Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which of the following sets of quantum number is correct for an electron in 4f orbitals ? |
|
Answer» n = 4,1 = 3, m = + 4, S = +1/2 |
|
| 2. |
Which of the following types of forces bind together the carbon atoms in diamond ? |
|
Answer» Ionic |
|
| 3. |
Why an Insert can walk on ordinary water but sinks in soapy water ? |
| Answer» SOLUTION :This is because soapy WATER has REDUCED SURFACE TENSION. | |
| 4. |
Van der Waal's constant a and b are related with _________ respectively. |
|
Answer» attrative force and VOLUME of molecules. |
|
| 5. |
When sulphuric acid is added carefully to water, what is formedm a compound or mixture ? |
|
Answer» |
|
| 6. |
Which of the following statements is wrong : |
|
Answer» `A`. HYPER conjugation is the simultaneous shift of SIGMA and `pi` electrons at 1, 3-positions without themovement of any atom |
|
| 7. |
The salt of strong acid and strong base does not undergo hydrolysis. Comment. |
| Answer» Solution :In this case, neither the cation nor the anion undergoes hydrolysis. Therefore, the solution remains neutral. For EXAMPLE, in the AQUEOUS solution of NaCl, its ions `Na^+` and `Cl^-` have no tendency to react with the `H^+` or `OH^-` ions of water. This is because the possible products of such reaction are NAOH and HCl, which are themselves completely dissociated. As a RESULT, there is no change in the concentration of `H^+` and `OH^-` ions and hence the solution CONTINUES to remain neutral. | |
| 8. |
What are main products formed during oxidation of glycerol with Fenton's reagent ? |
|
Answer» GLYCERALDEHYDE and Dihydroxyacetone |
|
| 9. |
Whichof the following are not resolvable? |
|
Answer» `2,3-` Pentadiene 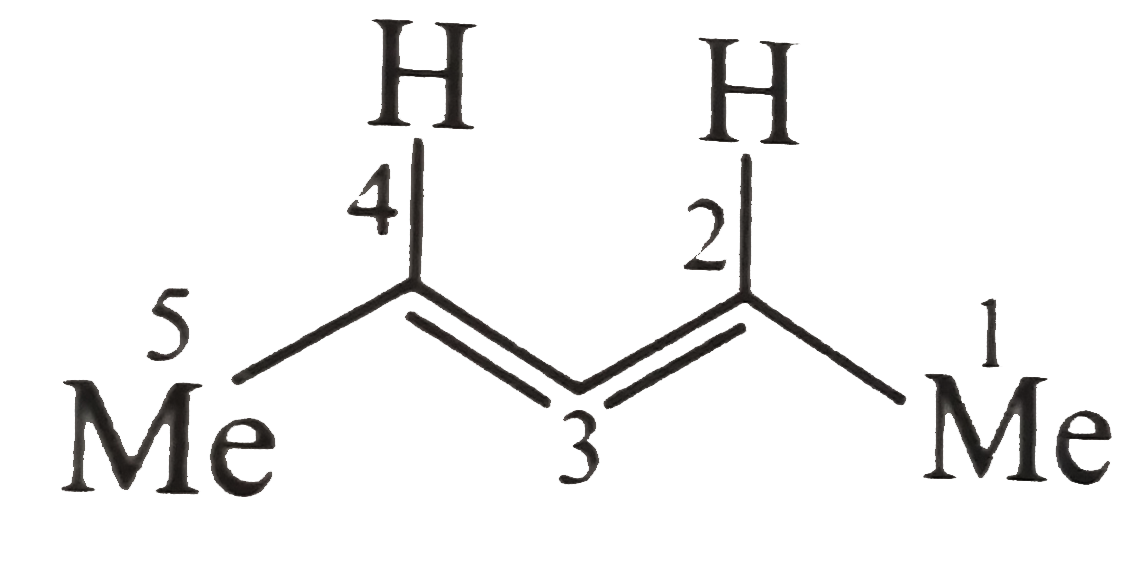 ,(Allenes of this type SHOW `O.I`) ,(Allenes of this type SHOW `O.I`) b.The cis form shows `O.I` c. The TRANS form does not show `O.I` due to the CENTRE of symmetry. d. Does not show `O.I`. Due to ALTERNATING AXIS of symmetry. |
|
| 10. |
The van der Waal's equation of state for 1 mole real gas is (P + (a)/(V^(2))) (v - b) = RT....(i) The Virial equation for 1 mole real gas is PV = RT [1 + (x)/(V) + (y)/(V^(2)) + (z)/(V^(3))+....." To higher power of " V] ....(ii) when x, y and z are constants which are known as 2nd, 3rd adn 4th co-efficients respectively. The temperature at which real gas obeys ideal gas equation i.e., (PV = nRT) is known as Boyle's temperature. Answer the following questions on the basis of the above write up : If equation (ii) can be one of the way of writing of equation (i), then what will be the value of y? |
|
Answer» `B - (a)/(RT)` |
|
| 11. |
The van der Waal's equation of state for 1 mole real gas is (P + (a)/(V^(2))) (v - b) = RT....(i) The Virial equation for 1 mole real gas is PV = RT [1 + (x)/(V) + (y)/(V^(2)) + (z)/(V^(3))+....." To higher power of " V] ....(ii) when x, y and z are constants which are known as 2nd, 3rd adn 4th co-efficients respectively. The temperature at which real gas obeys ideal gas equation i.e., (PV = nRT) is known as Boyle's temperature. Answer the following questions on the basis of the above write up : Which of the following is the incorrect statement about the Boyle's temperature ? |
