or O(3)= 6 - 4 - `(1)/(2) xx 4 = 0 ` Formal charge on O(2) or O(4) = 6 - 6 - `(1)/(2) xx2 = -1` Formal charge on S = 6 - 0 - `(1)/(2) xx 8 = + 2 ` |
|
| 27. |
What is green fuel ? Give the information about waste recycling. |
|
Answer» Solution :Fuel obtained from plastic WASTE has high octane rating. It contains no lead and is known as "green fuel". Due to recent developments made in CHEMICAL and textile industries, clothes will be made from recycled plastic waste. TECHNOLOGY has now been developed to produce electricity from the garbage. A pilot plant has been set up, where after removing ferrous metals, plastic, glass, paper etc. from garbage, it is mixed with water. It is mixed with water. It is then caltured with bacterial species for producing methane, which is known as biogas. Biogas is used to produce electricity and the REMAINING PRODUCT is used as manure. |
|
| 28. |
What will be the stability order of Group-14 ? |
|
Answer» `CX_2 LT SiX_2 lt GeX_2 lt SnX_2 lt PbX_2` |
|
| 29. |
Which of the following alkene gives four monochloro (structural isomer) products after hydrogenation ? |
|
Answer» Pent-2-ene |
|
| 30. |
Which of the following pairs would have higher boiling points ? |
|
Answer» Solution :(a) (II) because carbonyl GROUP is more polar than DOUBLE bond. (b) (I) acid forms dimar due to `H-`bonding. ( C) (II) cis-compound has high dipole moment `(mu)`, so more polar than trans-compound `(mu = 0)` (d) (I) it forms `H-`bonding1 through `H` atom in `(-NH)` group. ( e) (II) it forms `H-`bonding (f) (II) alcohols form `H`-bonding. (G) (II) two `(OH)` groups can form more `H-`bonding with each other. (h) (I) alcohold form `H-`bonding (i) (II) because of high MOLECULAR mass and large size, have larger van der Waals forces of attraction. |
|
| 31. |
What is added to increase the setting velocity of plaster of paris? |
|
Answer» Alum |
|
| 32. |
When an amide is reduced with sodium/alcohol is yields |
|
Answer» PRIMARY AMINE |
|
| 33. |
What are the uses of Heavy Water ? |
|
