Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
What is the composition of 'London smog'? |
| Answer» SOLUTION :FOG of `H_2SO_4` DROPLETS DEPOSITED on the PARTICULATES | |
| 2. |
Volume strength of perhydrol is |
|
Answer» 15 |
|
| 3. |
The substance that does not have sharp ,melting point is |
|
Answer» KCl |
|
| 4. |
What will be the value of DeltaG for the ice at 8°C temperature ? |
|
Answer» ZERO |
|
| 5. |
What is the IUPAC name of the insecticide DDT? Why is their use banned in most of the countries? |
|
Answer» Solution :(i) The lUPAC name of the insecticide DDT is p, p-dichloro-diphenyl TRICHLOROETHANE. (ii) Even DDT is an effective insecticide. Now-adays it is banned because of its long TERM toxic effects. (iii) DDT is very persistent in the environment and it has a HIGH affinity for fatty tissues. As a result, DDT gets accumulated in animal tissue tat, in particular that of birds of prey with SUBSEQUENT thinning of their eggs shells and impacting their rate of REPRODUCTION. That is why DDT is banned in most of the countries. |
|
| 6. |
What is the order of reactivity of hydrogen atoms attached to carbon atom in an alkene? |
|
Answer» `3^(@) GT 1^(@) gt 2^(@)` |
|
| 7. |
Which of the following reaction(s) give(s) blue precipitate? |
|
Answer» `HG^(2+)+Co^(2+)+4SCN^(-) (AQ) RARR` |
|
| 8. |
What do you understand by bond pairs and lóne pairs of electrons? Illustrate by giving one example of each type. |
|
Answer» Solution :The electron pair involved in sharing between two atoms during covalent BONDING is CALLED SHARED pair or bond pair. At the same time, the electron pair which is not involved in sharing is called lone pair of electrons. For example, In `CH_(4),[Hunderset(H)UNDERSET(* *)OVERSET(H)overset(* *)C:H]` there are only 4 bond pairs, but in `H_(2)O,`  there are two bond pairs and two lone pairs. |
|
| 9. |
