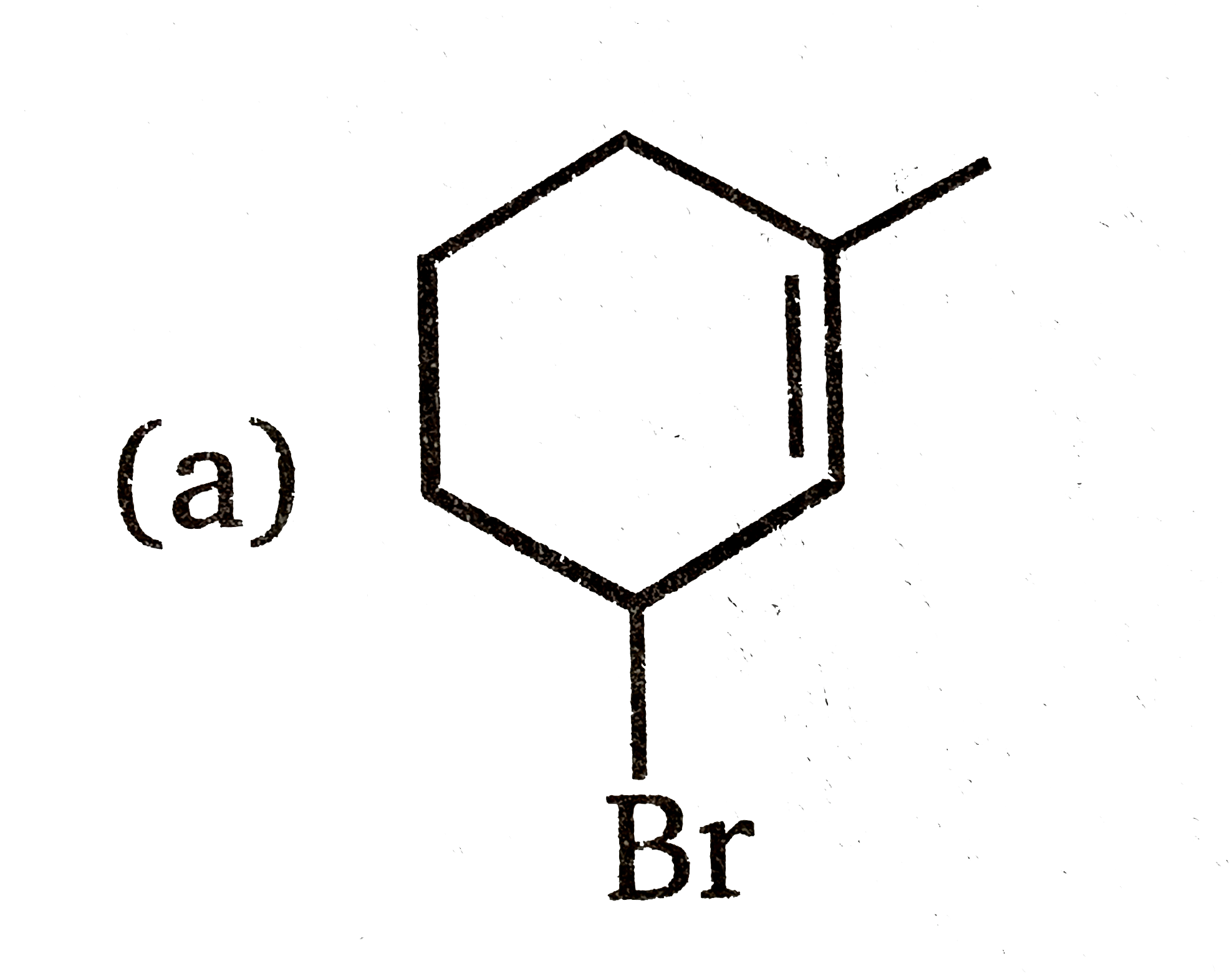Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
What do you mean by syaing that energy of the electron is quantized ? |
| Answer» Solution :This MEANS that the electrons in an ATOM have only definite VALUES of ENERGIES | |
| 2. |
Why are bonding molecular orbitals more stable than anti bonding molecular orbitals? |
| Answer» Solution :The bonding ORBITALS are at a lower energy than the antibonding orbitals, so they are the FIRST to FILL up. They are more stable | |
| 3. |
Which of the following compounds are formed when BCI_(3) is treated with water? |
|
Answer» `H_(3)BO_(3)` |
|
| 4. |
Which of the following is used to produce smoke screens ? |
|
Answer» <P>calcium phosphide Phosphate `(PH_(3))` so formed spontaneously catches fire to give `P_(2)O_(5)`. `2PH_(3)+4O_(2) rarr P_(2)O_(5)+3H_(2)O` `P_(2)O_(5)` forms a white DENSE SMOKE screen. |
|
| 5. |
VDs of N_(2)O_(4 (g)) at equilibrium at temperatures T_(1) & T_(2)K respectively are 40 & 30 . Ratio of total number of moles of components at equilibrium at T_(1) K to T_(2) K shall be |
| Answer» ANSWER :A | |
| 6. |
what is dipolemoment |
| Answer» Solution :If is the PRODUCT of the DISTANCES of CHARGE (positive or negative) CENTRES and distance between the centre. | |
| 7. |
Which one of the following is an example of disporoportionation reaction ? |
|
Answer» `3Cl_(2) (g) +6OH^(-) (aq) rarrClO_(3)^(-) (aq) +5CI^(-) (aq) +3H_(2)O(1)` `rarr ClO_(3)^(-) (aq) +5CI^(-)(aq) +3H_(2)O(l)` |
|
| 8. |
The value of planck constant is |
|
Answer» `6.62 xx 10^(-34)` Joule-second |
|
| 9. |
What is the coordination number of sodium in Na_(2)O ? |
|
Answer» 6 `THEREFORE`Co-ordination number of `Na^(+)`ion =4 |
|
| 10. |
Which acid has lowest pK_(a) |
|
Answer» <P>`p-`Methoxybenzoic acid |
|
| 11. |
Which of the following compound will not undergo nucleophilic substitution reaction ? |
|
Answer»
|
|
| 12. |
The relative adsorption of each component of the mixture is expressed in terms of |
