Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Which mineral is known as baryte ? |
|
Answer» BARIUM sulphide |
|
| 2. |
What is the hybrid state of BeCl_(2) in the vapour state ? What will be the change in hybrid state of BeCl_(2) in the solid state ? |
| Answer» Solution :In the vapour state at high temperature (1200 K) , Be is SP-hybridized and hence `BeCl_(2)` in linear . In the solid state , `BeCl_(2)` is polymeric. In this structure , each Be has two covalent and two coordinate BONDS . Therefore , in the solid state . Be has `sp^(3)`-HYBRIDIZATION . | |
| 3. |
Which of the following ionization energy values for Calcium show a sudden increase ? |
| Answer» ANSWER :A | |
| 4. |
What is inert pair effect ? |
| Answer» Solution :In case of elements of 4th, 5th and 6th period of p-block elements which COME after d-block elements, the electrons present in the INTERVENING d-or-and-f-orbitals do not shield the s-electrons of the valence shell effectively. As a RESULT, `ns^(2)`-electrons reamain more tightly held by the nucleus and HENCE do not participate in bonding. This is called inert PAIR effect. | |
| 5. |
The values of K_(sp) of two sparingly soluble salts NI(OH)_2 and AgCN are 2.0xx10^(-15)and 6xx10^(-17) respectively . Which salt is more soluble ? Explain . |
|
Answer» Solution :Value of `K_(sp)` is more than solubility is more according to this , Solubility of `Ni(OH)_2 gt` solubility of AGCN. Suppose , solubility of `Ni(OH)_2=S_1` solubility of AgCN = `S_2` CALCULATION of solubility `S_1` of `Ni(OH)_2` : EQUILIBRIUM in concentrated solution of `Ni(OH)_2` . `{:(Ni(OH)_(2(s)) hArr , Ni_((aq))^(2+) + , 2OH_((aq))^(-)),("Molarity in solution:", S_1,2S_1):}` `therefore K_(sp) [Ni(OH)_2] = [Ni^(2+)] [OH^-]^2` `=(S_1)(2S_1)^2` `=(4S_1)^3=2.0xx10^(-15)` `therefore S=(2.0/4.0 xx 10^(-15) )^(1/3)= (0.5 xx 10^(-15) )^(1/3)` `= 0.7937 xx 10^(-5) "mol L"^(-1)` `=7.937xx10^(-6)` The calculation of solubility of `S_2` in AgCN : `{:(AgCN_((s)) hArr , Ag_((aq))^(+) +, CN_((aq))^(-)),("Molar concentration", S_2,S_2):}` `therefore K_(sp)=[Ag^+][CN^-]` `=(S_2)(S_2)=S^2 =6xx10^(-17) = 60XX10^(-18)` `therefore S_2=sqrt(60xx10^(-18))=7.745xx10^(-8)` Thus solubility of `Ni(OH)_2` is `S_1= 7.937xx10^(-6)` solubility of AgCN is `S_2= 7.745xx10^(-8)` The solubility of `Ni(OH)_2` is more and the solubility of AgCN is less than it. |
|
| 6. |
Write IUPAC name of following: (i) (CH_(3))_(4)C (b) [(CH_(3))_(2) CH] CH_(2) (c ) (CH_(3))_(2) CH CH_(2)C (CH_(3))_(3) (d) (C_(2)H_(5))_(4)C (e ) (CH_(3))_(2) CHCH_(2) CH(CH_(3)) CH_(2)CH(CH_(3))_(2) (f) (C_(2)H_(5))_(2) C(CH_(3)) CH (CH_(3))_(2)(g) (CH_(3)CH_(2))_(2) CH- C(CH_(3))_(2)- CH_(2)CH_(2)CH_(3) (h) (CH_(3))_(3)C C(CH_(3))(C_(2)H_(5)) CH_(2)CH_(2)CH_(3) |