The weight of one litre of a gas at latm pressure and 300K is 4g. At what temperature the weight of the gas is 4g when the pressure is made 0.5 atm and volume is 1 litre? |
| Answer» Answer :B | |
| 10. |
Which elements were arranged based upon their properties by Mendeleev ? |
| Answer» Solution :Atomic NUMBER of IODINE is LESS Telunium. He arranged TE in group VI and iodine VII group. Iodine (I) `126.9 lt `Tellurium (Te) `127.6` | |
| 11. |
underline(A)overset(4KOH,O_(2))to2underline(B)+2H_(2)O 3underline(B)overset(4HCl)underset(("Purple"))(2underline(C))+MnO_(2)+2H_(2)O 2underline(C)overset(H_(2)O,Kl)to2underline(A)+2KOH+underline(D) In the above sequence of reactions, underline(A) and underline(D), respectively, are: |
|
Answer» `KlO_(3) and MnO_(2)` |
|
| 12. |
The volume of gas is reduced to half from its original volume. The specific heat will be .................... |
|
Answer» reduced to half |
|
| 13. |
What is dihedral angle in describing the structure of H_(2)O_(2) ? How it changes with a change in physical state ? |
| Answer» Solution :Dihedral angle is the angle between TWO pages of the open BOOK structure of hydrogen peroxide. The dihedral angle in gas phase of hydrogen peroxide is `11^(0)30.` and in solid STATE is `90^(0)15.`. This decrease in the dihedral angle is ATTRIBUTED to hydrogen bonding. | |
| 14. |
Which order is correct regarding stability of intermediates. (I) CH_(3)-CH_(2)^-,"(II) "CH_(2)=CH^-"(III) "CH equiv C^- |
|
Answer» a. `I = II = III` |
|
| 15. |
Total number of atoms in 44g of CO_2 is |
|
Answer» `6.02 xx 10^(23)` |
|
| 16. |
What type of magnetism is shown by a substance if magnetic moments of domains are arranged in same direction ? |
| Answer» SOLUTION :FERROMAGNETIC SUBSTANCES | |
| 17. |
Wurtz reaction of methyl iodide yields an organic compound X. Which of the following reaction also yields X ? |
|