|
Answer» ADSORPTION FACTOR |
|
| 13. |
Which one of the following always be negative? |
|
Answer» 1) HEAT of reaction |
|
| 14. |
Write limitation of lewis concept and main postulates of VSEPR theory. |
|
Answer» SOLUTION :Lewis concept is unable to explain the shapes of molecules. Valence SHELL electron pair repulsion theory : This theory provides a simple procedure to predict the shape of covalent molecules. Sidgwick and Powell in 1940, proposed a simpl.e theory based on the repulsive interactions of the electron pairs in the valence shell of the atoms so, its known of VSEPR theory. The postulates of VSEPR Theory : The shape of a molecule DEPENDS upon the number of valence shell electron pairs (bonded or nonbonded) around the central atom. pairs of electrons in the valence shell repel one another since their electron clouds are negatively charged. These pairs of electrons tend to OCCUPY such position in space that minimise repulsion and thus maximise distance between them. The valence shell is taken as a sphere with the electron pairs localising on the spherical surface at maximum distance from one another. A multiple bond is treated as if it is a sing]e electron pair and the two or three electron pairs of a multiple bond are treated as a single super pair. Where two or more resonance structures can represent a molecule, the VSEPR model is applicable to any such structure. The repulsive INTERACTION of electron pairs decrease in the order : [Lone pair (lp) - Lone pair (lp)] `gt` [Lone pair (lp) - Bond pair (bp)] `gt` [Bond pair (bp) - Bond pair (bp) ] |
|
| 15. |
What is valence boned approach for the formation of covalent bond and a coordinate bond ? |
|
Answer» SOLUTION :A covalent bond is FORMED by the overlap of half FILLED atomic ORBITALS whereas a coordinate bond formed by the oversap of an empty ORBITAL with a fully-filled orbital. |
|
| 16. |
Which of the following has maximum number of molecules ? (C = 12, O = 16, N = 14, H = 1) |
|