|
Answer» Temperature at which FIRST virial co-efficient BECOME zero |
|
| 12. |
The van der Waal's equation of state for 1 mole real gas is (P + (a)/(V^(2))) (v - b) = RT....(i) The Virial equation for 1 mole real gas is PV = RT [1 + (x)/(V) + (y)/(V^(2)) + (z)/(V^(3))+....." To higher power of " V] ....(ii) when x, y and z are constants which are known as 2nd, 3rd adn 4th co-efficients respectively. The temperature at which real gas obeys ideal gas equation i.e., (PV = nRT) is known as Boyle's temperature. Answer the following questions on the basis of the above write up : If the critical temperature of the gas be T_(C) = (8a)/(27 Rb) and T_(B) is the Boyle's temperature, then which of the following is the correct relation between T_(C) " and " T_(B)? |
|
Answer» `T_(C) = (8)/(28) T_(B)` `:. (T_(C))/(T_(B)) = (8)/(27)` |
|
| 13. |
The van der Waal's equation of state for 1 mole real gas is (P + (a)/(V^(2))) (v - b) = RT....(i) The Virial equation for 1 mole real gas is PV = RT [1 + (x)/(V) + (y)/(V^(2)) + (z)/(V^(3))+....." To higher power of " V] ....(ii) when x, y and z are constants which are known as 2nd, 3rd adn 4th co-efficients respectively. The temperature at which real gas obeys ideal gas equation i.e., (PV = nRT) is known as Boyle's temperature. Answer the following questions on the basis of the above write up : The third virial coefficient of He gas is 4 xx 10^(-2) ("litre/mole")^(2), then what will be volume of 2 mole He gas at NTP |
|
Answer» 22.0 litres i.e., `b = 2 xx 10^(-1)` litre/mole EXTENDED volume for 2 mole REAL gas. `:. V_("real") = V_(C) + nb = 44.8 + 0.4 = 45.3` litre |
|
| 15. |
What is the major poduct expected from the following reaction? |
|
Answer» `CH_(3)-underset(Cl)underset(|)(C)=CH_(2)` |
|
| 17. |
Which of the following is amide group? |
|
Answer» `CH_(3)NHCH_(2)CH_(3)` |
|
| 18. |
The s-block elements are characterised by their larger atomic sizes, lower ionisation enthalpies, invariable +1 oxidation state and solubilities of their oxosalts. In the light of these features describe the nature of their oxides, halides and oxosalts. |
|
Answer» Solution : The atom of alkali metals have large size and due to this they readily form cations and so their compounds are ionic in nature. Alkali metals have +1 oxidation state and give ionic compounds. Alkali metals give three TYPES of oxides - Normal oxides`(M_(2)O)`, Peroxide `(M_(2)O_(2))` and Super oxides`(MO_(2))`. Basic character of normal oxides increases from `Li_(2)O` to `Cs_(2)O` due to increase in their ionic character. Halides of alkali metals MX are also ionic except Lix, which is covalent because of small size and high polarizing power. Oxosalts: All alkali metals from solid carbonates of general formula `M_(2)C0_(3)`. Carbonates are STABLE except `Li_(2)CO_(3)` due to high polarising capacity of `Li^(+)`which is unstable and decomposes. All the alkali metals (except Li) form solid bicarbonates `MHCO_(3)`. All alkali metals form nitrates having formula `MNO_(3)`. They are colourless, water SOLUBLE, electrovalent compounds. |
