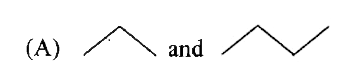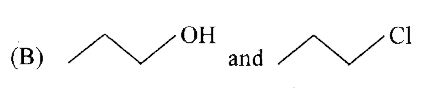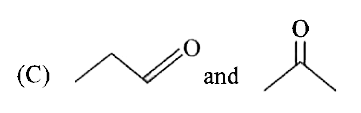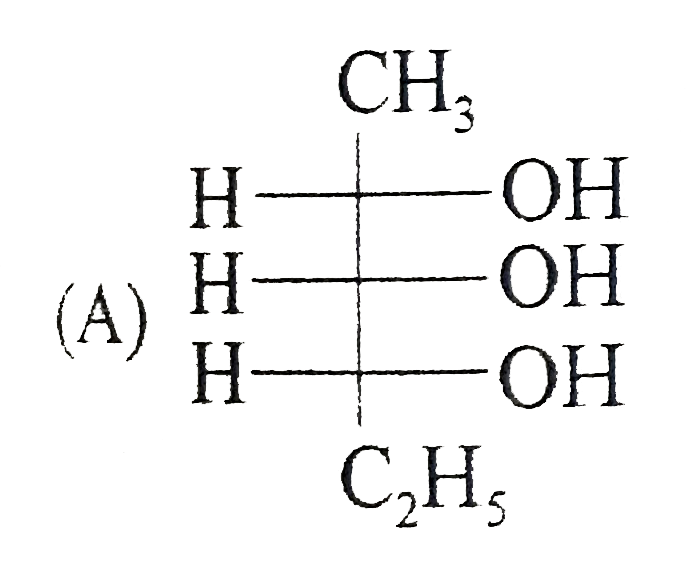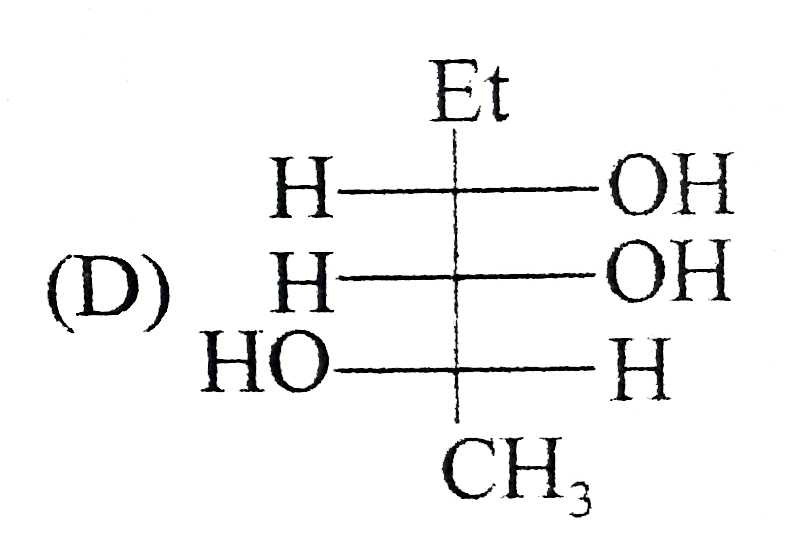Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which is the most abundant metal found in the human body? Explain how it Works inside the human body? |
|
Answer» Solution :Calcium is the most ABUNDANT metal found in the human body for egin TEETH and to (ii) 2% of an adult weight is made up of calcium. Calcium is PRESENT in teeth and bones in the form of calcium phosphate and carbonate and these make bone and teeth hard. Al Water in the human body as in the blood and inside cells. contain DISSOLVED calc (v) The calcium IONS are involved in muscle movement and sending electricity brain and along nerves |
|
| 2. |
What is the significance of Lewis symbols. |
|
Answer» Solution :The NUMBER of dots in the lewis SYMBOL represents the number of valence electrons. The common valency of the element is EITHER EQUAL to the number of dots or 8 minus the number of dots. Ex : For carbon the valency is 4. But valency of NITROGEN is 8-5=3 respectively. |
|
| 3. |
To determine the boiling point, liquid is filled in |
|
Answer» CAPILLARY TUBE |
|
| 4. |
Whichatom areindicatedby thefollowingconfiguration ? |
|
Answer» Solution :(a ) Electronicconfiguration(He ) `2s^(1)` Atomicnumber `(2+1) =3` so2=3`:. LI` (b ) electronicconfiguration: (Ne) `3s^(2) 3p^(3)` Totalelectronz=10 + 2+3 (C )electronicconfiguration(Ar ) `4s^(2) 3d^(3)` SCANDIUM (S c )element |
|
| 5. |