Answer» 1 mole of `H_2O` gas (B) `(6xx10^(23)xx32)/(28) = 6.86xx10^(23)` ( C) `(6xx10^(23))/(22.4) xx2.24 = 6xx10^(22)` (d) `(6xx10^(23))/(44)xx22 = 3xx10^(23)` CORRECT ANSWER is (b). |
|
| 17. |
Write the IUPAC names of the products obtained by the ozonolysis of the following compouds :2-Ethyl but-l-ene |
Answer» SOLUTION :
|
|
| 18. |
What is the difference between ionic product and solubility product ? |
| Answer» SOLUTION :SOLUBILITY PRODUCT is the product of the MOLAR concentrations of the IONS in a saturated solution but ionic product is for any solution. | |
| 19. |
Which hybridization is found in NH_(3) and H_(2)O? |
| Answer» ANSWER :A | |
| 20. |
Write the equations for the preparation of 1-iodobutane from: (i) 1-butanol (ii) 1-chlorobutane (iii) but-1-ene |
Answer» SOLUTION :
|
|
| 21. |
Write the following equation in ionic form. MnO_(2)+4HCl to MnCl_(2)+2H_(2)O+Cl_(2) |
|
Answer» Solution :In this REACTION HCl and `MnCl_(2)` are ionic in NATURE. Writing these comounds in ionic from, `MnO_(2)+4H^(+)+4Cl^(-)=MN^(2+)+2Cl^(-)+2H_(2)O+Cl_(2)` `2Cl^(-)` ION are common on both sides, so these are cancelled. The desired ionic equation reduceds, to , `MnO_(2)+4H^(+)+2Cl^(-)=Mn^(2+)+2H_(2)O+Cl_(2)` |
|
| 22. |
Which of the following is an endothemic reaction? |
|
Answer» `N _(2 (g)) + 3 H _(2(g)) - 92 KJ to 2 NH _(3(g))` |
|
| 23. |
The reduction of 2-Butyne with H_2 in the presence of Pd/BaSO_4 - Quinoline and Na/liquid NH_3 gives |
|
Answer» trans-2 butene in both the CASES |
|
| 24. |
What is the hybridisation of central atom of (D)? |
|
Answer» `d^(2)SP^(3)` |
|
| 25. |
What is true about saline hydrides |
|
Answer» They are binary compounds of hydrogen and METALLIC ELEMENTS |
|
| 26. |
What would be the SI unit for the quantity pV^(2)T^(2)//n ? |
| Answer» Solution :`((N m^(-2))(m^(3))^(2)(K)^(2))/("mol")=N m^(4)K^(2)" mol"^(-1)` | |
| 27. |
Which of the following gas deviates least from ideal nature at room temperature and atmospheric pressure |
|
Answer» NITROGEN |
|
| 28. |