Answer» `C_2H_5Cl + LiAlH_4 to ` 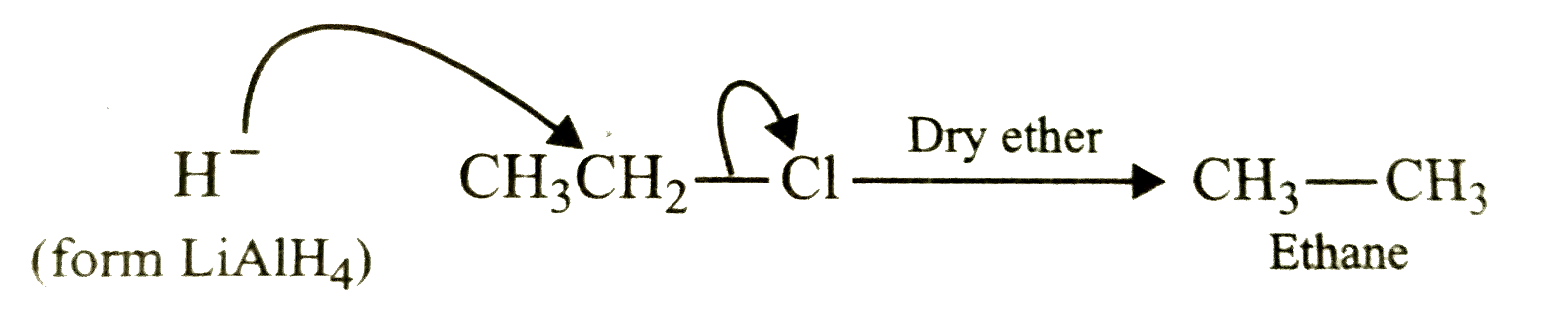 `LiAlH_4` reduces `1^@` alkyl HALIDES to ALKANES . |
|
| 19. |
Which of the following is a strong electrolyte |
|
Answer» `NH_4OH` |
|
| 20. |
The reagents used in the preparation of chloroform are …………………. . |
| Answer» SOLUTION :ETHANOL +BLEACHING POWDER | |
| 21. |
The successive ionization energy values for an element X are given below: (1) 1st ionization energy = 410 kJ mol^(-1) (2) 2nd ionization energy = 820 kJ mol^(-1) (3) 3rd ionization energy = 1100kJ mol^(-1) (4) 4th ionization energy = 1500 kJ mol^(-1) (5) 5th ionization energy =3200 kJ mol^(-1) Find out the number of valence electrons for the atom X |
|
Answer» 1st ionization energy =410 KJ `MOL^(-1)` |
|
| 23. |
Why alkane is known as "Paraffin" ? |
|
Answer» Solution :Saturated hydrocarbons are inert at normal CONDITION because they do not react with acid, BASE and any other REACTANTS. Paraffin is latin word : Parum means LITTLE and affinis means reactive, which suggest LESS reactive. Alkane is inert and less reactive so it is known as paraffins. |
|
| 24. |
The value of K_(c) " for the reaction , " 2 A hArr B+C " is "2*0 xx 10^(-3)" AT a given time, the composition of the reaction mixture is "[ A] = [B] = [C] = 3 xx 10^(-4) M In which direction , the reaction will proceed ? |
|
Answer» Solution :For the given reaction , `Q_(c) = ([B] [C])/[A]^(2)= ((3 xx 10^(-4)) (3 xx 10^(-4)))/((3 xx 10^(-4)) ^(2)) = 1` Thus , ` Q_(c) gt K_(c).` Hence , the reaction will proceed in the backward direction. |
|
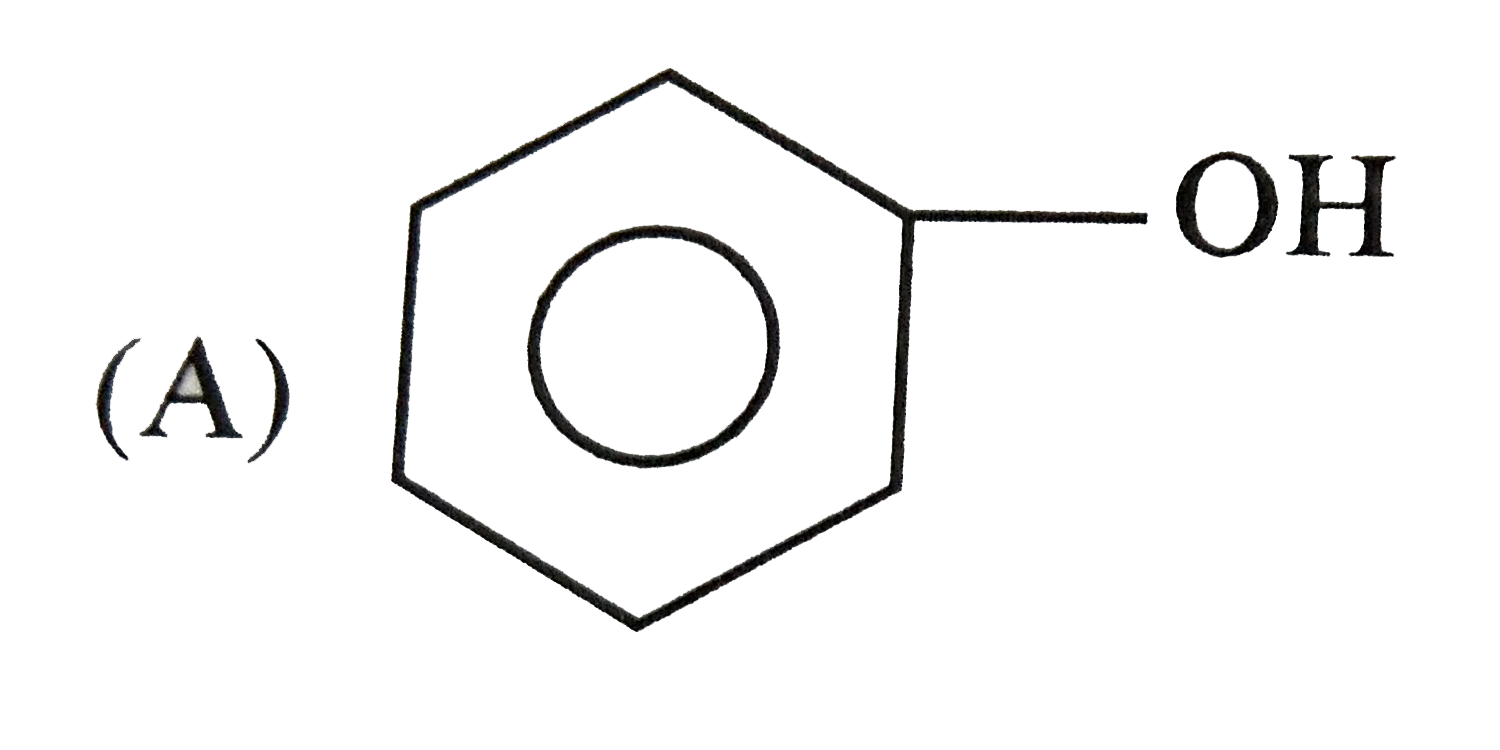
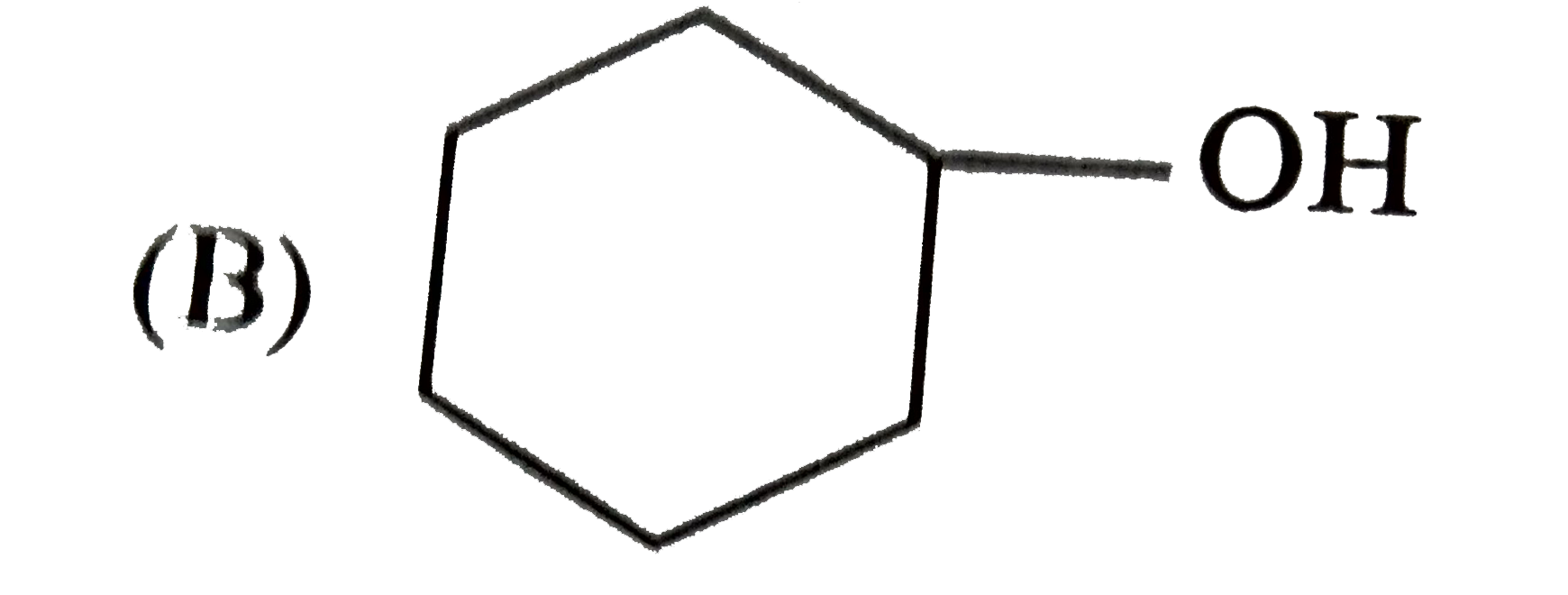
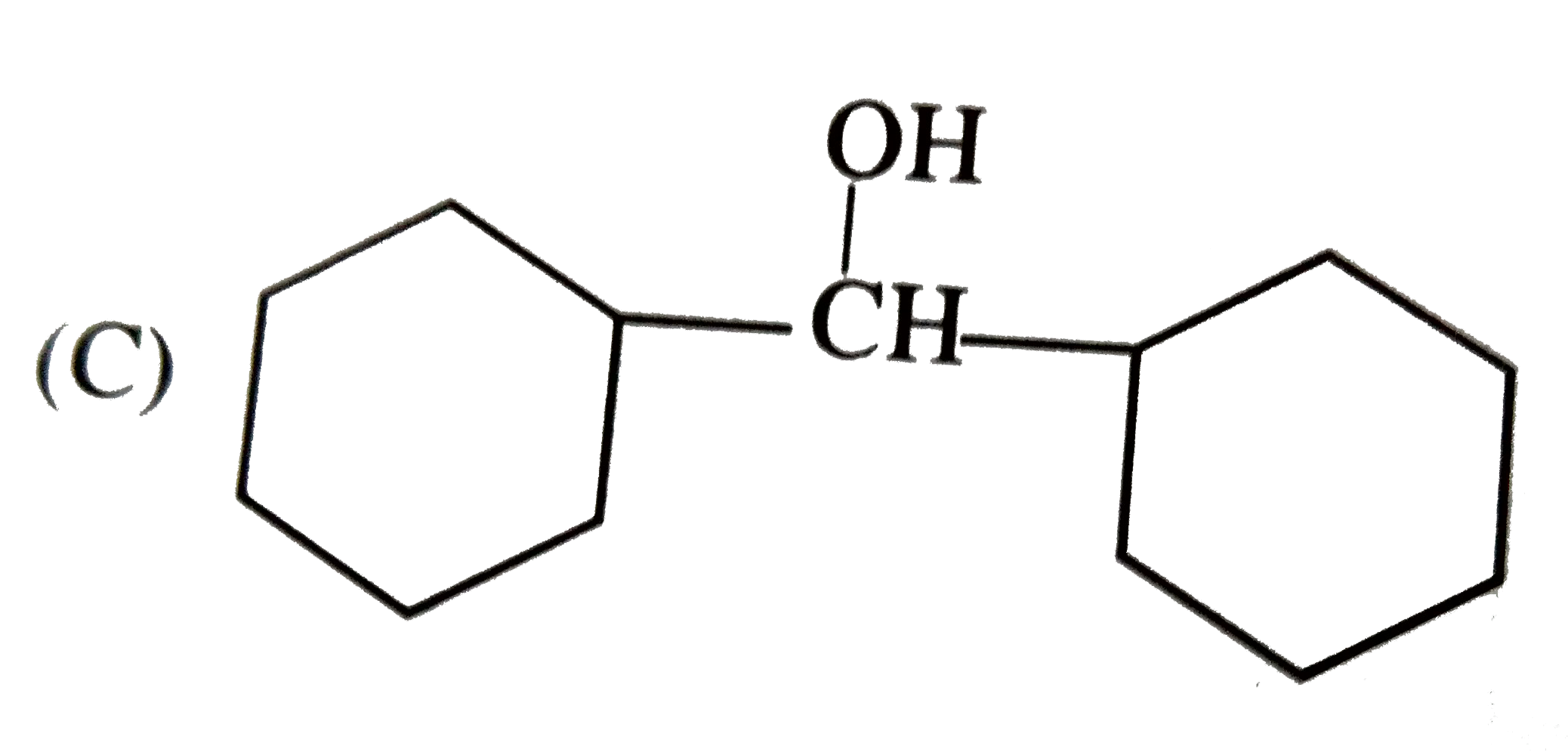
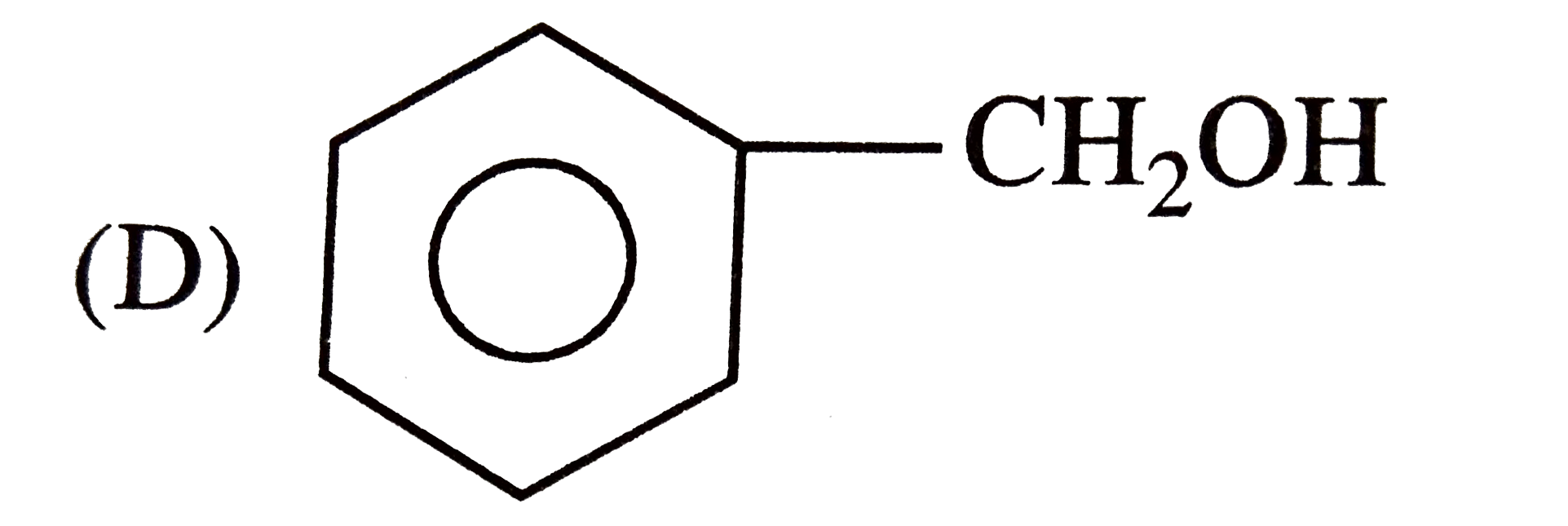
| 25. |
Which one of the following compounds has the most acidic nature? |
|
Answer»
|
|
| 26. |
Which of the following statements is CORRECT for Joule Thomson effect in liquefaction of permanent gases ? |