The substances which contain species with unpaired electrons in their orbitals behave as paramagnetic sustances. Such substances are weakly attracted by the magnetic field. The paramagnetism is expressed in terms of magnetic moment. The magnetic moment is related to the number of unpaired electrons according tot he following relation: Magnetic moment mu=sqrt(n(n+2))BM where n=number of unpaired electrons. BM stands for Bohr magneton, a unit of magnetic moment. 1BM=(eh)/(4pimc)=9.27xx10^(-24)Am^(2) or JT^(-1) Q. An ion of a d-block element has magnetic moment 5.92 BM. Select the ion among the following: |
|
Answer» `Zn^(2+)` |
|
| 6. |
Which of the following does not satisfy Huckel's rule?(a) Benzene (b) 1, 3-cyclobutadiene (c) Naphthalene (d) Furan |
| Answer» SOLUTION :1, 3-cyclobutadiene | |
| 7. |
The substances which contain species with unpaired electrons in their orbitals behave as paramagnetic sustances. Such substances are weakly attracted by the magnetic field. The paramagnetism is expressed in terms of magnetic moment. The magnetic moment is related to the number of unpaired electrons according tot he following relation: Magnetic moment mu=sqrt(n(n+2))BM where n=number of unpaired electrons. BM stands for Bohr magneton, a unit of magnetic moment. 1BM=(eh)/(4pimc)=9.27xx10^(-24)Am^(2) or JT^(-1) Q. Which of the following ions are diamagnetic? |
|
Answer» `He^(2+)` |
|
| 8. |
Which of the following non-metal reacts with ordinary water? |
|
Answer» CARBON |
|
| 9. |
What is the characteristics of inductive effect? |
| Answer» Solution :The INDUCTIVE effect is EXTENDED UPTO three carbon | |
| 10. |
Write electron dot structure (Lewis structure) of Na, Ca, B, Br, Xe, As, Ge, N^(3-). |
| Answer» SOLUTION :SEE the TEXTBOOK or SOURCE FILES | |
| 11. |