Answer» Solution :a. Heavy water is USED as a coolant and MODERATOR for neutrons in nuclear REACTORS. b. Heavy water is used in the preparation of deuterium and deuterated compounds. C. It is used as a tracer element in metabolic processes. |
|
| 34. |
The total number of acyclic isomers for C_(3)H_(4) is |
|
Answer» 1 |
|
| 35. |
Which of the following refers to the original description of reduction? |
|
Answer» Addition of hydrongen `2ZnO(s) overset(Delta)(rarr) 2ZN(s) +O_2(g)`. |
|
| 36. |
Which properties of gas can be measured ? |
| Answer» Solution :(i) Mass (ii) VOLUME (III) Temperature (IV) PRESSURE | |
| 37. |
Which one of the following will undergo S reaction faster? |
|
Answer»

|
|
| 38. |
Which of the followingis similar to graphite ? |
| Answer» Solution :BN haslayeredstructuresimilar to graphite. | |
| 39. |
Which of the following conformers for ethylene glycol is most stable ? |
|
Answer»
|
|
| 40. |
The thermochemical equation for solid and liquid rocaket fuel are given below 2Al(s) + 1(1)/(2)O_(g) rarr Al_(2)O_(3)(s) , Delta H = - 1667.8 kJ mol^(-1) H_(2)(g) + (1)/(2) O_(2)(g) rarr H_(2)O(l) , Delta H = - 285.9 kJ mol^(-1) (a) If equal masses fo aluminiumand hydrogen are used, which is a better rocket fuel? (b ) Determine Delta H for the reaction : Al_(2)O_(3)(s)rarr 2Al(s) + 1(1)/(2)O_(2)(g) |
|
Answer» Solution :(a) From the first given equation. 2 moles of Al `( i.e., 2 xx 27 g = 54G)` on combustion GIVE heat `= 1667.8 KJ` `:. 1 g` of Al combustion givesheat ` = (1667.8 ) / ( 54) = 30.9kJ` From the second given equation, 1 mole of `H_(2)( = 2g) ` on combustion gives heat `= 285.9 kJ` `:.` 1 g of `H_(2)` on combustion gives heat `= (285.9) /( 2) = 142.95kJ` Thus, `H_(2)` is a better rocket fuel. (b) Writing the reverse of the first REACTION, we have `Al_(2)O_(3)(s) rarr2Al(s) +1(1)/(2) O_(2)(g), DeltaH = + 1667.8 kJ mol^(-1)` Thus, for the reaction given in part (b) of the problem, `Delta H = + 1667.8 kJ mol^(-1)` |
|
| 41. |
What happens when quick lime is heated with silica? |
|
Answer» Solution :Quick lime (CaO) COMBINES with silica `(SiO_(2))` to FORM SLAG. `CaO+SiO_(2) overset("HEAT") to CaSiO_(3)` |