| Answer» SOLUTION :(a) 2, 2-dimethylpropane (b) 2, 4-dimethylpentane (C ) 2, 4, 4-trimethylpentane (d) 3, 3-dimethylpentane (e ) 2, 4, 6-trimethylpentane (F ) 3-ethyl-2, 3-dimethylpentane (g) 3-ethyl -4, 4-dimethylheptane (h) 3-ethyl-2, 2, 3-trimethylhexane | |
| 7. |
What is the two-dimensional coordination number of a molecule is square close packed layer ? |
| Answer» Solution :In The two-dimesional square CLOSE PACKED LAYER,the atom touches 4 nearest neighouring atoms. HENCE,its corrdination number = 4. | |
| 8. |
Which of the following is saturated hydrocarbon ? |
|
Answer» Ethene |
|
| 9. |
There is 1 mol liquid (molar volume 100 ml) in an adiabatic container initial, pressure being 1 bar. Now the pressure is steeply increased to 100 bar ,and the volume decreased by 1 mL under constant pressure of 100 bar. Calculate DeltaH "and" DeltaE. [Given 1 bar = 10^(5) N//m^(2)] |
|
Answer» `DeltaE=0 J, DeltaHne0 J` |
|
| 10. |
Which of the following is redox reaction |
|
Answer» `N_(2)O_5 +H_2O rarr 2HNO_3` |
|
| 11. |
Write separate equations for the oxidation and reduction reactioins occurring in the following redox reaction. 2FE+2HCltoFeCl_(2)+H_(2) |
|
Answer» Solution :`2Fe+2HCltoFeCl_(2)+H_(2)` `FetoFe^(2+)+2E^(-)` `2H^(+)+2e^(-)toH_(2)` |
|
| 12. |
Write the electronic configuration of alkali metals ""_(3)Li,_(11)Na, ""_(19)K,""_(37)Rb,""_(55)Cs and ""_(87)Fr |
Answer» SOLUTION :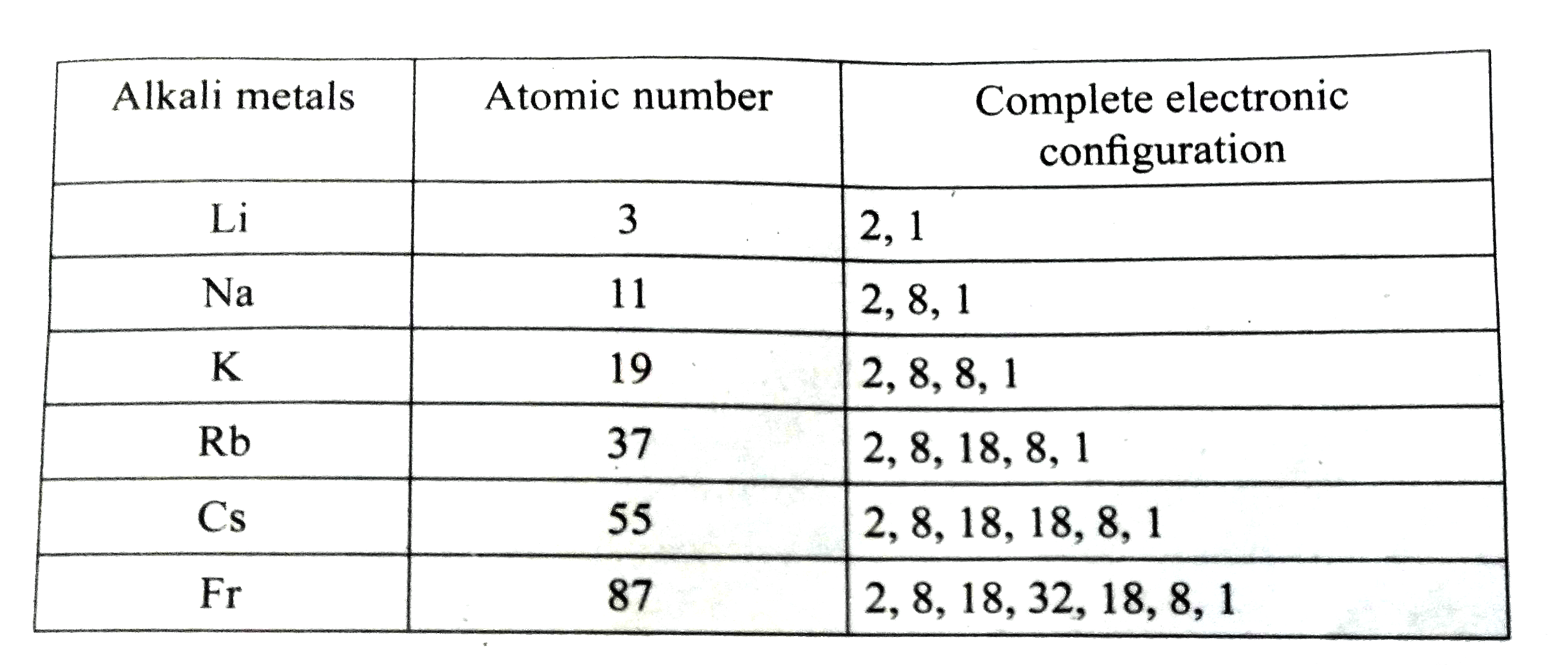
|
|
| 13. |
Using s, p, d, f notations, describe the orbital with the following quantum numbers: (a) n = 2, l = 1 (b) n =4, l =0 (c) n = 5, l = 3 (d) n = 3, l = 2 |
|
Answer» Solution :(a) n = 2, l = 1 means 2p orbital (B) n = 4, l =0 means 4s orbital (c) n = 5, l = 3 means 5F ORBITALS (d) n = 3, l = 2 means 3d orbital |
|
| 14. |
This compound shows : |
|
Answer» geometrical ISOMERISM |
|
| 15. |
Which is correct order of bond length ? |
|
Answer» `O_(2) LT O_(3) lt O_(2)^(2-)` `O_(3)` (ozone ) has V - shape it BOND length O - O`rarr ` 1.278 Å Bond order `prop (1)/("Bond length")` So, `O_(2) lt O_(2)^(2-)` 
|
|
| 16. |
Whichelectronicconfigurationof an elementhas abnormallyhighdifferencebetweensecondandthirdionizationenergy ? |
|
Answer» `1s^(2)2s^(2)2P^(6)3S^(1)` |
|
| 17. |
which orbital does not have directional characteristic ? |
| Answer» SOLUTION :s-orbital | |
| 18. |
Which one of the following is a hard base ? |
|
Answer» `AG^(+)` |
|
| 19. |