|
Answer» When a work of expansion is DONE at the cost of internal energy, the temperatureof GAS DROPS further. |
|
| 27. |
What is shell ? |
| Answer» Solution :The principle ENERGY LEVEL or principle QUANTUM number (N) is KNOWN as shell. | |
| 28. |
What is the pH of a 0.50 M aqueous NaCN solution ? pK_(b)of CN^(-) is 4.70. |
|
Answer» Solution :`NaCN + H_(2)O HARR NaOH + HCN` or `CN^(-) + H_(2)O hArr OH^(-) HCN` `K_(B)=([OH^(-)][HCN])/([CN^(_)])=([OH^(-)]^(2))/([CN^(-)])` `:. pK_(b) = - LOG K_(b) = - 2 log [OH^(-)]+log [CN^(-)]` or `4.70 = - 2 log [OH^(-)]+ log (0.5)`[`:' [CN^(-)]=[NaCN]]` or` 2log [OH^(-)]=-5.00 or log [OH^(-)]=-2.5` `-log [OH^(-)]=2.5` `p_(OH)=2.5` `pH = 14-25 = 11.5` |
|
| 29. |
The temperature at which oxygen molecules have the same mean velocity as that of Helium atoms at 0^@C is |
|
Answer» `2184^@C` |
|
| 30. |
Which one of the following is not a primary standard : |
|
Answer» OXALIC acid |
|
| 31. |
Water gas contains CO and H_(2) in mole ratio, 1:1. A nickel ore containing Ni_(2)O_(3) may be reduced into Ni as Ni_(2)O_(3)(s)+3H_(2)(g)rarr2Ni(s)+3H_(2)O(g) But due to presence of CO(g), the nickel formed get converted into Ni(CO)_(x)(g). Both the reaction occurs 100% If 240 gm water gas is needed to convert one mole of Ni_(2)O_(3)(s) into Ni(CO)_(x)(g), the value of x is: |
|
Answer» |
|
| 32. |
Which compound has incomplete octet ? |
|
Answer» `BCl_(3)` |
|
| 33. |
What possibly can the ratio be of the de Broglie wavelengths for two electrons having the same initial energy and accelerated through 50 volts and 200 volts ? |
|
Answer» `3:10` |
|
| 34. |
What is the minimum pH required to prevent the precipitation of Zn^(+2) in a solution that is 0.01 M ZnCl_(2) and saturated with 0.1 M H_(2) S (Given : K_(sp) ZnS = 10^(-21) and Ka_(1) xx Ka_(2)of H_(2)S= 10^(-20) ) |
|