Write IUPAC names of the products obtained by addition reactions of HBr to but-l-ene: (i) in the absence of peroxide and (ii) in the presence of peroxide. |
|
Answer» Solution :(i) In absence of PEROXIDE, the product FORMED is according to Markovnikov rule. `CH_(3)-CH_(2)-CH-=CH_(2)+HBroverset("no peroxide")toCH_(3)-CH_(2)-CHBr-CH_(3)` (ii) In presence of peroxide, the product formed is according to antiMarkovnikov rule. `CH_(3)-CH_(2)-CH=CH_(2)+HBroverset("peroxide")toCH_(3)-CH_(2)-CH_(2)Br` |
|
| 12. |
Which of the following molecule/ion have not unpaired /single electron ? |
|
Answer» `N_(2)^(+)` |
|
| 13. |
What is BHC ? How will you prepare BHC ? Mention its uses. |
|
Answer» Solution :(i) BHC is Benzene hexachloride. (ii) Benzene reacts with three molecule of `Cl_(2)` in the PRESENCE of sunlight or UV light to yield BHC. This is also called as GAMMAXANE or LINDANE. 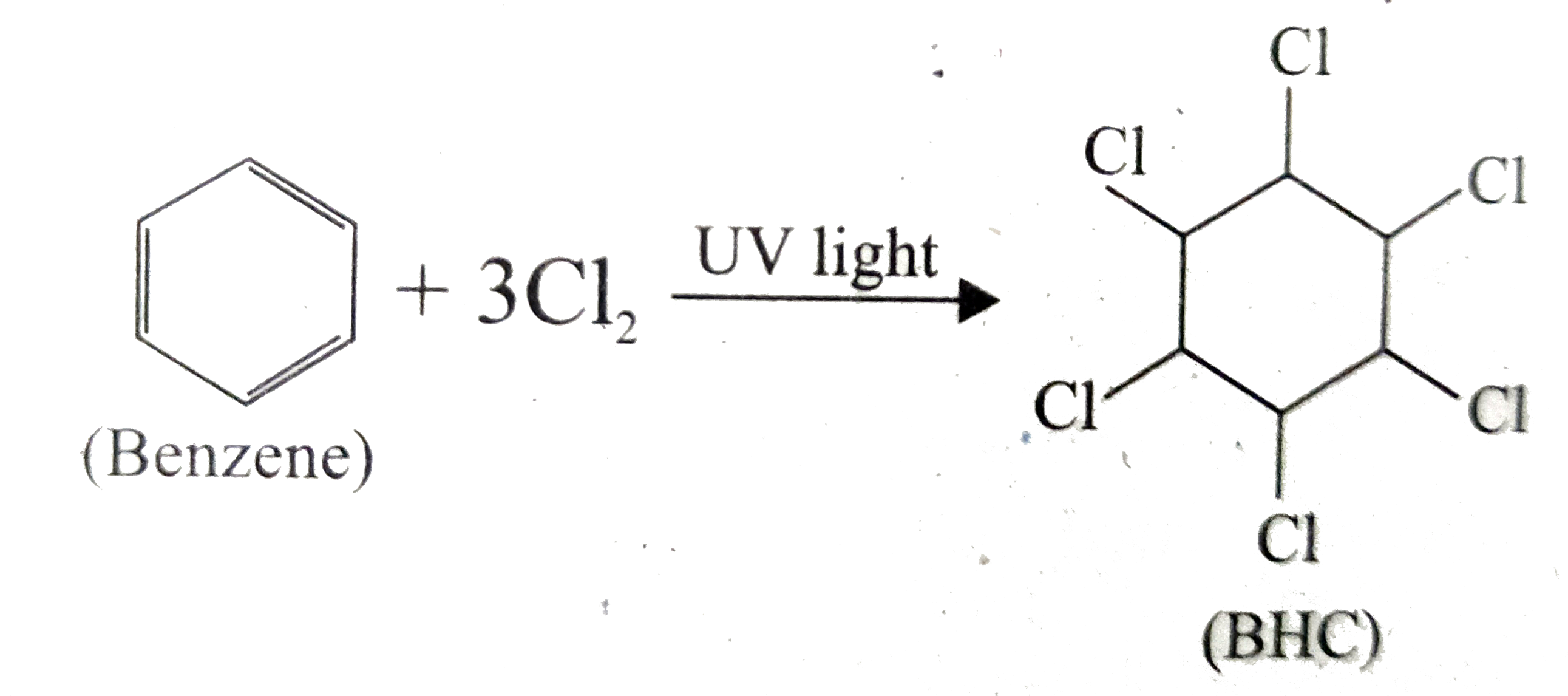 (iii) BHC is a powerful insecticide. |
|
| 14. |
When aqueous solution of borax isacidified with hydrochloric acid,a white crystalline solid is formed which is soapy to touch . Is thissolidacidic or basic in nature ? Explain. |
|
Answer» Solution :When an aqueoussolution of borax is acidifiedwith HCl,boric acid is formed. `underset("Borax")(Na_(2)B_(4)O_7)+2HCl + 5H_(2)O rarr2NaCl + underset("Boric acid")(4H_(3)BO_(3))`. Boric ACIDIS a white crystalline solid. Due to its planar layered structure , it is soapy to touch . it is not a protonic acidsince it does not ionize in aqueous solution to liberate `H^(+)` ions. HOWEVER,due to SMALL size of boron and presenceof only sixelectrons in its VALENCE shell, `B(OH)_(3)` accepts a pair of electrons from OXYGENATOM of a molecule of `H_(2)O`releasing a proton. Thus, it acts as Lewis acid. 