Two elements X (at.mass 16) ard Y (at. mass 14) combine to form compounds A, which combine with a fixed mass of X in A, B and C is 1:3:5. If 32 parts by mas of X combines with 84 parts by mass of Y in B, then in C 16 parts by mass of X will combine with |
|
Answer» 14 PARTS by MASS of Y |
|
| 20. |
What is the state of hybridisation of carbon in graphite ? |
| Answer» SOLUTION :Graphite : Each CARBON atom in graphite is `sp^2` hybridised and is BOUND to three other carbon ATOMS. | |
| 21. |
Two vessels are connected by a valve of negligible volume. One container, A has 2.8 g N_2 at T_1K. The other container, B is completely evacuated. The container A is heated to T_1K, while container B is maintained at (T_2)/3 K. Volume of A is half that of B. If the valve is opened, then |
|
Answer» 6/70 moles of `N_2` will be present in B `((0.1 - x)RT_2)/(v//2) = (x RT_3)/V` |
|
| 22. |
Which of the following molecules has sufficient C bar(---)O double bond character? |
|
Answer» Phenol |
|
| 23. |
What is the energy in joules required to shift the electron of the hydrogen atom from the first Bohr orbit to the fifth Bohr orbit and what is the wavelength of the light emitted when the electron returns to the ground state ? The ground state energy is -2.18xx10^(-11) ergs. |
|
Answer» SOLUTION :As ground state electronic ENERGY is `-2.18xx10^(-11)` ergs, this means that `E_(n)=-(2.18xx10^(-11))/n^(2)` ergs `DeltaE=E_(5)-E_(1)=2.18xx10^(-11) (1/1^(2)-1/5^(2))=2.18xx10^(-11) (24/25)=2.09xx10^(-11)` ergs `=2.09xx10^(-18)J (1 erg =10^(-7) J)` When electron returns to ground state (i.E., to `n=1`), energy emitted `=2.09xx10^(-11)` ergs. As `E=hv=hc/LAMBDA or lambda=(hc)/E=((6.626xx10^(-27)" erg sec")(3xx10^(10)" cm s"^(-1)))/(2.09xx10^(-11) ergs)` `=9.51xx10^(-6) cm=951xx10^(-8) cm=951 Å` |
|
| 24. |
What are f-block elements? How many series are there? Why they are called f-block elements? |
| Answer» SOLUTION :• The elements, WHOSE LAST electron enters into the f-orbital of the ante-penultimate shell (n-2) are having similar properties are called f-block elements. In these elements (n 2)f orbitas are being FILLED progressively. • The TWO rows of elements placed at the bottom of the neriodic table. They are lanthanid and actinides. | |
| 26. |
What is W ? |
| Answer» SOLUTION :W iswavefunctionis itmathermaticaloperator. It DOESNOT havephysicalmeaning . | |
| 27. |
Wurtzite has ___________ formula units per unit cell whereas zinc blende has ______ formula units per unit cell. |
|
Answer» |
|
| 28. |
When boron atom undergoes sp^3 hybridization |
|
Answer» all the four `sp^3` orbitals contain one electron in each of them |
|
| 29. |