Answer» `10 ^(-21)= (10 ^(_2))[S^(-2)]rArr [S^(-2) ]=10 ^(-19)M` `H_2S hArr 2H^(+) +S^(-2)rArr K_(eq)=([H^(+)][S^(-2)])/( [H_2S]) ` ` 10 ^(-20)=([H^(+) ]^(2) (10^(-19)))/(10^(-1)) rArr [H^(+) ]=0.1 M` ` PH =1` |
|
| 35. |
The wavelength of the first line in the balmer series is 656 nm . Calculate the wavelengthof thesecond lineand the limitingline in the Balmer series. |
|
Answer» SOLUTION :According to Rydberg formula, `bar(v) = (1)/(lamda) = R ((1)/(n_(1)^(2)) - (1)/(n_(2)^(2)))` For the Balmer series, `n_(1) = 2` and for the 1st line, `n_(2) = 3` `:. (1)/(656) = R ((1)/(2^(2)) - (1)/(3^(2))) = R xx ((1)/(4)- (1)/(9)) = R xx (5)/(36) = (5R)/(36)`...(i) For the second line,`n_(1) = 2, n_(2) = 4` `:. (1)/(lamda) = R ((1)/(2^(2)) - (1)/(4^(2))) = R ((1)/(4) - (1)/(16)) = R xx (3)/(16) xx (3R)/(16)`...(ii) Dividing eqn (i) by eqn. (ii) we get `(lamda)/(656) = (5)/(36) xx (16)/(3) or lamda = 485.9 nm` For the limiting line, `n_(1) = 2, n_(2) = oo` `:. (1)/(lamda) = R ((1)/(2^(2)) - (1)/(oo^(2))) = (R)/(4)`....(iii) Dividing eqn. (i) by eqn. (iii), we get `(lamda)/(656) = (5)/(36) xx 4 or lamda = 364.4 nm` ALTERNATIVELY first calculate R from eqn. (i) and substitute in eqns (ii) and (iii) |
|
| 36. |
Write IUPAC name CH_(3)-underset(CH_(3))underset(|)(CH)-underset(OH)underset(|)(CH)-CH_(3) |
|
Answer» |
|
| 37. |
Two moles of a triatomic linear gas (neglect vibration degree of freedom) are taken through a reversible processideal starting from A as shown in figure. The volume ratio (V_(B))/(V_(A))=4. If the temperature at A is -73^(@)C, then : Work done by the gas in AB process is : 6.16 kJ 308.3 kJ 9.97 kJ 0 J (ii) Total enthalpy change in both steps is : |
|
Answer» 3000 R `=-9.97kJ` (ii) (b) `DeltaH_("total")=DeltaH_(AB)+DeltaH_(BC)=nC_(p,m)DeltaT+0` `=2XX(7)/(2)xxRxx(800-200)` = 4200 R |
|
| 38. |
What is thebasicthemeof organisation in theperiodic table ? |