|
| 19. |
To remove stains from clothes "____________" is used. |
|
Answer» `NaCl` |
|
| 20. |
Which one of the following is a metalloid? |
|
Answer» N |
|
| 21. |
Which one of the following is a homogeneous mixture ? |
|
Answer» SEA WATER |
|
| 22. |
What is the effect of temperature of the reactions? (i) PCl_(5)(g)hArrPCl_(3)(g)+Cl_(2)(g) (ii) N_(2)(g)+O_(2)(g)hArr2NO(g). |
| Answer» Solution :(i) The EQUILIBRIUM will SHIFT towards the backward REACTION. (ii) No effect. | |
| 24. |
Through a solution containing Cu^(2+) and Ni^(2+),H_(2)S gas is passed after adding dil HCl, which will precipitate out and why ? |
| Answer» SOLUTION :`CU^(2+)` will precipitate out because in the acidic MEDIUM, only IONIC product `[Cu^(2+)][S^(2-)]` exceeds the solubility product of CUS. | |
| 25. |
Which of the following has highest stable oxidation state ? |
|
Answer» Al |
|
| 26. |
What is the basic difference the terms electron gain enthalpy and electronegativity ? |
Answer» SOLUTION :
|
|
| 27. |
Which of the following decreases in going down the halogen group ? |
|
Answer» ionic RADIUS |
|
| 28. |
Which one of the following is not used as an adsorbent in chromatography? |
|
Answer» Alumina |
|
| 29. |
What is meant by active mass? Give its unit. |
|
Answer» Solution :The active mass represents the molar concentration of the REACTANTS (or) products `"Active mass "= (n)/(V) = ("number of moles ")/("VOLUME in litre ")` unit of active mass= MOL `dm^(-3)` (or ) mol `L^(-1)` |
|
| 30. |
Write the conditions in terms of DeltaHand DeltaS when a reaction would be always spontaneous. |
| Answer» SOLUTION :The reaction would be always SPONTANEOUS when both energy factor and randmness factor favour it, i.e., `DELTAH =-ve` and `DELTAS= + ve` | |
| 31. |
Which of the following are stronger bases than ammonia |
|
Answer» `(CH_(3))_(2)NH` |
|
| 32. |
Which has maximum pKa value |
|
Answer» `MeO-CH_(2)-COOH` |
|
| 33. |
The solubility of AgCl(s) with solubility product 1.6 xx 10^(-10) in 0.1 M NaCl solution would be |
|
Answer» `1.6xx10^(-11)` M `{:(AgCl hArr , Ag^(+)+, Cl^-),(0.1 M, 0,0),(x-S,S,S+0.1):}` `K_(SP)=1.6xx10^(-10) = [Ag^+][Ci]=[S]` [S+0.1] `because K_(sp)` is small , S is neglected with respect to 0.1 M `1.6xx10^(-10) =Sxx0.1` `S=1.6xx10^(-9)` M |
|
| 34. |
Which of the following alcohols is least soluble in water? |
|
Answer» `CH_(3)OH` |
|
| 35. |
Define Biochemical Oxygen Demand (BOD). |
| Answer» Solution :The Amount of OXYGEN required by BACTERIA to BREAK down the organic matter present in certain VOLUME of sample of water is CALLED BOD. | |
| 36. |
Which of thefollowingsets ofionsrepresentsa collectionof isoelectronicspecies ? |
|