|
|
| 15. |
The variation of boiling points of hydrogen halides is in tha order HF gt HI gt HBr gt HCl . What explains the higher boiling point of hydrogen fluoride ? |
|
Answer» There is strong hydrogen BONDING between HF molecules due to high elecrtonegatively of fluorine. Hence , the boiling point of HF is abnormally high. Boiling points of other hydrogen halides GRADUALLY increase from HCl to HI due to increase in the size of halogen atom from Cl to I whihc results in the increase of van Waal forces . |
|
| 16. |
Which of the following is most soluble ? |
|
Answer» ` Bi_2S_3 (K_(sp) =1xx 10^(-17))` `Bi_2S_3 : S = 5 sqrt(( 1xx 10 ^(-17))/( 108) ) , MnS: S =sqrt( 7xx 10 ^(-16))` ` CuS: S =sqrt(8xx 10 ^(-37) ) , Ag_2 S : S =3sqrt( ( 6 xx 1 0 ^(-51))/( 4))` |
|
| 17. |
Which one of the following species does not exist under normal conditions ? |
|
Answer» `Li_(2)` |
|
| 18. |
The secondary precursors of photochemical smog are : |
|
Answer» `SO_(2)` and `NO_(2)` |
|
| 19. |
What will be the wavelength of a ball of mass 0.1 kg moving with a velocity of 10 ms^(-1) ? |
| Answer» Solution :ACCORDING to de Broglie equation, `lamda = (h)/(MV) = (6.626 XX 10^(-34) kg m^(2) s^(-1))/((0.1kg) (10 MS^(-1))) = 6.626 xx 10^(-34) m` | |
| 20. |
What is the effect of introducing an alkyl group on the stability of carbocation? |
| Answer» SOLUTION :It INCREASES because of HYPER CONJUGATION EFFECT. | |
| 21. |
What are Freons ? Discuss their uses and environmental effects. |
|
Answer» Solution :Freons are the chlorofluoro derivatives of methane and ethane. Freon is represented as Freon - cba Where, c = number of carbon atoms, b = number of hydrogen atoms, a total number of fluorine atoms. `CF_(2)Cl_(2)""c=1-1=0` `H=0+1=1` `F=2` So Freom - 12 is `CF_(2)Cl_(2)` Uses of Freons (i) Freons are used as REFRIGERANTS in refrigerators and air conditioners. (ii) It is used as a propellents for aerosols and foams. (iii) It is used as propellant for foams to spray