| Answer» Solution :The basicthemeof organisation of elements in theperiodictable ISTO SIMPLIFYAND SYSTEMATIZE thestudyof theproperties of all theelementsand millions of theircompound . Onthe BASIS ofsimilarities in CHEMICALPROPERTIES, the variouselements have now been dividedintodifferentgroups . This hasmade the studysimplebecausethe propertiesof elements are nowstudied informof groupsrather thanindividually. | |
| 39. |
Which of the following is not an electrophile? |
|
Answer» `SO_(3)` (B) `NH_(3)` is a Lewis base it has nonbonding electron pair on N. It is not an electrophile but is a nucleophile |
|
| 40. |
When an electron withdrawing or electron releasing group is attached to carbon chain, polarity is induced on the carbon atom and on the substituent attach to it. This permanent polarity is due electron displacement due to difference in electronegativities this is called inductive effect or I effect. The inducitve effect depends in the electronegativity of the substituent. The inductive effect is broadly classified as (i) negative inductive effect or -I effect. (ii) positive inductive effect or +I effect. The group which exhibits the maximum -I effect |
| Answer» Solution :(-I.E ORDER `-NO_2gt -CN GTCI GT -OH`) | |
| 41. |
When an electron withdrawing or electron releasing group is attached to carbon chain, polarity is induced on the carbon atom and on the substituent attach to it. This permanent polarity is due electron displacement due to difference in electronegativities this is called inductive effect or I effect. The inducitve effect depends in the electronegativity of the substituent. The inductive effect is broadly classified as (i) negative inductive effect or -I effect. (ii) positive inductive effect or +I effect. Which of the following statements is correct about inductive effect ? |
|
Answer» Polarization of a `SIGMA-` BOND is called inductive effect. |