underset([S])([M(NH_(3))_(6)]^(2+)) to underset([S])(M(NH_(2))_(2))+xunderset((g))(NH_(3))+yunderset((g))(H_(2)) |
|
Answer» `x+y=4+1=5` |
|
| 29. |
Using the outcome fo previous example, arrange benzene, 1,3-cyclohexadiene, and cyclohexene in the order of decreasing rate of monohydrogenation. Strategy: Since the change in the number of moles is the same for each hydrogentaion,we assume that the DeltaS's (entropy changes) are about the same, and that the Delta_(h)H's are directly related to the DeltaH's. |
| Answer» Solution :The order of chlohexene `(-120 kJmol^(-1))gt1,3-`CYCLOHEXADIENE `(-112kJmol^(-1))GT` benzene `(+24kJmol^(-1))` | |
| 30. |
The solution which consumes [H^(+)] or [OH^(-)] or both simultaneously from externally added base in order to give negligible change in pH, is known as buffer solution. In general, the solution resists the change in pH. Buffer solution does not mean that there does not occur a pH change in pH. Buffer solution des not mean that there does not occur a pH change at all. It implies the pH change occurs but in neglibible amount. There are two types of buffer (i) Acidic buffer: it is a mixture of weak acid and its salt acid strong base. (ii) Basicbuffer : It is a mixture ofweak base and its salt with strong acid. To prepare a buffer of pH 8.26, amount of (NH_(4))_(2)OH solution [pK_(a)(NH_(4^(+)) = 9.26] |
|
Answer» `0.05`MOL |
|
| 31. |
Which one of the following indicates the correct order of atomic size? (1). C gt F gt Ne""(2) Be lt C lt F lt Ne (3). Be gt C gt F" "(4) F gt Ne gt Be gt C |
|
Answer» |
|
| 32. |
When a liquid evaporates, which is true about the signs of the enthalpy and entropy changes? |
|
Answer» `{:(Delta H,Delta S),(+,-):}` |
|
| 34. |
Which are two isotherm graph according to Boyle.s Law. |
|
Answer» SOLUTION :(i) Graph p `to` V is curve. (II) Graph of p `to(1)/(V)` is straight line PASSES through ORIGIN. |
|
| 35. |
Which of the following reactions produce the same alkane as major product ? |
|
Answer»

|