Which of the following element does not form compound with coordination number 5. |
|
Answer» Si |
|
| 30. |
What do you understand by the form non stoichiometric hydrides ? Do you expect this type of the hydrides to be formed by alkali metals ? Justify your answer. |
|
Answer» Solution :The Metallic hydride are formed by many d -block and f-block elements. However, the metals of group 7, and 9 do not form hydride. Even from group 6, only chromium forms CrH. These hydrides CONDUCT heat and electricity though not as efficiently as their parent metals do. Unlike saline hydrides, they are almost always non-stoichiometric, being deficient in hydrogen. For example : `LaH_(2.87), YbH_(2.55),TiH_(1.5-1.8),ZrH_(1.3-1.75), VH_(0.56), NiH_(0.6-0.7), PdH_(0.6-0.8)` etc. In such hydrides, the law of constant composition does not hold GOOD. In these hydrides, hydrogen occupies interstitial space in the metal lattice producing distortion without any change in its type. Consequently, they were termed as interstitial hydrides. The except for hydrides of Ni, Pd, Ce and Ac, other hydrides of this CLASS have lattice different from that of the parent metal. The property of absorption of hydrogen on transition metals is WIDELY used in catalytic reduction / hydrogenation reactions for the preparation of large number of compounds. Some of the metals (e.g., Pd, Pt) can accommodate a very large volume of hydrogen and, therefore, can be used as its storage media. This property has high POTENTIAL for hydrogen storage and as a source of energy. |
|
| 31. |
Which of the following gases have same rate of diffusion at constant P and T ? |
|
Answer» `C_2H_6` |
|
| 32. |
What is the product formed when acetylene reacts with excess of hypochlorous acid ? |
|
Answer» `CH_3COCl` |
|
| 33. |
Which one of the alkaline earth metals carbonate is thermally most and last stalbe. Why? |
| Answer» Solution :`BaCO_(3)` is thermally most stable DUE to greater inoic CHARACTER and high LATTICE energy whereas `BaCO_(3)` is thermally LEAST stable because it is COVALENT and has less lattice energy. | |
| 34. |
what is the electronic configuration of Cu^(2+) (Z=29) of least position |
|
Answer» `[AR]4s^1 3d^8` GROUND state of `Cu^29=1s^2 2s^2 2p^6 3s^2 3p^6 3d^10 4s^1` `Cu^(2+)=1s^2 , 2s^2 2p^6 , 3s^2 3p^6 3d^9` |
|
| 35. |
What is the temperature in the rotary Kiln used for the manufacture of cement? |
| Answer» SOLUTION :1670 -1870 K | |
| 36. |
Why can dilute solutions of hydrogen peroxide not be concentrated by heating. How can be concentrated solutions of hydrogen peroxide be obtained ? |
|