|
| 42. |
When an electron withdrawing or electron releasing group is attached to carbon chain, polarity is induced on the carbon atom and on the substituent attach to it. This permanent polarity is due electron displacement due to difference in electronegativities this is called inductive effect or I effect. The inducitve effect depends in the electronegativity of the substituent. The inductive effect is broadly classified as (i) negative inductive effect or -I effect. (ii) positive inductive effect or +I effect. Stability of given anions in the decreasing order is (i) bar(C)H_(3) "(ii) "bar(C)H_(2)-NO_(2)"(iii) "bar(C)H_(2)Cl"(iv) "CH_(3)-bar(C)H_(2) |
|
Answer» `i GT II gt III gt IV` 
|
|
| 43. |
Which of the following names is correct for underset(CHO)underset(|)(C)H_(2)-underset(CHO)underset(|)(C)H-underset(CHO)underset(|)(C)H_(2)? |
|
Answer» 3-Formylpentane-1,3-dial |
|
| 44. |
Which metal is purified by Mond's carbonyl process ? |
|
Answer» Ni |
|
| 45. |
What is the tell us the direction on the process. |
| Answer» SOLUTION :It cannot TELL US the DIRECTION of the PROCESS. | |
| 46. |
Why alkali metal elements are more reactive ? |
| Answer» Solution :Because IONISATION enthalpy of alkali metal in a PERIOD is very LESS. | |
| 47. |
What is the difference in reaction of alkene with HBr in presence of peroxide and in absence of peroxide ? |
| Answer» SOLUTION :In PRESENCE of peroxide according to ANTI Markovnikov rule and in absence of peroxide according to Markovnikov rule reaction PERFORMED by alkenes. | |
| 48. |
Which of the following pairs of substances on reaction will not evolve H_2 gas ? |
|
Answer» Fe and `H_2SO_4` (aqeous) |
|
| 49. |
Which of the following statement are wrong ? |
|
Answer» Equilibrium constant of a reaction is doubled if the equilibrium CONCENTRATION of the products become double |
|