Answer» `K^(+) ,C1^(-) ,Mg^(2+) , SC^(3+)` `Ca^(2+)(20- 2 =18) , Sc^(3+)(21 = 3=18)` represent acollectionof ISOELECTRONIC species. |
|
| 37. |
Which of the following equation depicts reducing nature of H_(2)O_(2) ? |
|
Answer» `2[FE(CN)_(6)]^(4-) + 2H^(+) + H_(2)O_(2) to 2[Fe(CN)_(6)]^(3-) + 2H_(2)O` |
|
| 38. |
Theshortestwavelengthin hydrogenspectrum oflaymanserieswhen R_(H) = 109678 cm^(-1)' is |
|
Answer» 1002.7A |
|
| 39. |
Why is the solution of an alkali metal in ammonia blue? |
| Answer» SOLUTION :BLUE, CONDUCTING, ammoniated ELECTRONS | |
| 40. |
Which of the following cannot exhibit hypercojugation - |
|
Answer» `CH_(3)OVERSET(.)CH_(2)` |
|
| 41. |
Which two organic compound are purify by sublimation technique? |
| Answer» SOLUTION :(i) CAMPHOUR (II) Nalpthaline | |
| 42. |
Which relationship holds true for Boltzmann constnat 'K' and gas constant 'R' (N_(A) is Avogadro number)? |
|
Answer» `R=kxxN_(A)` |
|
| 43. |
Which of the following statements is true about the following conformer (X) |
|
Answer» X is the most stable CONFORMER of meso-2,3-Dichloro-2,3-dimethylbutanedioc acid |
|
| 44. |
Which of the following statement is correct for the spontaneous reaction ? |
|
Answer» ENTROPY of the SYSTEM is ALWAYS increased. |
|
| 45. |
What is thechangeobservein ironon heatingwith differenttemperature ? |
|
Answer» Solution :Whenan ironis heateditscolouris beingchangedas under . Whenan ironrod is heatedits COLOURIS beingchangedas under. dull red `to`more and`to`WHITE `to `BLUE `to `frequencyv increasing `to |
|
| 46. |
The wavelength of the first member of theBalmer series in hydrogen spectrum is xÅ. Then the wave length ("in "Å) of the first member of Lyman series in the same spectrum |
|
Answer» `(5)/(27) x ` For Balmer series , `bar(upsilon_1) = R (1/((1)^2) - 1/((3)^2))` |
|
| 47. |
X' is a smallest optically active alkanol. On dehydration it can form Y number of alkenes (including stereoisomers). On reaction with Lucas reagent it forms Z number of alkyl halides (including stereoisomers). Report your answer as ZY. |
|
Answer» |
|
| 48. |
Which among the following is most soluble in alcohol ? |
|
Answer» `CsClO_(4)` |
|
| 49. |
Which of the following mixtures can act as a buffer? |
|
Answer» `NaOH + HCOONA(1:1 "molar ratio")` |
|
| 50. |
When 6.3 g of NaHCO_(3) (sodium bicarbonate) is added to a solution of CH_(3)COOH (acetic acid) weighing 15 g, it is observed that 3.3 g CO_(2) is released to the atmosphere. The residue is found to weigh 18.0 g. Show that the data is in agreement with the law of conservation of mass. |
|
Answer» Solution :The chemical EQUATION for the reaction is : `NaHCO_(3)+CH_(2)COOHrarrCH_(3)COONa+H_(2)O+CO_(2)` Sod. Bicarbonate Acetic ACID `underset(("Residue"))(underbrace("Sod. acetate"))` Mass of reactants = Mass of `NaHCO_(3) + "Mass of "CH_(3)COOH = 6.3 + 15 = 21.3 g` Mass of product = Mass of residue + Mass of `CO_(2)=18 + 3.3 = 21.3 g` Since these is no change in mass during the chemical reaction the DATA illustrates the Law of conservation of Mass. |
|