|
| 36. |
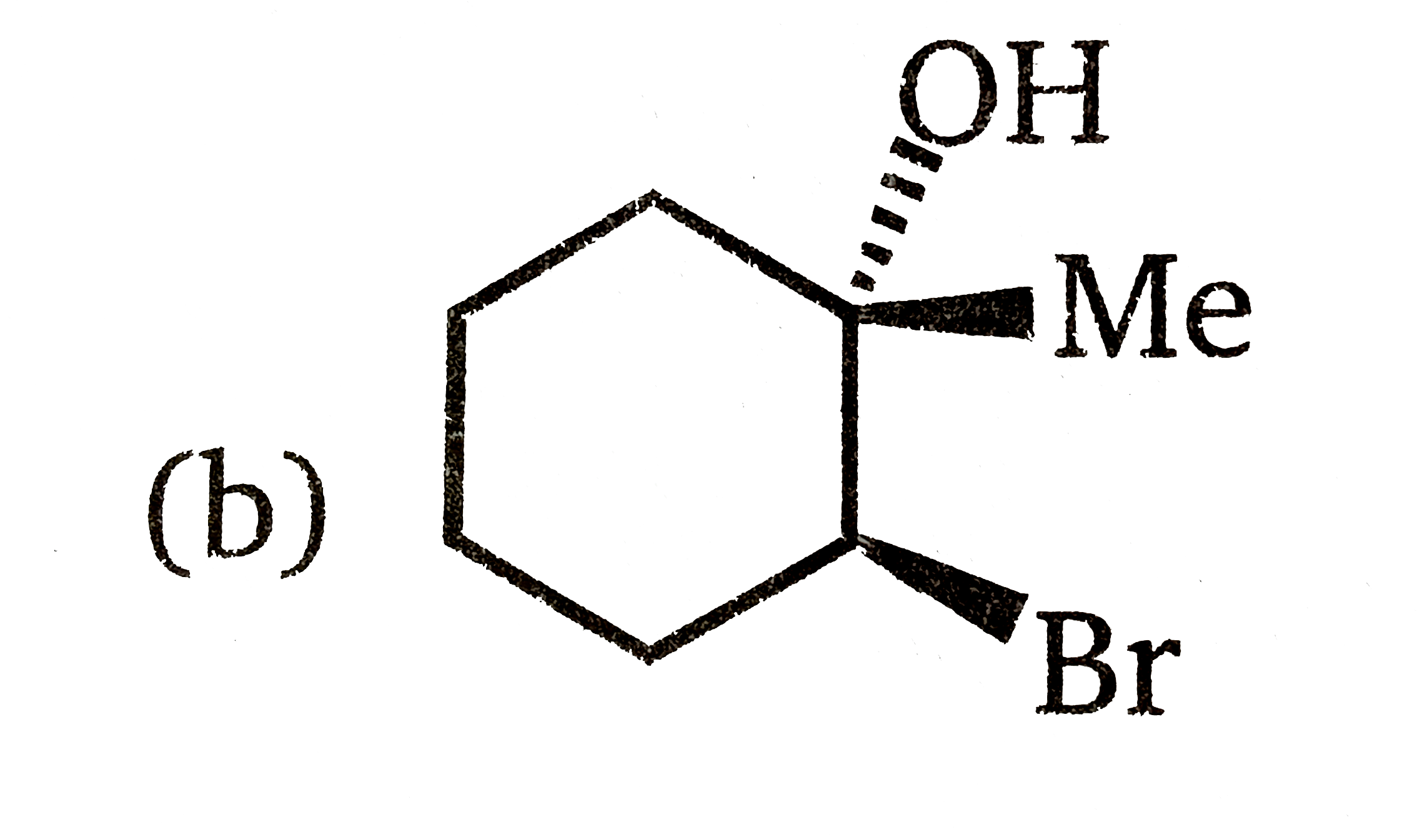
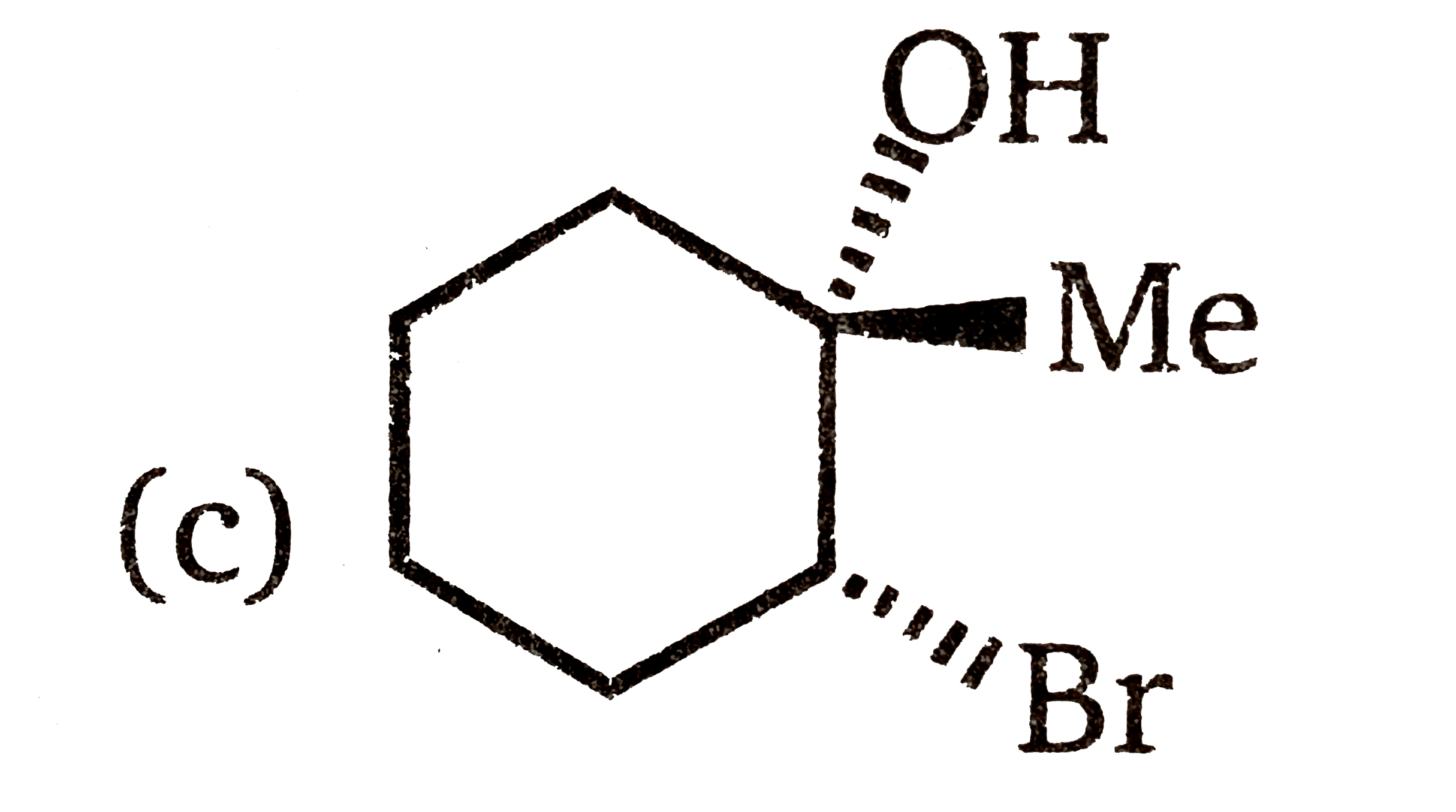
Which of the following is the best stereochemical representation when reaction between 1-methylcyclohexene and NBS react in aqueous dimethyl sulfoxide? |
|
Answer»
|
|
| 37. |
When gypsum is heated at 393K, the compound formed is CaSO_(4).XH_(2)O value of 6X is |
|
Answer» `x=1//2`, `6x=6xx(1)/(2)=3` |
|
| 38. |
What do you understand by the term mole? |
| Answer» Solution :The mole is defined as the amount of a substance which contains 6.023* 1023 PARTICLES such as ATOMS, molecules or IONS. It is represented by the SYMBOL n. | |
| 39. |
What type of non-idealities are exhibited by cyclohexane-ethanol and acetone-chloroform mixture ? Give reason for your answer. |
|
Answer» Solution :Ideal SOLUTIONS are those which obey Raoult.s law over extreme range of concentration. Ideal solution have another important PROPERTIES : (i) `Delta H_(mix) =0 (ii) Delta V_(mix) =0` Here-forces of attraction between A-A, B-B and A-B are of the same order. Non ideal solutions do not obey Raoult.s law over the entire range of concentration. `Delta H_(mix) NE 0 and Delta V _(mix) ne 0` Cyclohexane-ethanol mixture shows POSITIVE deviation from Raoult.s law because forces of attraction between cyclohexane and ethanol are less than in between puire cyclohexane as well as pure ethanol. Acetone-Chloroform mixture shows negative deviation from Raoult.s law because forces of attraction between acetone and chloroform are higher than that in between pure acetone and pure chloroform molecules. |
|
| 40. |
The treatment of CH_3MgX with CH_3C-=C-H produces |
|
Answer» a. CH_3 - CH = CH_2` |
|
| 41. |
The substance which added to the ore in order to remove impurities during smelting is called |
|
Answer» slag |
|
| 42. |
Three substances normally considered as primary pollutant are ______,______,and,____. |
|
Answer» |
|
| 43. |
Which of the following reactions is an example of use of water gas in the synthesis of other compounds / |