out deodroants, shaving creams and insecticides. Environmental effects of Freons (i) Freon gas is a very poerful greenhouse gas which mean that it traps the heat normally RADIATED from the earth out into the space. This causes the earth.s temperature to increase resulting in rising sea levels, droughts, stronger storms, flash floods and a host of other very unpleasant effect. (ii) As freon MOVES throughout the air, its chemical ingredients causes DEPLETION of ozone layer. Depletion of ozone increase the amount of ultraviolet radiations that reaches the EARTHS surface, redulting in serious risk to human health. high levels of ozone, in turn, causes respiratory problems and can also kill plants. |
|
| 22. |
Which one/ones of the following reactions will yield 2-propanol ? Choose the right asnwer from (A), (B), (C) and (D) I. CH_(2) = CH-CH_(3)+H_(2)O overset(H^(+))rarr II. CH_(3)-CHO underset((ii) H_(2)O)overset((i) CH_(3)MgI)rarr III. CH_(2)O underset((ii) H_(2)O)overset((i) C_(2)H_(5)MgI)rarr IV. CH_(3)=CH-CH_(3) overset("Neutral" KMnO_(4))rarr |
|
Answer» I and II `CH_(2)=CH-CH_(3)+H_(2)O underset("M.Rule")overset(H^(+))rarr CH_(3)-overset(OH)overset(|)(C)-CH_(3)` `CH_(3)-CHO underset((ii) H_(2)O //H^(+))overset((i) CH_(3)MgI)rarr CH_(3)-overset(OH)overset(|)(C)-CH_(3)`. |
|
| 23. |
Which postulate of kinetic theory are invalid at low temperature of high pressure? |
| Answer» Solution :If the collision are non-elastic, there WOULD be constant LOSS of energy. This would cause MOLECULAR motion to slow down and ultimately settle down at the bottom of the container like precipitate. | |
| 24. |
whichof thefollowingrepresentsahomologousseries ? |
|
Answer»
|
|
| 25. |
Which of the following is optically active? |
| Answer» Solution :2-Methylpentane | |
| 26. |
The self ionization of water is endothermic. While increase the temp. than what is the effect on pH and value of K_w ? |
|