Answer» SOLUTION :`H_(2)O_(2)` cannot be concentrated simply by heating since it decomposes much below its b.p. to give `H_(2)O` and `O_(2)` `"" 2H_(2)O_(2) to 2H_(2)O+O_(2)` Therefore, concentration of `H_(2)O_(2)` is CARRIED out in a NUMBER of stages, |
|
| 37. |
Which of the following reaction is incorrect? |
|
Answer» `BF_(3)(G) + F^(-)(AQ) rarr BF_(4)^(-)` |
|
| 38. |
Which of the following reaction takes place when a mixture of concentrated HNO_3 and H_2SO_4 reacts on benzene at 300 K. |
|
Answer» Sulphonation |
|
| 40. |
Van dar Waal's equation explains the behaviour of rea gases. What does the van der Waal's constant 'a' indicate? |
| Answer» Solution :The van der Waal.s CONSTANT .a. is a measure of the magnitude of intermolecular attractive FORCES in the GAS. | |
| 41. |
What would be the IUPAC name for an element with atomic number 222 ? |
|
Answer» bibibiium |
|
| 42. |
What are substitution reaction? |
|
Answer» Solution :In this REACTION an atom or a group of atoms ATTACHED to a carbon atom is replaced by a new atom or a group of atoms. For EXAMPLE , ` underset("Bromomethane") (CH_(3)) overset ("aq.OH") to underset("Methanol")(CH_(3)OH+ Br^(oplus))` Here, -Br is replaced by -OH group. |
|
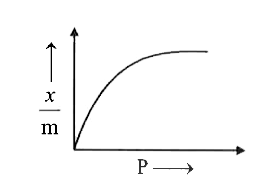
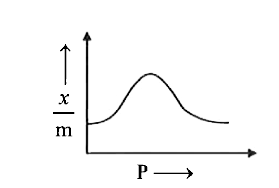
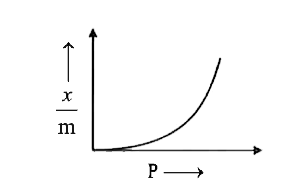
| 43. |
Which of the following graph CORRECTLY shows the variation of (x)/(m) with P according to Freundlich equation? |
|
Answer»
|
|
| 44. |
Which of the following molecules constants both polar and non-polar bonds- |
|
Answer» `NH_(4)CL` |
|
| 45. |
Which of the following molecule have least bond order ? O_(2) , O_(2)^(+) and O_(2)^(-) ? |
Answer» SOLUTION :`THEREFORE O_(2)^(-)` has least bond order. 
|
|
| 46. |
Write the structures of the following compounds : (i) But-2-enoic acid (ii) But-1-en-3-yne (iii) 3, 4, 4-Trimethylhex-1-yne |
|
Answer» Solution :(i) `CH_(3)-CH=CH-COOH` (II) `CH equiv C-CH=CH_(2)` (iii) `CH_(3)-CH_(2)-underset(CH_(3))underset(|)OVERSET(CH_(3))overset(|)(C)-underset(CH_(3))underset(|)(CH)-C equiv CH` |
|
| 47. |
What is quick lime? How is it prepared? |
| Answer» SOLUTION :QUICK LIME is calcium oxide. It is prepared by HEATING LIMESTONE. | |
| 48. |
Which of the following represent homogeneous catalysis? |
|
Answer» OIL`+H_(2) overset(Ni)rarr "SATURATED FAT "` |
|
| 49. |
Which of the following is not a general characteristic of equilibria involving physical processes ? |
|
Answer» Equilibrium is possible only in a closed system at a given temperature. |
|
| 50. |
The salt that is added to table salt to make it flow freely in rainy season is |
|
Answer» KCl |
|