|
Answer» `CH_(4)(G) + H_(2)O(g) UNDERSET(Ni)overset(1270K)to CO(g) + H_(2)(g)` |
|
| 44. |
When 'n' moles of CO combines with (2n+1) moles of H_2 , the hydrocarbon formed is |
| Answer» ANSWER :B | |
| 45. |
Which of the followingg intermediates contains three paris of electrons in its valence shell? |
|
Answer» Carbocations |
|
| 46. |
Which of the following statement(s) is/are correct about red vapour gas D? |
|
Answer» D is `CrO_(2)CI_(2)` (C) `rarr HgO.Hg(NH_(2))I` (D) `rarr CrO_(2)CI_(2)`, (E) `rarr Na_(2)CrO_(4)` |
|
| 47. |
The solution which consumes [H^(+)] or [OH^(-)] or both simultaneously from externally added base in order to give negligible change in pH, is known as buffer solution. In general, the solution resists the change in pH. Buffer solution does not mean that there does not occur a pH change in pH. Buffer solution des not mean that there does not occur a pH change at all. It implies the pH change occurs but in neglibible amount. There are two types of buffer (i) Acidic buffer: it is a mixture of weak acid and its salt acid strong base. (ii) Basicbuffer : It is a mixture ofweak base and its salt with strong acid. Which of the following mixture will be a buffer solution when dissolved in 500 mL of water: |
|
Answer» `0.200` MOL of aniline and `0.200` mol of HCL |
|
| 48. |
What is called a Ideal gas ? Why ? |
|
Answer» Solution :A gas that follows Boyle.s law, Charle.s law and Avogadro law STRICTLY is called an ideal gas. Such a gas is hypothetical. It is assumed that intermolecular forces are not present between the MOLECULES of an ideal gas. When the inter molecular forces are NEGLIGIBLE in real gas it acts as an ideal gas and FOLLOW the gas rules. |
|
| 49. |
The reagent used for Friedel-Craft's reaction is : |
|
Answer» DRY ETHER |
|
| 50. |
What type of substances would make better permanent magnets , ferromagnetic or ferrimagnetic ? |
| Answer» Solution :FERROMAGNETIC SUBSTANCES would MAKE better permanent MAGNETS. | |