Answer» Solution :`2H_2O_((l)) + "Heat" hArr H_3O_((aq))^(+)+OH_((aq))^(-)` Then increase the TEMPERATURE and according to Le-Chatelier.s principle the reaction will be forward and increase products and `K_w` increase. The VALUE of pH is LESS than 7. At higher temperature the pOH of water is high. |
|
| 27. |
What are electron deficient compounds ? Are BCl_(3)" and "SiCl_(4) electron deficient species ? Explain. |
| Answer» SOLUTION :The species in which the central atom possesses an incomplete octet (i.e., does not have 8 electrons in its VALENCE shell) are called electron DEFICIENT compounds. In `BCl_3`, the central BORON atom has only six electrons. Therefore, it is an electron deficient species. In `SiCl_4` the central silicon atom has eight electrons. Therefore, in accordance to the above definition, it is not an electron deficient species. | |
| 28. |
Which of the following hydrated salts will not become anhydrous on heating? |
|
Answer» `MgCI_(2).6H_(2)O` |
|
| 29. |
What is hybridization ? Give example. What is the meaning of hybridization? |
|
Answer» Solution :ln order to explain the characteristic geometrical shapes of polyatomic molecules like `CH_(4), NH_(3)`and `H_(2)O` etc. Pauling introduced the concept of hybridization. Defutition of Pauling : According to him the atomic ORBITALS combine to form new set of equivalent orbitals known as hybrid orbitals. Unlike PURE orbitals, the hybrid orbitals are used in bond formation. Definition of hybridization : The process of intermixing of the orbitals of slighting different engorging so as to redistribute their on orgies RESULTING in the formation of new set of orbitals of equivalent energies and SHAPE known as hybridization. e.g. : One 2s orbital and there 2p orbitals of carbon mix & form equivalent four sp3 hybrid orbitals. |
|
| 30. |
What are organic reduction reactions? Give an example. |
|
Answer» SOLUTION :Most of the reduction reaction of ORGANIC COMPOUNDS involves GAIN of hydrogen or LOSS ofoxygen. Example: 
|
|
| 31. |