|
| 42. |
Which of the following is a redox reaction |
|
Answer» `CH_(3)COOH + C_(2)H_(5)OH rarr CH_(3) COOC_(2)H_(5)+H_(2)O` 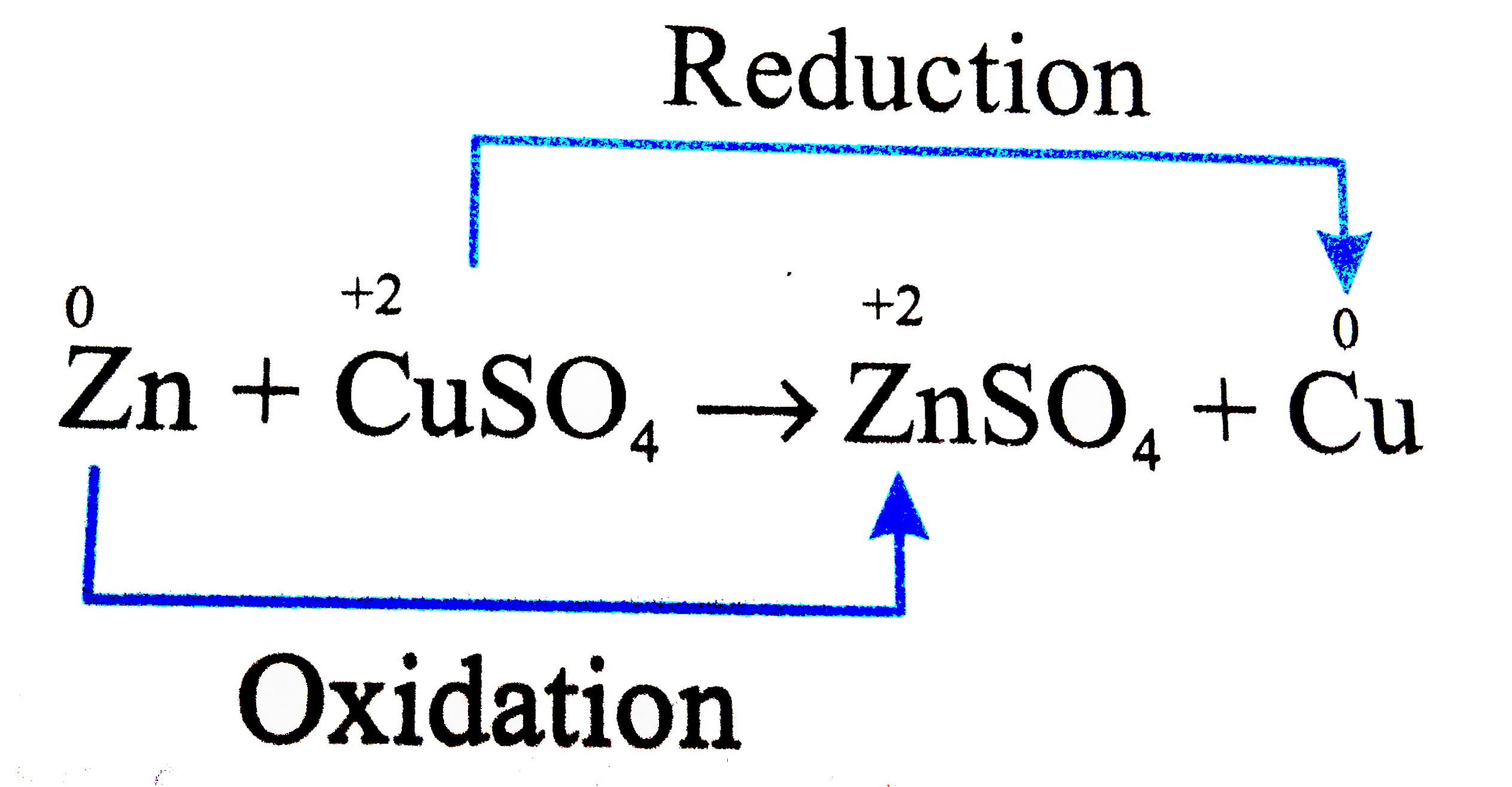
|
|
| 43. |
Which of the following atomic numbers represent group 13 elements ? |
| Answer» Solution :Group 13 ELEMENTS have atomic number 5,13 31, 49 and 81. | |
| 44. |
Write the balanced equations for the reactions between (i) Na_(2)O_(2) and water (ii) KO_(2) and water (iii) Na_(2)O and CO_(2) |
|
Answer» Solution :(a) `Na_(2)O_(2) (s) + 2H_(2)O (l) to 2 NaOH (aq) + H_(2)O_(2) (aq)` (B) `2KO_(2) (s) + 2H_(2)O (l) to 2 KOH (aq) + H_(2)O_(2) (aq) + O_(2)(g)` or `4 KO_(2) (s) + 2H_(2)O (l) to 4KOH (aq) + 3O_(2) (g) ` (c) `Na_(2) O + CO_(2) to Na_(2) CO_(3)` |
|
| 45. |
Which statement is incorrect |
|
Answer» Phenol is a weak acid  `:. C-C` bond is FORMED. Hydroboration-oxidation: `RCH=CR_(2)^(') underset((II) H_(2)O_(2), OH^(-))OVERSET((i) B_(2)H_(6))rarr R-underset(OH)underset(|)(CH)-CHR_(2)^(')` `:. C-O` bond is formed. Cannizzaro reaction: `2RCHO+50% NaOH rarr RCH_(2)OH+RCOONa` `:. C-OH` and `C=O` bonds are formed. Reaction of `1^(@)` alcohol by PCC `RCH_(2)OH underset([O])overset(PC C)rarrRCHO` `:. C=O` bond is formed. |
|
| 46. |
Tungsten has a density of 19.35 g cm^(-3) and the length of the side of the unit cell is 316 pm. The unit cell in the most important crystalline form of tungsten is the body centred cubic unit cell. How many atoms of the element does 50 g of the element contain ? |
|
Answer» |
|
| 47. |
Write the structure of the alkene which on reductive ozonolysis gives butanone and ethanal. |
Answer» SOLUTION :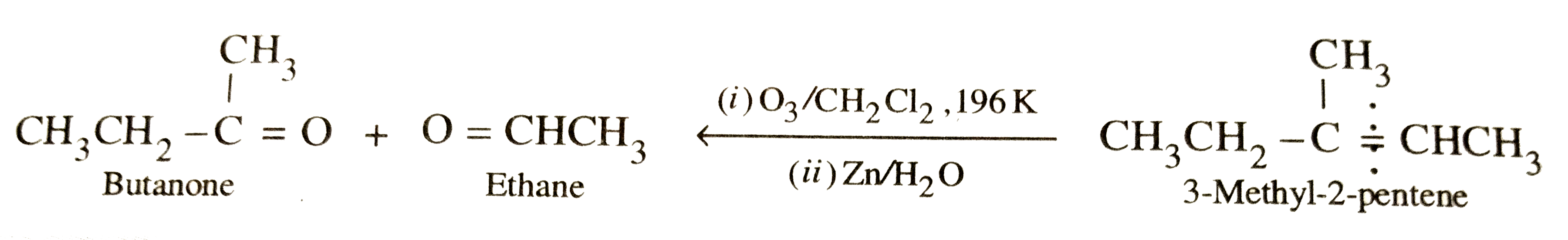
|
|
| 48. |
Which of the following will show geometrical isomerism? |
|
Answer»
|
|
| 49. |
Two elements 'P' and 'Q' combine to form a compound. Atomic mass of 'p' is 12 and 'Q' is 16. percentage of 'P' in the compound is 27.3. What will be the empirical formula of the compound ? |
|
Answer» `P_2Q_2` 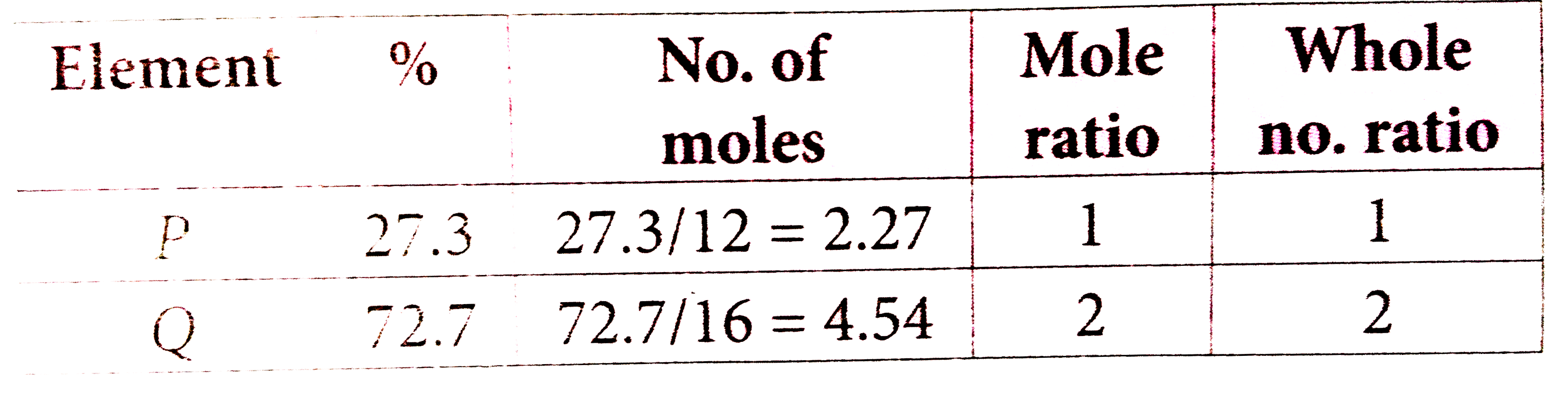 EMPIRICAL FORMULA `=PQ_2` |
|