Which one of the following statements is not true? |
|
Answer» pH of drinkingg water should be between 5.5-9.5 |
|
| 32. |
Which of the following functional group is in CH_(3)(CH_(2))_(2)COCl? |
|
Answer» Aldhyde |
|
| 33. |
What is electronegativity? How is this useful in understanding the nature of elements? |
|
Answer» |
|
| 34. |
Which of the following is a mono-atomic molecule? |
|
Answer» Hydrogen |
|
| 35. |
The relative reactivity of 1^@,2^@,3^@ hydrogen's towards chlorination is 1:3.8:5. Calculate the percentages of all monochlorinated products obtained from 2-methylbutane. |
|
Answer» Solution :2-Metylbutane has 4 different sets of equivalent HYDROGEN atoms marked as 'a', 'b' , 'c' and 'd' `oversetaCH_3-oversetoverset(.^aCH_3)|(.^BCH)-oversetcCH_2-oversetdCH_3` 2-Methylbutane and HENCE gives four different types of monochlorinated products . These are : 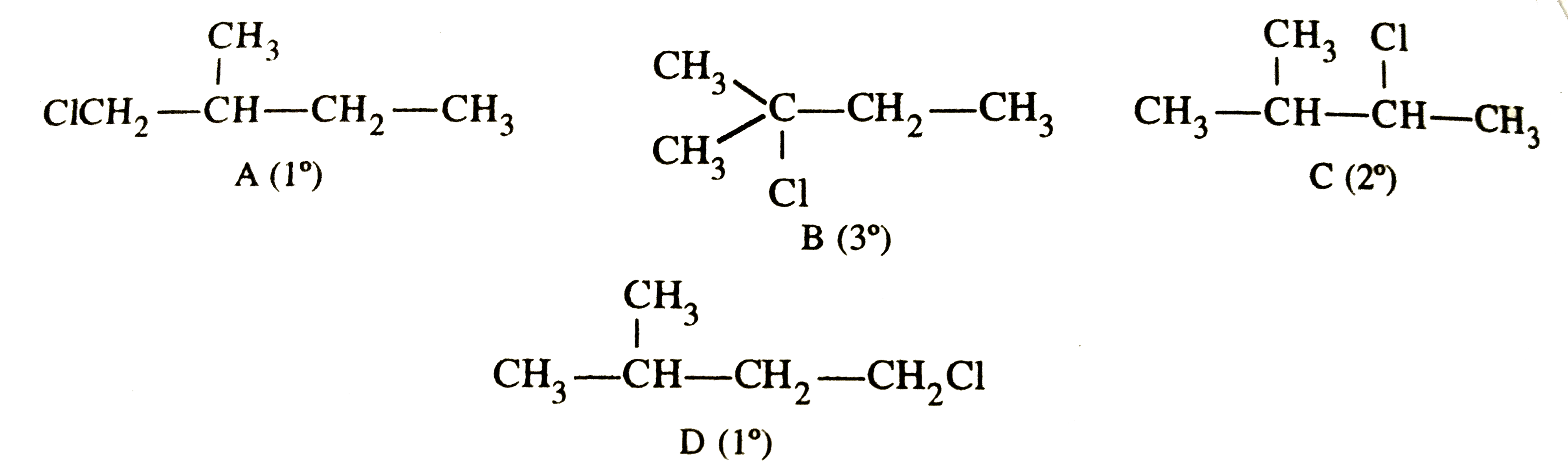 Relative number of equivalent hydrogens , a:b:c :d =6:1:2:3 Relative amounts of A, B, C and D = No. of hydrogen x relative REACTIVITY `{:("Relative amounts :",A(1^@),B(3^@), C (2^@),D (1^@)),(,6xx1=6,1xx5=5,2xx3.8=7.6,3xx1=3):}` Total amount of monohaloginated COMPOUNDS = 6+5+7.6+3=21.6 % A =`(6xx100)/21.6`=27.72 , %B=`5/21.6xx100`=23.15, %C=`7.6/21.6xx100=35.22` , %D=`3/21.6xx100=13.85` |
|
| 37. |
The role of 'Molybdenum' in Uaber's synthesis is |
|
Answer» A POSITIVE CATALYST |
|
| 38. |
What happen if HCl gas passed in NaCl solution ? |
|
Answer» Solution :(i)`NaCl_((s)) hArr Na_((aq))^(+) + Cl_((aq))^(-)` saturated solution (ii)`HCl_((g)) overset(H_2O)to H_((aq))^(+) + Cl_((aq))^(-)` Here, `Cl^-` is COMMON ION so concentration is increase so reaction TAKE place in reverse DIRECTION and solid precipitation of NaCl take place. So with HIGH purity solid NaCl obtained and impurities of `Na_2SO_4` and `MgSO_4` removed. |
|
| 39. |
What is meant by nodal surface ? |
| Answer» Solution :(I) The REGION where there is probability DENSITY function reduces to ZERO is called nodal surface or a radial surface. ltbr GT (ii) For ns orbital.(n-1)NODES are found in it. | |
| 40. |
Write the conjugate acids for the bronsted bases. NH_2^(-) , NH_3 and HCOO^- |
| Answer» SOLUTION :`NH_3, NH_4^(+) , HCOOH` | |
| 41. |
Which one of the following does NOT involve coagulation:- |
|
Answer» Formation of delta regions |
|
| 42. |
What type of structures are exhibited by (a) BaCl_(2) (b) Na_(2)O |
| Answer» SOLUTION :FLUORITE TYPE STRUCTURE (B) Antifluorite type structure. | |
| 43. |
Which of the following is the least stable resonance structure of CH_(2)=CH-CHO |
|
Answer»
`overset(Ɵ)(C)H_2 - CH - overset( overset(OPLUS)O) overset(|)CH` is the least stable one since more EN atom, O is carrying positive charge. |
|
| 44. |
Whichof the following is a homongenous mixture? |
|
Answer» Milk |
|
| 45. |
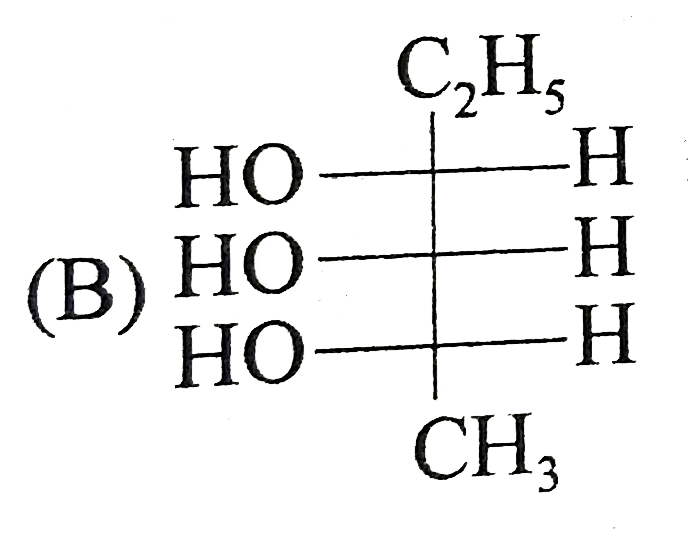
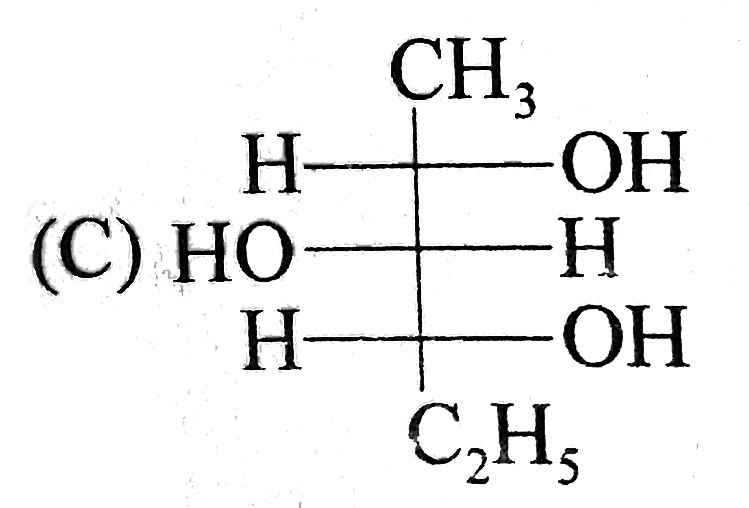
Which of the following is/are the correct Fischer projection of the followings |
|
Answer»
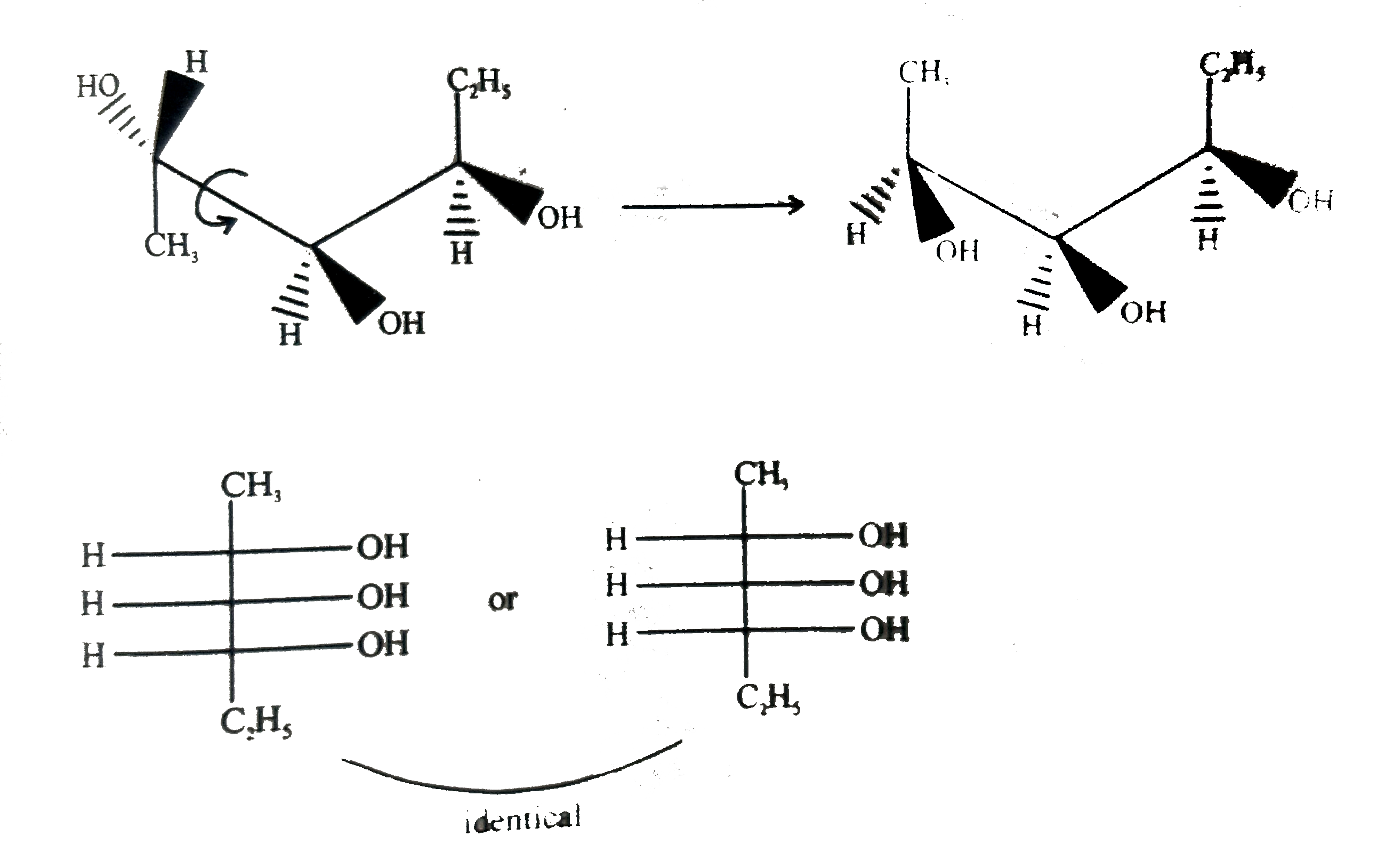
|
|
| 46. |
The substituents which are deactivating but ortho para directing in aromatic substitutions are -NO , -CHO, -F , -Cl, -Br , -CH = CHCHO, - C_6H_5, - COCl, -NH_3 . |
| Answer» SOLUTION :AROMATIC SUBSTITUTIONS | |
| 47. |
What will be the molarity of a solution, which contains 5.85g of NaCl_((s))per 500 mL ? |
|
Answer» 4 mol `L^(-1)` Molarity `=("weight" xx 1000)/("molecular weight" xx "volume (mL)")` `=(5.85xx1000)/(58.5xx500) = 0.2 "mol "L^(-1)` Note : Molarity of solution depends UPON the temperature because volume of solution is CHANGE with temerature . |
|
| 48. |
Which of the following salts is the most basic in aqueous solution? |
|
Answer» `Al(CN)_3` 
|
|
| 49. |
Which of the following pairs can be cited as an example to illustrate the law of multiple proportion? |
|
Answer» `Na_(2)O,K_(2)O` |
|
| 50. |
Which is used forending charge on colloidal solution? |
|
Answer» Electrons |
|